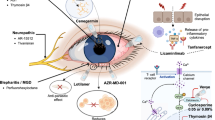
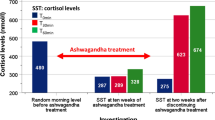
Abstract
Pituitary adenylate cyclase-activating polypeptide (PACAP) is a 27- or 38-amino acid neuropeptide, which belongs to the vasoactive intestinal polypeptide/glucagon/secretin family of peptides. PACAP and its three receptor subtypes are expressed in neural tissues and in the eye, including the retina, cornea, and lacrimal gland. PACAP is known to exert pleiotropic effects on the central nervous system and in eye tissues where it plays important roles in protecting against dry eye. This review provides an overview of current knowledge regarding dry eye symptoms in aged animals and humans and the protective effects, mechanisms of action. In addition, we also refer to the development of a new preventive/therapeutic method by PACAP of dry eye patients.





Similar content being viewed by others
References
Arimura A, Shioda S (1995) Pituitary adenylate cyclase activating polypeptide (PACAP) and its receptors: neuroendocrine and endocrine interaction. Front Neuroendocrinol 16(1):53–88
Asghar MS, Hansen AE, Amin FM, van der Geest RJ, Koning P, Larsson HBW, Olesen J, Ashina M (2011) Evidence for a vascular factor in migraine. Ann Neurol 69(4):635–645
Azcarate PM, Venincasa VD, Feuer W, Stanczyk F, Schally AV, Galor A (2014) Androgen deficiency and dry eye syndrome in the aging male. Invest Ophthalmol Vis Sci 55(8):5046–5053
Banks WA, Kastin AJ, Komaki G, Arimura A (1993) Passage of pituitary adenylate cyclase activating polypeptide1-27 and pituitary adenylate cyclase activating polypeptide1-38 across the blood-brain barrier. J Pharmacol Exp Ther 267(2):690–696
Banks WA, Uchida D, Arimura A, Somogyvari-Vigh A, Shioda S (1996) Transport of pituitary adenylate cyclase-activating polypeptide across the blood-brain barrier and the prevention of ischemia-induced death of hippocampal neurons. Ann N Y Acad Sci 805:270–277 discussion 277–279
Barabino S, Dana MR (2004) Animal models of dry eye: a critical assessment of opportunities and limitations. Invest Ophthalmol Vis Sci 45(6):1641–1646
Birk S, Sitarz JT, Petersen KA, Oturai PS, Kruuse C, Fahrenkrug J, Olesen J (2007) The effect of intravenous PACAP38 on cerebral hemodynamics in healthy volunteers. Regul Pept 140(3):185–191
Blehm C, Vishnu S, Khattak A, Mitra S, Yee RW (2005) Computer vision syndrome: a review. Surv Ophthalmol 50(3):253–262
Boni LJ, Ploug KB, Olesen J, Jansen-Olesen I, Gupta S (2009) The in vivo effect of VIP, PACAP-38 and PACAP-27 and mRNA expression of their receptors in rat middle meningeal artery. Cephalalgia 29(8):837–847
Borba JC, Henze IP, Silveira MS et al (2005) Pituitary adenylate cyclase-activating polypeptide (PACAP) can act as determinant of the tyrosine hydroxylase phenotype of dopaminergic cells during retina development. Brain Res Dev Brain Res 156(2):193–201
Castle NA (2005) Aquaporins as targets for drug discovery. Drug Discov Today 10(7):485–493
Chen Q, Wang J, Shen M, Cai C, Li J, Cui L, Qu J, Lu F (2009) Lower volumes of tear menisci in contact lens wearers with dry eye symptoms. Invest Ophthalmol Vis Sci 50(7):3159–3163
D’Agata V, Cavallaro S (1998) Functional and molecular expression of PACAP/VIP receptors in the rat retina. Brain Res Mol Brain Res 54(1):161–164
Dartt DA (1989) Signal transduction and control of lacrimal gland protein secretion: a review. Curr Eye Res 8(6):619–636
Dartt DA (1994) Regulation of tear secretion. Adv Exp Med Biol 350:1–9
Dartt DA (2009) Neural regulation of lacrimal gland secretory processes: relevance in dry eye diseases. Prog Retin Eye Res 28(3):155–177
Elsas T, Uddman R, Sundler F (1996) Pituitary adenylate cyclase-activating peptide-immunoreactive nerve fibers in the cat eye. Graefes Arch Clin Exp Ophthalmol 234(9):573–580
Fushimi K, Sasaki S, Marumo F (1997) Phosphorylation of serine 256 is required for cAMP-dependent regulatory exocytosis of the aquaporin-2 water channel. J Biol Chem 272(23):14800–14804
Gaal V, Mark L, Kiss P, Kustos I, Tamas A, Kocsis B, Lubics A, Nemeth V, Nemeth A, Lujber L, Pytel J, Toth G, Reglodi D (2008) Investigation of the effects of PACAP on the composition of tear and endolymph proteins. J Mol Neurosci 36(1–3):321–329
Grone BP, Sheng Z, Chen CC, Fernald RD (2007) Localization and diurnal expression of melanopsin, vertebrate ancient opsin, and pituitary adenylate cyclase-activating peptide mRNA in a teleost retina. J Biol Rhythm 22(6):558–561
Harmar AJ, Fahrenkrug J, Gozes I, Laburthe M, May V, Pisegna JR, Vaudry D, Vaudry H, Waschek JA, Said SI (2012) Pharmacology and functions of receptors for vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide: IUPHAR review 1. Br J Pharmacol 166(1):4–17
Hodges RR, Dartt DA (2003) Regulatory pathways in lacrimal gland epithelium. Int Rev Cytol 231:129–196
Horsefield R, Norden K, Fellert M, Backmark A, Tornroth-Horsefield S, Terwisscha van Scheltinga AC, Kvassman J, Kjellbom P, Johanson U, Neutze R (2008) High-resolution x-ray structure of human aquaporin 5. Proc Natl Acad Sci U S A 105(36):13327–13332
Il’inskii OB, Spevak SE, Kochetkov NV, Solov’eva AI, Krasnikova TL (1985) Participation of the lacrimal glands in wound healing processes. Biull Eksp Biol Med 100(7):91–93
Izumi S, Seki T, Shioda S, Zhou CJ, Arimura A, Koide R (2000) Ultrastructural localization of PACAP immunoreactivity in the rat retina. Ann N Y Acad Sci 921:317–320
Jozsa R, Somogyvari-Vigh A, Reglodi D, Hollosy T, Arimura A (2001) Distribution and daily variations of PACAP in the chicken brain. Peptides 22(9):1371–1377
Kosugi-Tanaka C, Li X, Yao C, Akamatsu T, Kanamori N, Hosoi K (2006) Protein kinase A-regulated membrane trafficking of a green fluorescent protein-aquaporin 5 chimera in MDCK cells. Biochim Biophys Acta 1763(4):337–344
Kubrusly RC, da Cunha MC, Reis RA et al (2005) Expression of functional receptors and transmitter enzymes in cultured Muller cells. Brain Res 1038(2):141–149
Labrie F, Belanger A, Cusan L, Gomez JL, Candas B (1997) Marked decline in serum concentrations of adrenal C19 sex steroid precursors and conjugated androgen metabolites during aging. J Clin Endocrinol Metab 82(8):2396–2402
Lakk M, Szabo B, Volgyi B, Gabriel R, Denes V (2012) Development-related splicing regulates pituitary adenylate cyclase-activating polypeptide (PACAP) receptors in the retina. Invest Ophthalmol Vis Sci 53(12):7825–7832
Mathis U, Schaeffel F (2007) Glucagon-related peptides in the mouse retina and the effects of deprivation of form vision. Graefes Arch Clin Exp Ophthalmol 245(2):267–275
Miyata A, Arimura A, Dahl RR, Minamino N, Uehara A, Jiang L, Culler MD, Coy DH (1989) Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem Biophys Res Commun 164(1):567–574
Moss SE, Klein R, Klein BE (2000) Prevalence of and risk factors for dry eye syndrome. Arch Ophthalmol 118(9):1264–1268
Nakamachi T, Ohtaki H, Seki T, Yofu S, Kagami N, Hashimoto H, Shintani N, Baba A, Mark L, Lanekoff I, Kiss P, Farkas J, Reglodi D, Shioda S (2016) PACAP suppresses dry eye signs by stimulating tear secretion. Nat Commun 7:12034
Nedvetsky PI, Tamma G, Beulshausen S et al (2009) Regulation of aquaporin-2 trafficking. Handb Exp Pharmacol 190:133–157
Nilsson SF, De Neef P, Robberecht P, Christophe J (1994) Characterization of ocular receptors for pituitary adenylate cyclase activating polypeptide (PACAP) and their coupling to adenylate cyclase. Exp Eye Res 58(4):459–467
Nowak JZ, Jozwiak-Bebenista M, Bednarek K (2007) Effects of PACAP and VIP on cyclic AMP formation in rat neuronal and astrocyte cultures under normoxic and hypoxic condition. Peptides 28(9):1706–1712
O’Brien PD, Collum LM (2004) Dry eye: diagnosis and current treatment strategies. Curr Allergy Asthma Rep 4(4):314–319
Olianas MC, Ennas MG, Lampis G, Onali P (1996) Presence of pituitary adenylate cyclase-activating polypeptide receptors in Y-79 human retinoblastoma cells. J Neurochem 67(3):1293–1300
Olianas MC, Ingianni A, Sogos V, Onali P (1997) Expression of pituitary adenylate cyclase-activating polypeptide (PACAP) receptors and PACAP in human fetal retina. J Neurochem 69(3):1213–1218
Peck T, Olsakovsky L, Aggarwal S (2018) Dry eye syndrome in menopause and perimenopausal age group. J Mid-life Health. 160:122–147
Rahimi Darabad R, Suzuki T, Richards SM, Jakobiec FA, Zakka FR, Barabino S, Sullivan DA (2014) Does estrogen deficiency cause lacrimal gland inflammation and aqueous-deficient dry eye in mice? Exp Eye Res 127:153–160
Reglodi D, Somogyvari-Vigh A, Vigh J et al (2001) Pituitary adenylate cyclase activating polypeptide is highly abundant in the nervous system of anoxia-tolerant turtle, Pseudemys scripta elegans. Peptides 22(6):873–878
Schaumberg DA, Dana R, Buring JE, Sullivan DA (2009) Prevalence of dry eye disease among US men: estimates from the Physicians’ health studies. Arch Ophthalmol 127(6):763–768
Schaumberg DA, Sullivan DA, Buring JE, Dana MR (2003) Prevalence of dry eye syndrome among US women. Am J Ophthalmol 136(2):318–326
Schrader S, Mircheff AK, Geerling G (2008) Animal models of dry eye. Dev Ophthalmol 41:298–312
Seki T, Shioda S, Ogino D, Nakai Y, Arimura A, Koide R (1997) Distribution and ultrastructural localization of a receptor for pituitary adenylate cyclase activating polypeptide and its mRNA in the rat retina. Neurosci Lett 238(3):127–130
Seki T, Shioda S, Nakai Y, Arimura A, Koide R (1998) Distribution and ultrastructural localization of pituitary adenylate cyclase-activating polypeptide (PACAP) and its receptor in the rat retina. Ann N Y Acad Sci 865:408–411
Seki T, Izumi S, Shioda S, Zhou CJ, Arimura A, Koide R (2000a) Gene expression for PACAP receptor mRNA in the rat retina by in situ hybridization and in situ RT-PCR. Ann N Y Acad Sci 921:366–369
Seki T, Shioda S, Izumi S, Arimura A, Koide R (2000b) Electron microscopic observation of pituitary adenylate cyclase-activating polypeptide (PACAP)-containing neurons in the rat retina. Peptides 21(1):109–113
Sharma A, Hindman HB (2014) Aging: a predisposition to dry eyes. J Ophthalmol 2014:781683
Sherwood NM, Krueckl SL, McRory JE (2000) The origin and function of the pituitary adenylate cyclase-activating polypeptide (PACAP)/glucagon superfamily. Endocr Rev 21(6):619–670
Shioda S, Wascheck J (2002) VIP and PACAP receptors. In: Pangalos MN and Davies CH (eds) Understanding G protein-coupled receptors and their role in the CNS. Oxford Univ Press, pp 527–545
Shioda S, Nakamachi T (2015) PACAP as a neuroprotective factor in ischemic neuronal injuries. Peptides 72:202–207
Shioda S, Takenoya F, Wada N, Hirabayashi T, Seki T, Nakamachi T (2016) Pleiotropic and retinoprotective functions of PACAP. Anat Sci Int 91(4):313–324
Sidhaye V, Hoffert JD, King LS (2005) cAMP has distinct acute and chronic effects on aquaporin-5 in lung epithelial cells. J Biol Chem 280(5):3590–3596
Silveira MS, Costa MR, Bozza M, Linden R (2002) Pituitary adenylyl cyclase-activating polypeptide prevents induced cell death in retinal tissue through activation of cyclic AMP-dependent protein kinase. J Biol Chem 277(18):16075–16080
Sullivan DA, Jensen RV, Suzuki T, Richards SM (2009) Do sex steroids exert sex-specific and/or opposite effects on gene expression in lacrimal and meibomian glands? Mol Vis 15:1553–1572
Suzuki T, Schirra F, Richards SM, Treister NS, Lombardi MJ, Rowley P, Jensen RV, Sullivan DA (2006) Estrogen’s and progesterone’s impact on gene expression in the mouse lacrimal gland. Invest Ophthalmol Vis Sci 47(1):158–168
Tsubota K, Hirai S, King LS, Agre P, Ishida N (2001) Defective cellular trafficking of lacrimal gland aquaporin-5 in Sjogren’s syndrome. Lancet 357(9257):688–689
Valtysdottir ST, Wide L, Hallgren R (2003) Mental wellbeing and quality of sexual life in women with primary Sjogren’s syndrome are related to circulating dehydroepiandrosterone sulphate. Ann Rheum Dis 62(9):875–879
Vaudry D, Falluel-Morel A, Bourgault S, Basille M, Burel D, Wurtz O, Fournier A, Chow BKC, Hashimoto H, Galas L, Vaudry H (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61(3):283–357
Vaudry D, Gonzalez BJ, Basille M, Yon L, Fournier A, Vaudry H (2000) Pituitary adenylate cyclase-activating polypeptide and its receptors: from structure to functions. Pharmacol Rev 52(2):269–324
Wang ZY, Alm P, Hakanson R (1995) Distribution and effects of pituitary adenylate cyclase-activating peptide in the rabbit eye. Neuroscience 69(1):297–308
Yang F, Kawedia JD, Menon AG (2003) Cyclic AMP regulates aquaporin 5 expression at both transcriptional and post-transcriptional levels through a protein kinase A pathway. J Biol Chem 278(34):32173–32180
Zhang XY, Hayasaka S, Chi ZL, Cui HS, Hayasaka Y (2005) Effect of pituitary adenylate cyclase-activating polypeptide (PACAP) on IL-6, IL-8, and MCP-1 expression in human retinal pigment epithelial cell line. Curr Eye Res 30(12):1105–1111
Acknowledgments
We are indebted to co-researchers for assistance, especially to Drs. Dora Reglodi of Pecs University, Hitoshi Hashimoto of Osaka University, Rakwal Randeep of Tsukuba University and Ms. Junko Shibato of Hoshi University for the research described in this review paper.
Funding
This work was supported by the JSPS KAKENHI grant numbers 2459280, 2459268, 23249079, 15K15670, 16H02684, and 15H01288.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Shioda, S., Takenoya, F., Hirabayashi, T. et al. Effects of PACAP on Dry Eye Symptoms, and Possible Use for Therapeutic Application. J Mol Neurosci 68, 420–426 (2019). https://doi.org/10.1007/s12031-018-1087-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1087-1