Abstract
Purpose of Review
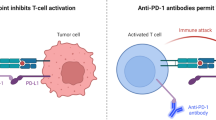
T-cell immunoglobulin and ITIM domain (TIGIT) is a next-generation inhibitory receptor with multiple antibodies under exploration in cancer therapy. Here, we review the available data from the early trials and overview upcoming clinical trials on anti-TIGIT antibodies.
Recent Findings
There is a promising activity of anti-TIGIT, particularly in combination with anti-PD-1/PD-L1 in non-small cell lung cancer (NSCLC) with already phase 3 trials currently ongoing to confirm these early findings.
Summary
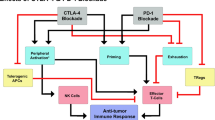
Numerous anti-TIGIT antibodies are in clinical trials currently, and others are in preclinical development. Therefore, more data are expected in the next few years regarding the efficacy of this new checkpoint inhibitor in multiple solid and hematologic malignancies. However, preliminary data are promising, and anti-TIGIT treatment seems to confer more favorable responses when combined with anti-PD-1/anti-PD-L1 compared to either agent alone.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Anderson AC, Joller N, Kuchroo VK. L1 Anderson, A. C., Joller, N. and Kuchroo, V. K. (2016) Lag-3, Tim-3, and TIGIT: co-inhibitory receptors with specialized functions in immune regulation. Immunity, NIH Public Access 44, 989–1004.ag-3, Tim-3, and TIGIT: Co-inhibitory Receptors with Specia. Immunity. 2016;44:989–1004.
Levin SD, Taft DW, Brandt CS, et al. Vstm3 is a member of the CD28 family and an important modulator of T-cell function. Eur J Immunol. 2011;41:902–15.
Stanietsky N, Simic H, Arapovic J, et al. The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc Natl Acad Sci U S A. 2009;106:17858–63.
• Yu X, Harden K, Gonzalez LC, et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol. 2009;10:48–57. (One of the first reports on TIGIT immunosuppressive role.)
Boles KS, Vermi W, Facchetti F, Fuchs A, Wilson TJ, Diacovo TG, Cella M, Colonna M. A novel molecular interaction for the adhesion of follicular CD4 T cells to follicular DC. Eur J Immunol. 2009;39:695–703.
Liu S, Zhang H, Li M, Hu D, Li C, Ge B, Jin B, Fan Z. Recruitment of Grb2 and SHIP1 by the ITT-like motif of TIGIT suppresses granule polarization and cytotoxicity of NK cells. Cell Death Differ. 2013;20:456–64.
Joller N, Hafler JP, Brynedal B, Kassam N, Spoerl S, Levin SD, Sharpe AH, Kuchroo VK. Cutting edge: TIGIT has T cell-intrinsic inhibitory functions. J Immunol. 2011;186:1338–42.
Josefsson SE, Huse K, Kolstad A, et al. T cells expressing checkpoint receptor TIGIT are enriched in follicular lymphoma tumors and characterized by reversible suppression of T-cell receptor signaling. Clin Cancer Res. 2018;24:870–81.
Dougall WC, Kurtulus S, Smyth MJ, Anderson AC. TIGIT and CD96: new checkpoint receptor targets for cancer immunotherapy. Immunol Rev. 2017;276:112–20.
Johnston RJ, Comps-Agrar L, Hackney J, et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8(+) T cell effector function. Cancer Cell. 2014;26:923–37.
Chauvin J-M, Pagliano O, Fourcade J, et al. TIGIT and PD-1 impair tumor antigen-specific CD8+ T cells in melanoma patients. J Clin Invest. 2015;125:2046–58.
• Rodriguez-Abreu D, Johnson ML, Hussein MA, et al. Primary analysis of a randomized, double-blind, phase II study of the anti-TIGIT antibody tiragolumab (tira) plus atezolizumab (atezo) versus placebo plus atezo as first-line (1L) treatment in patients with PD-L1-selected NSCLC (CITYSCAPE). J Clin Oncol. 2020;38:9503–9503. (Key studies on anti-TIGIT antibodies in the treatment of lung cancer.)
• Cho BC, Rodriguez-Abreu D, Hussein M, et al (2021) LBA2 Updated analysis and patient-reported outcomes (PROs) from CITYSCAPE: a randomised, double-blind, phase II study of the anti-TIGIT antibody tiragolumab + atezolizumab (TA) versus placebo + atezolizumab (PA) as first-line treatment for PD-L1+ NSCLC. Ann Oncol 32:S1428. (Key studies on anti-TIGIT antibodies in the treatment of lung cancer.)
• Niu J, Maurice-Dror C, Lee DH, et al. First-in-human phase 1 study of the anti-TIGIT antibody vibostolimab as monotherapy or with pembrolizumab for advanced solid tumors, including non-small cell lung cancer. Ann Oncol Off J Eur Soc Med Oncol. 2021. https://doi.org/10.1016/j.annonc.2021.11.002. (Key studies on anti-TIGIT antibodies in the treatment of lung cancer.)
Van den Mooter TFA, Migeotte A, Jungels C, et al (2021) Abstract CT118: Preliminary data from phase I first-in-human study of EOS884448, a novel potent anti-TIGIT antibody, monotherapy shows favorable tolerability profile and early signsof clinical activity in immune-resistant advanced cancers. In: Clin. Trials. American Association for Cancer Research, pp CT118–CT118
Piovesan D, Lopez A, Schweickert P, et al. 258 AB308 is an anti-TIGIT antibody that enhances immune activation and anti-tumor immunity alone and in combination with other I-O therapeutic agents. J Immunother Cancer. 2021;9:A280–A280.
Chaudhry A, Johnson M, Colburn D, Gilbert H, Garofalo A, Paoloni M, Krishnan K. 1419TiP ARC-7: a phase II study to evaluate the safety and efficacy of zimberelimab alone, AB154 in combination with zimberelimab, and AB154 in combination with zimberelimab and AB928 in front-line, PD-L1 expressing, non-small cell lung cancer (NSCLC). Ann Oncol. 2020;31:S897.
Frentzas S, Meniawy T, Kao SC-H, Wang R, Zuo Y, Zheng H, Tan W. AdvanTIG-105: phase 1 dose-escalation study of anti-TIGIT monoclonal antibody ociperlimab (BGB-A1217) in combination with tislelizumab in patients with advanced solid tumors. J Clin Oncol. 2021;39:2583–2583.
Xu R, Kim S-B, Tougeron D, Zuo Y, Yang H, Zhang J, Shi J, Van Cutsem E. AdvanTIG-203: A randomized phase 2 study comparing anti-TIGIT ociperlimab plus tislelizumab versus tislelizumab plus placebo as second-line treatment in patients with advanced or recurrent esophageal squamous cell carcinoma (ESCC) expressing programmed de. J Clin Oncol. 2021;39:TPS4150–TPS4150.
Wu L, Wang P-H, Hsiao S-Y, et al. AdvanTIG-202: a phase 2 study investigating anti-TIGIT monoclonal antibody ociperlimab plus anti-PD-1 monoclonal antibody tislelizumab in patients with previously treated recurrent or metastatic cervical cancer. J Clin Oncol. 2021;39:TPS5595–TPS5595.
Socinski MA, Spira AI, Paz-Ares LG, et al. AdvanTIG-302: Anti-TIGIT monoclonal antibody (mAb) ociperlimab (OCI) plus tislelizumab (TIS) versus pembrolizumab (PEM) in programmed death ligand-1 (PD-L1) selected, previously untreated, locally advanced, unresectable or metastatic non-small cell lung c. J Clin Oncol. 2021;39:TPS9128–TPS9128.
Mettu NB, Ulahannan S V., Bendell JC, Garrido-Laguna I, Strickler JH, Moore KN, Stagg R, Kapoun AM, Faoro L, Sharma S (2021) A phase 1a/b open‑label, dose‑escalation study of etigilimab alone or in combination with nivolumab in patients with locally advanced or metastatic solid tumors. Clin Cancer Res 2780.2021
Dumbrava E, Sharma M, Fleming G, et al (2021) COM701 in combination with BMS-986207 (anti-TIGIT antibody) and nivolumab - preliminary results of safety, tolerability and pharmacokinetics in patients with advanced solid tumors. In: SITC Annu. Meet. p Abstract 478
Garralda E, Sanborn RE, Minchom AR, et al. SGNTGT-001: a phase 1 study of SEA-TGT, an effector-function enhanced monoclonal antibody (mAb), in advanced malignancies (trial in progress). J Clin Oncol. 2021;39:TPS2657–TPS2657.
Smith A, Zeng W, Lucas S, Grogan B, Blackmarr A, Wo S, Peterson S, Gardai S (2021) Abstract 1583: SEA-TGT is an empowered anti-TIGIT antibody that displays superior combinatorial activity with several therapeutic agents. In: Immunology. Am Assoc Cancer Res 1583–1583
Shirasuna K, Koelsch G, Seidel-Dugan C, et al (2021) Characterization of ASP8374, a fully-human, antagonistic anti-TIGIT monoclonal antibody. Cancer Treat Res Commun 28:100433
Author information
Authors and Affiliations
Ethics declarations
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Conflict of Interest
Dr. Vaia Florou has nothing to disclose. Dr. Ignacio Garrido-Laguna reports personal fees from Array BioPharma, and research funding (institutional) from Novartis, Ignyta, Halozyme, Bristol-Myers Squibb, Bayer, Pfizer, Newlink Genetics, MedImmune, Lilly, Incyte, GlaxoSmithKline, OncoMed, ARMO BioSciences, Glennmark.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical collection on Immuno-oncology
Rights and permissions
About this article
Cite this article
Florou, V., Garrido-Laguna, I. Clinical Development of Anti-TIGIT Antibodies for Immunotherapy of Cancer. Curr Oncol Rep 24, 1107–1112 (2022). https://doi.org/10.1007/s11912-022-01281-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-022-01281-5