Abstract
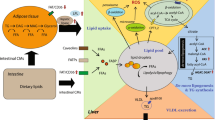
Excessive fat accumulation and increased oxidative stress contribute to the pathogenesis of nonalcoholic fatty liver disease (NAFLD). However, the mechanisms underlying the development of steatosis are not entirely understood. The present study was undertaken to establish an experimental model of hepatocellular steatosis with a fat overaccumulation profile in which the effects of oxidative stress could be studied in L-02 cells. We investigated the effects of free fatty acids (FFA) (palmitate:oleate, 1:2) on lipid accumulation and oxidative stress and their possible mechanisms in L-02 cells. High concentrations of fatty acids significantly induced excessive lipid accumulation and oxidative stress in L-02 cells, which could only be reversed with 50 μΜ WY14643 (the PPARα agonist). Immunoblotting and qPCR analyses revealed that FFA downregulated the expression of proliferator-activated receptor alpha (PPARα), which contributed to the increased activation of sterol regulatory element binding protein-1c (SREBP-1c). These results suggest that FFA induce lipid accumulation and oxidative stress in L-02 cells by upregulating SREBP-1c expression through the suppression of PPARα.





Similar content being viewed by others
Abbreviations
- NAFLD:
-
Nonalcoholic fatty liver disease
- FFA:
-
Free fatty acids
- PPARα:
-
Proliferator-activated receptor alpha
- SREBP-1c:
-
Sterol regulatory element binding protein-1c
- LDH:
-
Lactate dehydrogenase
- ROS:
-
Reactive oxygen species
- GSH:
-
Glutathione
- SOD:
-
Superoxide dismutase
- DCFH-DA:
-
2′-7′-Dichlorofluorescein diacetate
References
Hesham AH (2009) Nonalcoholic fatty liver disease in children living in the obeseogenic society. World J Pediatr 5:245–254
Trauner M, Arrese M, Wagner M (2010) Fatty liver and lipotoxicity. Biochim Biophys Acta 1801:299–310
Cohen JC, Horton JD, Hobbs HH (2011) Human fatty liver disease: old questions and new insights. Science 332:1519–1523
El-Zayadi AR (2008) Hepatic steatosis: a benign disease or a silent killer. World J Gastroenterol 14:4120–4126
Gomez-Lechon MJ, Donato MT, Castell JV, Jover R (2004) Human hepatocytes in primary culture: the choice to investigate drug metabolism in man. Curr Drug Metab 5:443–462
Gaens KH, Niessen PM, Rensen SS, Buurman WA, Greve JW, Driessen A, Wolfs MG, Hofker MH, Bloemen JG, Dejong CH, Stehouwer CD, Schalkwijk CG (2012) Endogenous formation of N epsilon-(carboxymethyl) lysine is increased in fatty livers and induces inflammatory markers in an in vitro model of hepatic steatosis. J Hepatol 56:647–655
Jiang P, Huang Z, Zhao H, Wei T (2013) Hydrogen peroxide impairs autophagic flux in a cell model of nonalcoholic fatty liver disease. Biochem Biophys Res Commun 433:408–414
Malhi H, Bronk SF, Werneburg NW, Gores GJ (2006) Free fatty acids induce JNK-dependent hepatocyte lipoapoptosis. J Biol Chem 281:12093–12101
Vidyashankar S, Sharath KL, Barooah V, Sandeep VR, Nandakumar KS, Patki PS (2012) Liv. 52 up-regulates cellular antioxidants and increase glucose uptake to circumvent oleic acid induced hepatic steatosis in HepG2 cells. Phytomedicine 19:1156–1165
Knowles BB, Howe CC, Aden DP (1980) Human hepatocellular carcinoma cell lines secrete the major plasma proteins and hepatitis B surface antigen. Science 209:497–499
Byrne CD (2010) Fatty liver: role of inflammation and fatty acid nutrition. Prostaglandins Leukot Essent Fat Acids 82:265–271
Shi LJ, Shi L, Song GY, Zhang HF, Hu ZJ, Wang C, Zhang DH (2013) Oxymatrine attenuates hepatic steatosis in non-alcoholic fatty liver disease rats fed with high fructose diet through inhibition of sterol regulatory element binding transcription factor 1 (Srebf1) and activation of peroxisome proliferator activated receptor alpha (Pparalpha). Eur J Pharmacol 714:89–95
Tailleux A, Wouters K, Staels B (2012) Roles of PPARs in NAFLD: potential therapeutic targets. Biochim Biophys Acta 1821:809–818
Zheng L, Lv GC, Sheng J, Yang YD (2010) Effect of miRNA-10b in regulating cellular steatosis level by targeting PPAR-alpha expression, a novel mechanism for the pathogenesis of NAFLD. J Gastroenterol Hepatol 25:156–163
Ferre P, Foufelle F (2010) Hepatic steatosis: a role for de novo lipogenesis and the transcription factor SREBP-1c. Diabetes Obes Metab 12(Suppl 2):83–92
Granado-Serrano AB, Martin MA, Izquierdo-Pulido M, Goya L, Bravo L, Ramos S (2007) Molecular mechanisms of (-)-epicatechin and chlorogenic acid on the regulation of the apoptotic and survival/proliferation pathways in a human hepatoma cell line. J Agric Food Chem 55:2020–2027
Wang GL, Fu YC, Xu WC, Feng YQ, Fang SR, Zhou XH (2009) Resveratrol inhibits the expression of SREBP1 in cell model of steatosis via Sirt1-FOXO1 signaling pathway. Biochem Biophys Res Commun 380:644–649
Petta S, Muratore C, Craxi A (2009) Non-alcoholic fatty liver disease pathogenesis: the present and the future. Digest Liver Dis 41:615–625
Vidyashankar S, Sandeep VR, Patki PS (2013) Quercetin ameliorate insulin resistance and up-regulates cellular antioxidants during oleic acid induced hepatic steatosis in HepG2 cells. Toxicol In Vitro 27:945–953
Vitaglione P, Morisco F, Caporaso N, Fogliano V (2004) Dietary antioxidant compounds and liver health. Crit Rev Food Sci Nutr 44:575–586
Adachi M, Ishii H (2002) Role of mitochondria in alcoholic liver injury. Free Radic Biol Med 32:487–491
Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M (2006) Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact 160:1–40
Qiao Y, Xiang Q, Yuan L, Xu L, Liu Z, Liu X (2013) Herbacetin induces apoptosis in HepG2 cells: involvements of ROS and PI3K/Akt pathway. Food Chem Toxicol 51:426–433
Malaguarnera M, Di Rosa M, Nicoletti F, Malaguarnera L (2009) Molecular mechanisms involved in NAFLD progression. J Mol Med (Berl) 87:679–695
Leung TM, Nieto N (2013) CYP2E1 and oxidant stress in alcoholic and non-alcoholic fatty liver disease. J Hepatol 58:395–398
Shin JW, Wang JH, Kim HG, Park HJ, Bok HS, Son CG (2010) CGX, a traditional Korean medicine ameliorates concanavalin A-induced acute liver injury. Food Chem Toxicol 48:3308–3315
Gonzalez FJ, Shah YM (2008) PPARalpha: mechanism of species differences and hepatocarcinogenesis of peroxisome proliferators. Toxicology 246:2–8
Mandard S, Muller M, Kersten S (2004) Peroxisome proliferator-activated receptor alpha target genes. Cell Mol Life Sci 61:393–416
Ahmed MH, Byrne CD (2007) Modulation of sterol regulatory element binding proteins (SREBPs) as potential treatments for non-alcoholic fatty liver disease (NAFLD). Drug Discov Today 12:740–747
Fernandez-Alvarez A, Alvarez MS, Gonzalez R, Cucarella C, Muntane J, Casado M (2011) Human SREBP1c expression in liver is directly regulated by peroxisome proliferator-activated receptor alpha (PPARalpha). J Biol Chem 286:21466–21477
Ziamajidi N, Khaghani S, Hassanzadeh G, Vardasbi S, Ahmadian S, Nowrouzi A, Ghaffari SM, Abdirad A (2013) Amelioration by chicory seed extract of diabetes- and oleic acid-induced non-alcoholic fatty liver disease (NAFLD)/non-alcoholic steatohepatitis (NASH) via modulation of PPARalpha and SREBP-1. Food Chem Toxicol 58:198–209
Acknowledgments
This study was supported by grants from the Natural Science Foundation of China (No. 81274160), the Natural Science Foundation of Guangdong Province (No. S2012010009380) and the Science and Technology Development project of Guangdong province (No. 2010B060900056).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
About this article
Cite this article
Qin, S., Yin, J. & Huang, K. Free Fatty Acids Increase Intracellular Lipid Accumulation and Oxidative Stress by Modulating PPARα and SREBP-1c in L-02 Cells. Lipids 51, 797–805 (2016). https://doi.org/10.1007/s11745-016-4160-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-016-4160-y