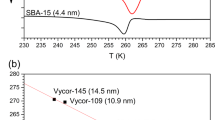
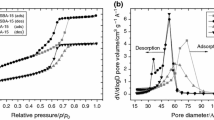
Abstract
In this work, water confined in silica and carbon nanopores has been examined. The purpose of this study is to describe the melting behaviour and structure of ice confined in silica nanopores, KIT-6 and ordered carbon nanopores, CMK-3, having pore diameters of 5.9 and 5.2 nm, respectively. To determine the melting temperature of ice inside the nanopores, we performed differential scanning calorimetry measurements of the systems studied. We found that the melting temperature of confined ice is reduced relative to the bulk melting point and this shift is 16 K for water confined in KIT-6 and 21 K for water confined in CMK-3. The structural properties of water at the interfaces were analysed by using the neutron diffraction method (ND). The ND measurements for all the systems studied, showed the features of both hexagonal ice, \(I_\mathrm{h}\), and cubic ice, \(I_\mathrm{c}\). However, we show that the ice confined in nanopores does not have a structure corresponding to the typical hexagonal form or the metastable cubic form. The ice confined in nanopores has a structure made up of cubic sequences interlaced with hexagonal sequences, which produce the stacking disordered ice (ice \(I_\mathrm{sd})\).





Similar content being viewed by others
References
Salzmann C G, Radaelli P G, Slater B and Finney J L 2011 Phys. Chem. Chem. Phys. 13 18468
Loerting T, Winkel K, Seidl M, Bauer M, Mitterdorfer C, Handle P H et al 2011 Phys. Chem. Chem. Phys. 13 8783
Palmer J C, Martelli F, Liu Y, Car R, Panagiotopoulos A Z and Debenedetti P G 2014 Nature 510 385
Johari G P and Andersson O 2015 J. Chem. Phys. 143 054505
Carr T H G, Shephard J J and Salzmann C G 2014 J. Phys. Chem. Lett. 5 2469
König H 1943 Z. Kristallogr. 105 279
Kuhs W F, Sippel C, Falenty A and Hansen T C 2012 Proc. Natl. Acad. Sci. 109 21259
Dowell L G and Rinfret A P 1960 Nature 188 1144
Mcmillan J A and Los S C 1965 Nature 206 806
Kuhs W F, Bliss D V and Finney J L 1987 J. Phys. Colloq. 48 631
Bertie J E, Calvert L D and Whalley E 1963 J. Chem. Phys. 38 840
Huang J and Bartell LS 1995 J. Chem. Phys. 99 3924
Bartell L S and Lennon P L 2009 J. Chem. Phys. 130 084303
Morishige K, Yasunaga H and Uematsu H 2009 J. Phys. Chem. C 113 3056
Morishige K and Uematsu H 2005 J. Chem. Phys. 122 44711
Steytler D C, Dore J C and Wright C J 1983 J. Phys. Chem. 87 2458
Hansen T C, Koza M M and Kuhs W F 2008 J. Phys. Condens. Matter 20 285104
Liu E, Dore J C, Webber J B W, Khushalani D, Jahnert S, Findenegg G H et al 2006 J. Phys. Condens. Matter 18 10009
Dunn M, Dore J C and Chieux P 1988 J. Cryst. Growth 92 233
Dore J, Webber B, Hartl M, Behrens P and Hansen T 2002 Physica A 314 501
Baker J M, Dore J C and Behrens P 1997 J. Chem. Phys. 101 6226
Handa Y P, Zakrzewski M and Fairbridge C 1992 J. Phys. Chem. 96 8594
Dore J 2000 Chem. Phys. 258 327
Takamuku T, Yamagami M, Wakita H, Masuda Y and Ya- maguchi T 1997 J. Phys. Chem. B 101 5730
Seyed-Yazdi J, Farman H, Dore J C, Webber J B W and Findenegg G H 2008 J. Phys. Condens. Matter 20 205108
Seyed-Yazdi J, Farman H, Dore J C, Webber J B W, Findenegg G H and Hansen T 2008 J. Phys. Condens. Matter 20 205107
Moore E B, Llave E, Welke K, Scherlis D A and Molinero V 2010 Phys. Chem. Chem. Phys. 12 4124
Fitzgerald R J 2013 Phys. Today 66 16
Amaya A J, Pathak H, Modak V P, Laksmono H, Loh N D, Sellberg J A et al 2017 J. Phys. Chem. Lett. 8 3216
Lupi L, Hudait A, Peters B, Grünwald M, Mullen R G, Nguyen A H et al 2017 Nature 551 218
Malkin T L, Murray B J, Salzmann C G, Molinero V, Pickering S J and Whale T F 2015 Phys. Chem. Chem. Phys. 17 60
Hansen T C, Koza M M, Lindner P and Kuhs W F 2008 J. Phys. Condens. Matter 20 285105
Hansen T C, Sippel C and Kuhs W F 2015 Z. Kristallogr. 230 75
Hansen T C, Falenty A and Kuhs W F 2007 in Physics and chemistry of ice W F Kuhs (ed.) (Cambridge: Royal Society of Chemistry) p 201
Kuhs W F, Genov G, Staykova D K and Hansen T 2004 Phys. Chem. Chem. Phys. 6 4917
Kohl I, Mayer E and Hallbrucker A 2000 Phys. Chem. Chem. Phys. 2 1579
Hudait A, Qiu S, Lupi L and Molinero V 2016 Phys. Chem. Chem. Phys. 18 9544
Malkin T L, Murray B J, Brukhno A V, Anwar J and Salzmann C G 2012 Proc. Natl. Acad. Sci. 109 1041
Moore E B and Molinero V 2011 Phys. Chem. Chem. Phys. 13 20008
González Solveyra E, Llave E, Scherlis D A and Molinero V 2011 J. Phys. Chem. B 115 14196
Johnston J C and Molinero V 2012 J. Am. Chem. Soc. 134 6650
Alba-Simionesco C, Coasne B, Dosseh G, Dudziak G, Gubbins K E, Radhakrishnan R et al 2006 J. Phys. Condens. Matter 18 R15
Gelb L D, Gubbins K E, Radhakrishnan R and Sliwinska- Bartkowiak M 1999 Rep. Prog. Phys. 62 1573
Radhakrishnan R, Gubbins K E and Sliwinska-Bartkowiak M 2002 J. Chem. Phys. 116 1147
Sliwinska-Bartkowiak M, Dudziak G, Sikorski R, Gras R, Radhakrishnan R and Gubbins K E 2001 J. Chem. Phys. 114 950
Morishige K, Yasunaga H and Matsutani Y 2010 J. Phys. Chem. C 114 4028
Sliwinska-Bartkowiak M, Jazdzewska M, Huang L L and Gubbins K E 2008 Phys. Chem. Chem. Phys. 10 4909
Jun S, Joo S H, Ryoo R, Kruk M, Jaroniec M, Liu Z et al 2000 J. Am. Chem. Soc. 122 10712
Kleitz F, Choi S H and Ryoo R 2003 Chem. Commun. 17 2136
Domin K 2016 PhD thesis (A. Mickiewicz University)
Balagurov A M, Beskrovnyy A I, Zhuravlev V V, Mironova G M, Bobrikov I A, Neov D et al 2016 J. Surf. Invest.: X-ray, Synchrotron Neutron Tech. 10 467
Real-Time Neutron Diffractometer. Available: http://flnph.jinr.ru/en/facilities/ibr-2/instruments/dn-2
Rietveld H M 1969 J. Appl. Cryst. 2 65
Zlokazov V B and Chernyshev V V 1992 J. Appl. Cryst. 25 447
Domin K, Chan K Y, Yung H, Gubbins K E, Jarek M, Sterc-zynska A et al 2016 J. Chem. Eng. Data 61 4252
Suzuki Y, Duran H, Steinhart M, Kappl M, Butt H J and Floudas G 2015 Nano Lett. 15 1987
Acknowledgements
Financial support from the National Center of Science under grants DEC-2013/09/B/ST4/03711 and UMO-2016/22/A/ST4/00092 and from the grant of Research Group at JINR and Research Centers in Poland under grant number 04-4-1121-2015/2020 is gratefully acknowledged. This work was also partially supported by the International PhD Projects Programme of the Foundation for Polish Science operated within the Innovative Economy Operational Programme (IE OP) 2007–2013 within the European Regional Development Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jażdżewska, M., Śliwińska-Bartkowiak, M., Domin, K. et al. Structure of ice confined in carbon and silica nanopores. Bull Mater Sci 42, 184 (2019). https://doi.org/10.1007/s12034-019-1846-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-019-1846-9