Abstract
Purpose of Review
Imaging features of lymphoma vary regionally. Awareness of site-specific key imaging characteristics of lymphoma can aid in rapid staging and assist in prompt treatment. FDG PET/CT and conventional MRI are readily available diagnostic modalities with excellent sensitivity and good specificity. Diagnostic specificity can be enhanced using emerging PET radiotracers, e.g., FLT and FET.
Recent Findings
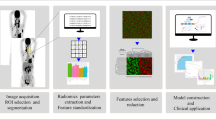
Emerging research has shown higher dimensional analysis (radiomics and radiogenomics) of imaging data can yield information of the underlying genetic aberrations in lymphoma, which can aid in assessing real-time evolution of tumor.
Summary
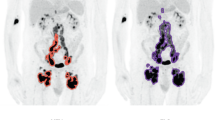
CT, PET/CT, MRI, and ultrasound accentuate the intrinsic qualities of lymphoma (e.g., FDG PET/CT for increased metabolic activity, FLT PET/CT for increased proliferation index, and DWI for increased cellularity) and play an essential role in its diagnosis and examination. Advanced radiogenomic analyses use radiomic parameters to deduce genetic variations of lymphoma, providing noninvasive, repeatable, and real-time surveillance of its genetic progression.








Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
LaCasce AS, et al. Clinical presentation and diagnosis of classic Hodgkin lymphoma in adults. UpToDate. A. G. Rosmarin and A. S. Freedman. 2019.
LaCasce AS, Ng AK. Hodgkin lymphoma: epidemiology and risk factors. UpToDate. A. G. Rosmarin and A. S. Freedman. 2020.
Bastidas Torres AN, Cats D, Mei H, Szuhai K, Willemze R, Vermeer MH, et al. Genomic analysis reveals recurrent deletion of JAK-STAT signaling inhibitors HNRNPK and SOCS1 in mycosis fungoides. Genes Chromosomes Cancer. 2018;57(12):653–64.
Salati M, Cesaretti M, Macchia M, Mistiri ME, Federico M. Epidemiological overview of Hodgkin lymphoma across the Mediterranean Basin. Mediterr J Hematol Infect Dis. 2014;6(1):e2014048.
Martin LJ. Understanding Hodgkin lymphoma (Hodgkin's disease) -- basic information. 2017.
Johnston PB, et al. Everolimus combined with R-CHOP-21 for new, untreated, diffuse large B-cell lymphoma (NCCTG 1085 [Alliance]): safety and efficacy results of a phase 1 and feasibility trial. Lancet Haematol. 2016;3:e309.
Ghielmini M, Vitolo U, Kimby E, Montoto S, Walewski J, Pfreundschuh M, et al. ESMO guidelines consensus conference on malignant lymphoma 2011 part 1: diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL) and chronic lymphocytic leukemia (CLL). Ann Oncol. 2013;24(3):561–76.
Zenz T, Kreuz M, Fuge M, Klapper W, Horn H, Staiger AM, et al. TP53 mutation and survival in aggressive B cell lymphoma. Int J Cancer. 2017;141(7):1381–8.
Vaque JP, et al. B-cell lymphoma mutations: improving diagnostics and enabling targeted therapies. Haematologica. 2014;99(2):222–31.
Alexandrow M, et al. Differences between Hodgkin & non-Hodgkin lymphoma. Moffit Cancer Center.
Portell CA. Marginal zone lymphoma. Lymphoma. 2018.
Swerdlow E, Campo N, Harris NL, et al. WHO classification of tumours of hematopoietic and lymphoid tissues. 4th ed. Lyon: IARC Press Lyon; 2008.
Skibola CF, Curry JD, Nieters A. Genetic susceptibility to lymphoma. Haematologica. 2007;92(7):960–9.
Bell DJ,Gaillard F.Primary CNS Lymphoma. Radiopaedia.
Huret JL, Huret JL. Anaplasic large cell lymphoma (ALCL). Atlas Genet Cytogenet Oncol Haematol. 2003;7(4):262–5.
Ke L, Xu M, Jiang X, Sun X. Epidermal growth factor receptor (EGFR) mutations and anaplastic lymphoma kinase/oncogene or C-Ros oncogene 1 (ALK/ROS1) fusions inflict non-small cell lung Cancer (NSCLC) female patients older than 60 years of age. Med Sci Monit. 2018;24:9364–9.
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32(27):3059–68.
Fujinaga Y, Lall C, Patel A, Matsushita T, Sanyal R, Kadoya M. MR features of primary and secondary malignant lymphoma of the pancreas: a pictorial review. Insights Imaging. 2013;4(3):321–9.
Batchelor T, LoefflerJS. Clinical presentation, pathologic features, and diagnosis of primary central nervous system lymphoma. UpToDate. A. F. Eichler and A. S. Freedman. 2019.
Weerakkody Y, Gaillard F. Orbital Lymphoma. Radiopaedia.
Hui KH, Pfeiffer ML, Esmaeli B. Value of positron emission tomography/computed tomography in diagnosis and staging of primary ocular and orbital tumors. Saudi J Ophthalmol. 2012;26(4):365–71.
Kato H, et al. Mucosa-associated lymphoid tissue lymphoma of the salivary glands: MR imaging findings including diffusion-weighted imaging. Eur J Radiol. 2012;81(4):e612–7.
Hadiprodjo D, Ryan T, Truong MT, Mercier G, Subramaniam RM. Parotid gland tumors: preliminary data for the value of FDG PET/CT diagnostic parameters. AJR Am J Roentgenol. 2012, Feb;198(2):W185–90. https://doi.org/10.2214/AJR.11.7172.
Jones J, KnipeH. Thyroid Lymphoma. Radiopaedia.
Friedberg JW, et al. Thyroid lymphoma. UpToDate. J. E. Mulder and D. S. Ross. 2019.
Cho KS, et al. Differential diagnosis of primary nasopharyngeal lymphoma and nasopharyngeal carcinoma focusing on CT, MRI, and PET/CT. Otolaryngol Head Neck Surg. 2012;146(4):574–8.
Zhou ML, Zhao K, Zhou SH, Wang QY, Zheng ZJ, Lu ZJ. Role of PET/CT in the diagnosis, staging, and follow-up of a nasal-type natural killer T-cell lymphoma in the larynx: a case report and literature review. Int J Clin Exp Med. 2014;7(11):4483–91.
Suh HB, Choi YS, Bae S, Ahn SS, Chang JH, Kang S-G, et al. Primary central nervous system lymphoma and atypical glioblastoma: differentiation using radiomics approach. Eur Radiol. 2018;28(9):3832–9. https://doi.org/10.1007/s00330-018-5368-4.
Kong F-L, et al. Managing lymphoma with non-FDG radiotracers: current clinical and preclinical applications. Biomed Res Int. 2013;2013:12.
•• Kong Z, et al. (18)F-FDG-PET-based radiomics features to distinguish primary central nervous system lymphoma from glioblastoma. Neuroimage Clin. 2019;23:101912. The authors sought to determine if (18)F-FDG-PET-based radiomics could provide a non-invasive method for differentiating between primary CNS lymphoma and Glioblastoma.
Politi LS, et al. Ocular adnexal lymphoma: diffusion-weighted MR imaging for differential diagnosis and therapeutic monitoring. Radiology. 2010;256(2):565–74.
Sepahdari AR, Politi LS, Aakalu VK, Kim HJ, Razek AA. Diffusion-weighted imaging of orbital masses: multi-institutional data support a 2-ADC threshold model to categorize lesions as benign, malignant, or indeterminate. AJNR Am J Neuroradiol. 2014;35(1):170–5.
Sepahdari AR, Aakalu VK, Setabutr P, Shiehmorteza M, Naheedy JH, Mafee MF. Indeterminate orbital masses: restricted diffusion at MR imaging with echo-planar diffusion-weighted imaging predicts malignancy. Radiology. 2010;256(2):554–64.
Song MK, Chung JS, Yhim HY, Lim SN, Kim SJ, Han YH, et al. Tumor necrosis and complete resection has significant impacts on survival in patients with limited-stage upper aerodigestive tract NK/T cell lymphoma. Oncotarget. 2017;8(45):79337–46.
Freedman AS, Aster JC. Clinical manifestations, pathologic features, and diagnosis of extranodal NK/T cell lymphoma, nasal type. UpToDate. A. G. Rosmarin, A. Lister, FRCP, FRCPath and FRCR. 2019.
Paes FM, Kalkanis DG, Sideras PA, Serafini AN. FDG PET/CT of extranodal involvement in non-Hodgkin lymphoma and Hodgkin disease. Radiographics. 2010;30(1):269–91.
Alnouby A, Ibraheem Nasr IM, Ali I, Rezk M. F-18 FDG PET-CT versus contrast enhanced CT in detection of extra nodal involvement in patients with lymphoma. Indian J Nucl Med. 2018;33(3):183–9.
Krishnan A, et al. Primary bone lymphoma: radiographic-MR imaging correlation. RadioGraphics. 2003;23(6):1371–83 discussion 1384-1377.
Lim CY, Ong KO. Imaging of musculoskeletal lymphoma. Cancer Imaging. 2013;13(4):448–57.
Navarro SM, Matcuk GR, Patel DB, Skalski M, White EA, Tomasian A, et al. Musculoskeletal imaging findings of hematologic malignancies. RadioGraphics. 2017;37(3):881–900.
Genovese E, et al. F. Radiol Med. 2011;116(4):644–56.
Juan YH, Saboo SS, Tirumani SH, Khandelwal A, Shinagare AB, Ramaiya N, et al. Malignant skin and subcutaneous neoplasms in adults: multimodality imaging with CT, MRI, and 18F-FDG PET/CT. AJR Am J Roentgenol. 2014;202(5):W422–38.
Messina C, et al. Primary and secondary bone lymphomas. Cancer Treat Rev. 2015;41(3):235–46.
LaCasce AS, Freedman AS. Primary lymphoma of bone. UpToDate. T. F. DeLaney and J. R. Park. 2019.
Alamdari A, Naderi N, Peiman S, Shahi F. Non-Hodgkin lymphoma with primary involvement of skeletal muscle. Int J Hematol Oncol Stem Cell Res. 2014;8(3):55–7.
Akuzawa N, et al. Malignant lymphoma in the psoas major muscle. Case Rep Hematol. 2017;2017:3902748.
Freedman AS, et al. Clinical presentation and diagnosis of non-Hodgkin lymphoma. UpToDate. A. G. Rosmarin, A. Lister, FRCP, FRCPath and FRCR. 2019.
Tokuda O, Harada Y, Matsunaga N. MRI of soft-tissue tumors: fast STIR sequence as substitute for T1-weighted fat-suppressed contrast-enhanced spin-echo sequence. AJR Am J Roentgenol. 2009;193(6):1607–14.
Carroll G, et al. Musculoskeletal lymphoma: MRI of bone or soft tissue presentations. J Med Imaging Radiat Oncol. 2013;57(6):663–73.
Suresh S, et al. Lymphoma presenting as a musculoskeletal soft tissue mass: MRI findings in 24 cases. Eur Radiol. 2008;18(11):2628–34.
Bagherani N, Smoller BR. An overview of cutaneous T cell lymphomas. F1000Res. 2016; 5.
Pfau D, et al. Primary mediastinal large B-cell lymphoma: a review for radiologists. AJR Am J Roentgenol. 2019;213(5):W194–w210.
Priola AM, Priola SM, Gned D, Giraudo MT, Veltri A. Nonsuppressing normal thymus on chemical-shift MR imaging and anterior mediastinal lymphoma: differentiation with diffusion-weighted MR imaging by using the apparent diffusion coefficient. Eur Radiol. 2018;28(4):1427–37.
Chen Y, Chen A, Jiang H, Zhang Y, Zhu L, Xia C, et al. HRCT in primary pulmonary lymphoma: can CT imaging phenotypes differentiate histological subtypes between mucosa-associated lymphoid tissue (MALT) lymphoma and non-MALT lymphoma? J Thorac Dis. 2018;10(11):6040–9.
Wang Y, et al. Patients with pulmonary B-cell non-Hodgkin’s lymphoma: characteristic CT findings and role in predicting the survival. 2019.
Kashefi A, Kuo J, Shelton DK. Molecular imaging in pulmonary diseases. AJR Am J Roentgenol. 2011;197(2):295–307.
Kumar A, et al. To study the efficacy of thallium-201 as tumor seeking agent and to study its role in therapeutic response. Med J Armed Forces India. 2017;73(3):242–9.
Trenker C, et al. Contrast-enhanced ultrasound in pulmonary lymphoma: a small pilot study. J Ultrasound Med. 2018;37(12):2943–7.
Juanpere S, et al. A diagnostic approach to the mediastinal masses. Insights Imaging. 2013;4(1):29–52.
Freedman AS, et al. Primary mediastinal large B cell lymphoma. UpToDate. A. G. Rosmarin, A. Lister, FRCP, FRCPath and FRCR. 2019.
Milgrom SA, Elhalawani H, Lee J, Wang Q, Mohamed ASR, Dabaja BS, et al. A PET radiomics model to predict refractory mediastinal Hodgkin lymphoma. Sci Rep. 2019;9(1):1322.
• Ceriani L, Barrington S, Biggi A, Malkowski B, Metser U, Versari A, et al. Training improves the interobserver agreement of the expert positron emission tomography review panel in primary mediastinal B-cell lymphoma: interim analysis in the ongoing international extranodal lymphoma study Group-37 study. Hematol Oncol. 2017;35(4):548–53. This study demonstrates the novelty of using radiomic parameters for risk stratification in the management of lymphoma.
Bligh MP, Borgaonkar JN, Burrell SC, MacDonald D, Manos D. Spectrum of CT findings in thoracic extranodal non-Hodgkin lymphoma. Radiographics. 2017;37(2):439–61.
Bi L, et al. Pulmonary MALT lymphoma: a case report and review of the literature. Exp Ther Med. 2015;9(1):147–50.
Hare SS, Souza CA, Bain G, Seely JM, Frcpc, Gomes MM, et al. The radiological spectrum of pulmonary lymphoproliferative disease. Br J Radiol. 2012;85(1015):848–64.
Kaplan LD, Ai W. HIV-related lymphomas: clinical manifestations and diagnosis. UpToDate. A. G. Rosmarin and A. S. Freedman. 2019.
Ko JP, et al. Approach to peribronchovascular disease on CT. Semin Ultrasound CT MR. 2019;40(3):187–99.
Saboo SS, Krajewski KM, O'Regan KN, Giardino A, Brown JR, Ramaiya N, et al. Spleen in haematological malignancies: spectrum of imaging findings. Br J Radiol. 2012;85(1009):81–92.
Kaushik C, et al. Unusual CT and MR imaging characteristics of splenic lymphoma. Case Rep Oncol Med. 2011;2011:298357.
Kellenberger CJ, Epelman M, Miller SF, Babyn PS. Fast STIR whole-body MR imaging in children. Radiographics. 2004;24(5):1317–30.
Buck AK, et al. Molecular imaging of proliferation in malignant lymphoma. Cancer Res. 2006;66(22):11055–61.
Rajesh S, Bansal K, Sureka B, Patidar Y, Bihari C, Arora A. The imaging conundrum of hepatic lymphoma revisited. Insights Imaging. 2015;6(6):679–92.
Padhan RK, Das P, Shalimar. Primary hepatic lymphoma. Trop Gastroenterol. 2015;36(1):14–20.
Colagrande S, Calistri L, Grazzini G, Nardi C, Busoni S, Morana G, et al. MRI features of primary hepatic lymphoma. Abdom Radiol (NY). 2018;43(9):2277–87.
Frampas E. Lymphomas: basic points that radiologists should know. Diagn Interv Imaging. 2013;94(2):131–44.
Chou CK, Chen LT, Sheu RS, Yang CW, Wang ML, Jaw TS, et al. MRI manifestations of gastrointestinal lymphoma. Abdom Imaging. 1994;19(6):495–500.
Bronowicki JP, Bineau C, Feugier P, Hermine O, Brousse N, Oberti F, et al. Primary lymphoma of the liver: clinical-pathological features and relationship with HCV infection in French patients. Hepatology. 2003;37(4):781–7.
Masood A, Kairouz S, Hudhud KH, Hegazi AZ, Banu A, Gupta NC. Primary non-Hodgkin lymphoma of liver. Curr Oncol. 2009;16(4):74–7.
Hwang SH, Yu JS, Chung JJ, Kim JH, Kim KW. Diagnosing small hepatic cysts on multidetector CT: an additional merit of thinner coronal reformations. Korean J Radiol. 2011;12(3):341–50.
Li Q, Zhang YY, Chiu S, Hu Z, Lan KH, Cha H, et al. Integrative functional genomics of hepatitis C virus infection identifies host dependencies in complete viral replication cycle. PLoS Pathog. 2014;10(5):e1004163.
Manzella A, et al. Abdominal manifestations of lymphoma: spectrum of imaging features. ISRN Radiol. 2013;2013:483069.
Rad N, Khafaf A, Mohammad Alizadeh AH. Primary pancreatic lymphoma: what we need to know. J Gastrointest Oncol. 2017;8(4):749–57.
Babu G, et al. Ki-67 and subtype as prognostic and predictive markers of diffuse large B-cell lymphoma. Clin Cancer Invest J. 2017;6(1):97–102.
•• Lanic H, Mareschal S, Mechken F, Picquenot JM, Cornic M, Maingonnat C, et al. Interim positron emission tomography scan associated with international prognostic index and germinal center B cell-like signature as prognostic index in diffuse large B-cell lymphoma. Leuk Lymphoma. 2012;53(1):34–42. Lanic et al. investigated the correlation between FDG PET/CT uptake values and gene set expression in DLBCL (GCB like vs ABC like).
Xie Y, Pittaluga S, Jaffe ES. The histological classification of diffuse large B-cell lymphomas. Semin Hematol. 2015;52(2):57–66.
Aide N, Talbot M, Fruchart C, Damaj G, Lasnon C. Diagnostic and prognostic value of baseline FDG PET/CT skeletal textural features in diffuse large B cell lymphoma. Eur J Nucl Med Mol Imaging. 2018;45(5):699–711.
Cottereau AS, Lanic H, Mareschal S, Meignan M, Vera P, Tilly H, et al. Molecular profile and FDG-PET/CT Total metabolic tumor volume improve risk classification at diagnosis for patients with diffuse large B-cell lymphoma. Clin Cancer Res. 2016;22(15):3801–9.
Kao CH, Tsai SC, Wang JJ, Ho YJ, Ho ST, Changlai SP. Technetium-99m-sestamethoxyisobutylisonitrile scan as a predictor of chemotherapy response in malignant lymphomas compared with P-glycoprotein expression, multidrug resistance-related protein expression and other prognosis factors. Br J Haematol. 2001;113(2):369–74.
• Camacho X, Machado CL, García MF, Gambini JP, Banchero A, Fernández M, et al. Technetium-99m- or Cy7-labeled rituximab as an imaging agent for non-Hodgkin lymphoma. Oncology. 2017;92(4):229–42. Pilot study by Camacho et al. demonstrates a paradigm for investigating radiolabeled monoclonal antibodies as a potential molecular imaging technique that can demonstrate both burden of disease and potential treatment response in NHL.
Acknowledgment
The authors of this paper would also like to thank research intern Meha Shezhad, who contributed to the literature review investigating genomics in lymphoma.
Funding
Financial support for this study was provided by a grant from the National Institute of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on T-Cell and Other Lymphoproliferative Malignancies
Rights and permissions
About this article
Cite this article
Khan, S., Naim, S., Bilwani, R. et al. Radiogenomics and Its Role in Lymphoma. Curr Hematol Malig Rep 15, 211–224 (2020). https://doi.org/10.1007/s11899-020-00577-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-020-00577-2