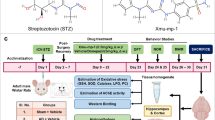
Abstract
Parkinson’s disease (PD), one of the motor system disorders, is characterized by the loss of dopamine-producing brain cells. Accumulating evidence has highlighted the involvement of microRNAs (miRs) in the development and progression of PD. Hence, we aimed at exploring possible effects of miR-326 on the progression of PD in mice in an attempt to elucidate the underlying mechanism associated with the kallikrein-related peptidase 7 (KLK7)–mediated mitogen-activated protein kinase (MAPK) signaling pathway. In order to identify the regulatory relationship between miR-326 and KLK7 and its biological significance in PD, PD mouse models were established and subsequently treated with mimics or inhibitors of miR-326 or siRNA-KLK7. The content of striatal dopamine (DA), 3,4-dihydroxyphenylacetic acid (DOPAC), homovanillic acid (HVA), 3-methoxytyrosine (3-MT), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and malondialdehyde (MDA); positive expression of tyrosine hydroxylase (TH) and inducible nitric oxide synthase (iNOS); and the levels of IL-1, IL-6, TNF-α, INF-γ, and MAPK signaling pathway–related genes were determined accordingly. The results obtained indicated that KLK7 was negatively targeted by miR-326, with lower miR-326 and higher KLK7 detected among PD mice. The overexpression of miR-326 or silencing of KLK7 was demonstrated to increase the content of DA, DOPAC, HVA, 3-MT, SOD, GSH-Px, and TH positive expression, while reducing iNOS positive expression, MDA content and cell apoptosis, as well as inhibited levels of IL-1, IL-6, TNF-α, INF-γ, and mRNA and protein levels of p38, ERK, JNK, and caspase-3. Taken together, these results provided evidence suggesting that miR-326 could inhibit iNOS activation and apoptosis of dopaminergic neurons through inhibiting the MAPK signaling pathway and negatively regulating KLK7 in mice with PD. These findings highlight the potential of miR-326 as a novel target for future PD treatment.










Similar content being viewed by others
Abbreviations
- PD:
-
Parkinson’s disease
- iNOS:
-
inducible nitric oxide synthase
- HPLC:
-
high-performance liquid chromatography
- DA:
-
dopamine
- SOD:
-
superoxide dismutase
- GSH-Px:
-
glutathione peroxidase
- MDA:
-
malondialdehyde
- TH:
-
tyrosine hydroxylase
- KLK7:
-
kallikrein-related peptidase 7
- DOPAC:
-
3,4-dihydroxyphenylacetic acid
- 3-MT:
-
3-methoxytyrosine
- TUNEL:
-
terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling
- HVA:
-
homovanillic acid
- MAPK:
-
mitogen-activated protein kinase
- ERK:
-
extracellular signal-regulated kinase
- JNK:
-
c-Jun NH2-terminal protein kinase
- 3′UTR:
-
3′untranslated region
- WT:
-
wild type
- MUT:
-
mutant
- PBS:
-
phosphate-buffered saline
- FBS:
-
fetal bovine serum
- RLU:
-
Ranilla luciferase
- NC:
-
negative control
- HBSS:
-
Hanks’ balanced salt solution
- SPF:
-
specific pathogen-free
- MPTP:
-
1-methyl-4-phenyl-1,2,4,5-tetrahydropyridine
- RT-qPCR:
-
reverse transcription quantitative polymerase chain reaction
- IgG:
-
immunoglobulin G
- DAB:
-
diaminobenzidine
- OCT:
-
optimal cutting temperature
- BSA:
-
bovine serum albumin
- GAPDH:
-
glyceraldehyde-3-phosphate dehydrogenase
- TRITC:
-
tetramethylrhodamine isothiocyanate
- FITC:
-
fluorescein isothiocyanate
- IL:
-
interleukin
- ddH2O:
-
double distilled water
- PVDF:
-
polyvinylidene fluoride
- TBST:
-
tris-buffered saline Tween-20
- ECL:
-
enhanced chemiluminescence
- SD:
-
Sprague Dawley
References
Ashby EL, Kehoe PG, Love S (2010) Kallikrein-related peptidase 6 in Alzheimer’s disease and vascular dementia. Brain Res 1363:1–10
Batra J, O’Mara T, Patnala R, Lose F, Clements JA (2012) Genetic polymorphisms in the human tissue kallikrein (KLK) locus and their implication in various malignant and non-malignant diseases. Biol Chem 393(12):1365–1390
Bellinger FP, Bellinger MT, Seale LA, Takemoto AS, Raman AV, Miki T, Manning-Bog AB, Berry MJ, White LR, Ross GW (2011) Glutathione peroxidase 4 is associated with neuromelanin in substantia nigra and dystrophic axons in putamen of Parkinson’s brain. Mol Neurodegener 6(1):8
Botella JA, Bayersdorfer F, Schneuwly S (2008) Superoxide dismutase overexpression protects dopaminergic neurons in a Drosophila model of Parkinson’s disease. Neurobiol Dis 30(1):65–73
Briggs CE, Wang Y, Kong B, Woo TU, Iyer LK, Sonntag KC (2015) Midbrain dopamine neurons in Parkinson’s disease exhibit a dysregulated miRNA and target-gene network. Brain Res 1618:111–121
Calabresi P, Di Filippo M, Ghiglieri V, Tambasco N, Picconi B (2010) Levodopa-induced dyskinesias in patients with Parkinson’s disease: filling the bench-to-bedside gap. Lancet Neurol 9(11):1106–1117
Chakraborty C, Sharma AR, Patra BC, Bhattacharya M, Sharma G, Lee SS (2016) MicroRNAs mediated regulation of MAPK signaling pathways in chronic myeloid leukemia. Oncotarget 7(27):42683–42697
Chaturvedi RK, Shukla S, Seth K, Agrawal AK (2006) Nerve growth factor increases survival of dopaminergic graft, rescue nigral dopaminergic neurons and restores functional deficits in rat model of Parkinson’s disease. Neurosci Lett 398(1–2):44–49
Cho HJ, Liu G, Jin SM, Parisiadou L, Xie C, Yu J, Sun L, Ma B, Ding J, Vancraenenbroeck R, Lobbestael E, Baekelandt V, Taymans JM, He P, Troncoso JC, Shen Y, Cai H (2013) MicroRNA-205 regulates the expression of Parkinson’s disease-related leucine-rich repeat kinase 2 protein. Hum Mol Genet 22(3):608–620
Choi I, Woo JH, Jou I, Joe EH (2016) PINK1 deficiency decreases expression levels of mir-326, mir-330, and mir-3099 during brain development and neural stem cell differentiation. Exp Neurobiol 25(1):14–23
Ci D, Song Y, Tian M, Zhang D (2015) Methylation of miRNA genes in the response to temperature stress in Populus simonii. Front Plant Sci 6:921
Corti O, Lesage S, Brice A (2011) What genetics tells us about the causes and mechanisms of Parkinson’s disease. Physiol Rev 91(4):1161–1218
Devetzi M, Trangas T, Scorilas A, Xynopoulos D, Talieri M (2013) Parallel overexpression and clinical significance of kallikrein-related peptidases 7 and 14 (KLK7KLK14) in colon cancer. Thromb Haemost 109(4):716–725
Dong Y, Tan OL, Loessner D, Stephens C, Walpole C, Boyle GM, Parsons PG, Clements JA (2010) Kallikrein-related peptidase 7 promotes multicellular aggregation via the alpha(5)beta(1) integrin pathway and paclitaxel chemoresistance in serous epithelial ovarian carcinoma. Cancer Res 70(7):2624–2633
Edwards TL, Scott WK, Almonte C, Burt A, Powell EH, Beecham GW, Wang L, Zuchner S, Konidari I, Wang G, Singer C, Nahab F, Scott B, Stajich JM, Pericak-Vance M, Haines J, Vance JM, Martin ER (2010) Genome-wide association study confirms SNPs in SNCA and the MAPT region as common risk factors for Parkinson disease. Ann Hum Genet 74(2):97–109
Feng G, Zhang Z, Bao Q, Zhang Z, Zhou L, Jiang J, Li S (2014) Protective effect of chinonin in MPTP-induced C57BL/6 mouse model of Parkinson’s disease. Biol Pharm Bull 37(8):1301–1307
Gille G, Radad K, Reichmann H, Rausch WD (2006) Synergistic effect of alpha-dihydroergocryptine and L-dopa or dopamine on dopaminergic neurons in primary culture. J Neural Transm 113(9):1107–1118
Guo Z, Shao L, Zheng L, Du Q, Li P, John B, Geller DA (2012) miRNA-939 regulates human inducible nitric oxide synthase posttranscriptional gene expression in human hepatocytes. Proc Natl Acad Sci U S A 109(15):5826–5831
Guo H, Qi RQ, Lv YN, Wang HX, Hong YX, Zheng S, Li JH, Gao XH, Chen HD (2016) miR-31 is distinctively overexpressed in primary male extramammary Paget’s disease. Oncotarget 7(17):24559–24563
Hao B, Chen X, Dai D, Zou C, Wu X, Chen J (2015) Bioinformatic analysis of microRNA expression in Parkinson’s disease. Mol Med Rep 11(2):1079–1084
Hebert SS, De Strooper B (2009) Alterations of the microRNA network cause neurodegenerative disease. Trends Neurosci 32(4):199–206
Jankovic J (2008) Parkinson’s disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry 79(4):368–376
Jha SK, Jha NK, Kar R, Ambasta RK, Kumar P (2015) p38 MAPK and PI3K/AKT signalling cascades in Parkinson’s disease. Int J Mol Cell Med 4(2):67–86
Johnstone DM, el Massri N, Moro C, Spana S, Wang XS, Torres N, Chabrol C, De Jaeger X, Reinhart F, Purushothuman S, Benabid AL, Stone J, Mitrofanis J (2014) Indirect application of near infrared light induces neuroprotection in a mouse model of parkinsonism - an abscopal neuroprotective effect. Neuroscience 274:93–101
Kefas B, Comeau L, Floyd DH, Seleverstov O, Godlewski J, Schmittgen T, Jiang J, diPierro CG, Li Y, Chiocca EA, Lee J, Fine H, Abounader R, Lawler S, Purow B (2009) The neuronal microRNA miR-326 acts in a feedback loop with notch and has therapeutic potential against brain tumors. J Neurosci 29(48):15161–15168
Khasnavis S, Ghosh A, Roy A, Pahan K (2013) Castration induces Parkinson disease pathologies in young male mice via inducible nitric-oxide synthase. J Biol Chem 288(29):20843–20855
Kim EK, Choi EJ (2015) Compromised MAPK signaling in human diseases: an update. Arch Toxicol 89(6):867–882
Kosik KS (2006) The neuronal microRNA system. Nat Rev Neurosci 7(12):911–920
Lauretani F, Maggio M, Silvestrini C, Nardelli A, Saccavini M, Ceda GP (2012) Parkinson’s disease (PD) in the elderly: an example of geriatric syndrome (GS)? Arch Gerontol Geriatr 54(1):242–246
Leal MC, Casabona JC, Puntel M, Pitossi FJ (2013) Interleukin-1beta and tumor necrosis factor-alpha: reliable targets for protective therapies in Parkinson’s disease? Front Cell Neurosci 7:53
Li S, Lv X, Zhai K, Xu R, Zhang Y, Zhao S, Qin X, Yin L, Lou J (2016) MicroRNA-7 inhibits neuronal apoptosis in a cellular Parkinson’s disease model by targeting Bax and Sirt2. Am J Transl Res 8(2):993–1004
Louter M, Tromp SC (2009) Parkinsonism due to the medication. Ned Tijdschr Geneeskd 153:A336
Martins M, Rosa A, Guedes LC, Fonseca BV, Gotovac K, Violante S, Mestre T, Coelho M, Rosa MM, Martin ER, Vance JM, Outeiro TF, Wang L, Borovecki F, Ferreira JJ, Oliveira SA (2011) Convergence of miRNA expression profiling, alpha-synuclein interacton and GWAS in Parkinson’s disease. PLoS One 6(10):e25443
Muthian G, Mackey V, King J, Charlton CG (2010) Modeling a sensitization stage and a precipitation stage for Parkinson’s disease using prenatal and postnatal 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine administration. Neuroscience 169(3):1085–1093
Nie K, Zhang Y, Gan R, Wang L, Zhao J, Huang Z, Tang H, Wang L (2013) Polymorphisms in immune/inflammatory cytokine genes are related to Parkinson’s disease with cognitive impairment in the Han Chinese population. Neurosci Lett 541:111–115
Pramanik D, Campbell NR, Karikari C, Chivukula R, Kent OA, Mendell JT, Maitra A (2011) Restitution of tumor suppressor microRNAs using a systemic nanovector inhibits pancreatic cancer growth in mice. Mol Cancer Ther 10(8):1470–1480
Saito T, Nakanishi H, Mochizuki Y, Ito S, Ito Y, Misawa K, Yatabe Y, Yamamichi K, Kondo E (2015) Preferential HER2 expression in liver metastases and EGFR expression in peritoneal metastases in patients with advanced gastric cancer. Gastric Cancer 18(4):711–719
Scalzo P, Kummer A, Cardoso F, Teixeira AL (2009) Increased serum levels of soluble tumor necrosis factor-alpha receptor-1 in patients with Parkinson’s disease. J Neuroimmunol 216(1–2):122–125
Scalzo P, Kummer A, Cardoso F, Teixeira AL (2010) Serum levels of interleukin-6 are elevated in patients with Parkinson’s disease and correlate with physical performance. Neurosci Lett 468(1):56–58
Sukumar N, Scott E, Dimitromanolakis A, Misiak A, Prassas I, Diamandis EP, Konvalinka A (2014) Mining for single nucleotide variants (SNVs) at the kallikrein locus with predicted functional consequences. Biol Chem 395(9):1037–1050
Thome AD, Harms AS, Volpicelli-Daley LA, Standaert DG (2016) microRNA-155 regulates alpha-synuclein-induced inflammatory responses in models of Parkinson disease. J Neurosci 36(8):2383–2390
Walker F, Nicole P, Jallane A, Soosaipillai A, Mosbach V, Oikonomopoulou K, Diamandis EP, Magdolen V, Darmoul D (2014) Kallikrein-related peptidase 7 (KLK7) is a proliferative factor that is aberrantly expressed in human colon cancer. Biol Chem 395(9):1075–1086
Wu D, Tseng IJ, Yuan RY, Hsieh CY, Hu CJ (2014) Memory consolidation and inducible nitric oxide synthase expression during different sleep stages in Parkinson disease. Sleep Med 15(1):116–120
Xia D, Tian S, Chen Z, Qin W, Liu Q (2018) miR302a inhibits the proliferation of esophageal cancer cells through the MAPK and PI3K/Akt signaling pathways. Oncol Lett 15(3):3937–3943
Xiao L, Zhou X, Liu F, Hu C, Zhu X, Luo Y, Wang M, Xu X, Yang S, Kanwar YS, Sun L (2015) MicroRNA-129-5p modulates epithelial-to-mesenchymal transition by targeting SIP1 and SOX4 during peritoneal dialysis. Lab Investig 95(7):817–832
Xie XX, Kou ST, Pu ZH, Hou CY, Tian YP (2007) Effects of scalp catgut embedding on SOD, NO, MDA in the rat with Parkinson’s disease. Zhongguo zhen jiu = Chin Acupunct moxibust 27(10):753–756
Xie YF, Shu R, Jiang SY, Liu DL, Ni J, Zhang XL (2013) MicroRNA-146 inhibits pro-inflammatory cytokine secretion through IL-1 receptor-associated kinase 1 in human gingival fibroblasts. J Inflamm 10(1):20
Yin XF, Xu HM, Jiang YX, Zhi YL, Liu YX, Xiang HW, Liu K, Ding XD, Sun P (2015) Lentivirus-mediated Persephin over-expression in Parkinson’s disease rats. Neural Regen Res 10(11):1814–1818
Yoon H, Blaber SI, Li W, Scarisbrick IA, Blaber M (2013) Activation profiles of human kallikrein-related peptidases by matrix metalloproteinases. Biol Chem 394(1):137–147
Yu SY, Zuo LJ, Wang F, Chen ZJ, Hu Y, Wang YJ, Wang XM, Zhang W (2014) Potential biomarkers relating pathological proteins, neuroinflammatory factors and free radicals in PD patients with cognitive impairment: a cross-sectional study. BMC Neurol 14:113
Zhang Z, Luo X, Ding S, Chen J, Chen T, Chen X, Zha H, Yao L, He X, Peng H (2012) MicroRNA-451 regulates p38 MAPK signaling by targeting of Ywhaz and suppresses the mesangial hypertrophy in early diabetic nephropathy. FEBS Lett 586(1):20–26
Zhang J, Li S, Yan Q, Chen X, Yang Y, Liu X, Wan X (2013) Interferon-beta induced microRNA-129-5p down-regulates HPV-18 E6 and E7 viral gene expression by targeting SP1 in cervical cancer cells. PLoS One 8(12):e81366
Zhou Q, Chen J, Yi S, Lou Y, Tang W, Liu Y, Zhang P (2012) Zhichan powder regulates nigrostriatal dopamine synthesis and metabolism in Parkinson’s disease rats. Neural Regen Res 7(27):2107–2114
Ziebarth JD, Bhattacharya A, Cui Y (2012) Integrative analysis of somatic mutations altering microRNA targeting in cancer genomes. PLoS One 7(10):e47137
Acknowledgments
We would like to acknowledge the helpful comments on this paper received from our reviewers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yizhi Zhang and Weiwei Xu are co-first authors.
Rights and permissions
About this article
Cite this article
Zhang, Y., Xu, W., Nan, S. et al. MicroRNA-326 Inhibits Apoptosis and Promotes Proliferation of Dopaminergic Neurons in Parkinson’s Disease Through Suppression of KLK7-Mediated MAPK Signaling Pathway. J Mol Neurosci 69, 197–214 (2019). https://doi.org/10.1007/s12031-019-01349-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-019-01349-1