Abstract
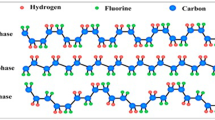
The capacitive behavior of the hydrophobic fluorinated polymeric ionic liquid, poly[2-(methacryloyloxy)ethyl]-trimethyl ammonium nonafluoro-1-butanesulfonate (poly-IL), has been investigated by current-voltage (C-V) measurements and electrochemical impedance spectroscopy (EIS) using a cell featured by a ITO/poly-IL/Al sandwich structure. The data were collected in the temperature range between 30 and 60 °C. The Nyquist plot shows a slope variation that indicates a moderate increasing of the ions mobility with temperature. The Bode diagram reveals that the sample exhibits a capacitive behavior below 104 Hz with no redox processes at the electrodes as confirmed by the C–V curves. The material shows a relatively high thermal stability, probably due to its polymeric nature making the capacitor almost insensitive to the variation of the operating temperature.



Similar content being viewed by others
References
Armand M, Endres F, MacFarlane DR, Ohno H, Scrosati B (2009) Ionic liquids materials for the electrochemical challenges of the future. Nature Mater 8:621–629. doi:10.1038/nmat2448
Smiglak M, Pringle JM, Lu X, Han L, Zhang S, Gao H, MacFarlane DR, Rogers RD (2014) Ionic liquids for energy, materials, and medicine. Chem Commun 50:9228–9250. doi:10.1039/C4CC02021A
MacFarlane DR, Tachikawa N, Forsyth M, Pringle JM, Howlett PC, Elliot GD, Davis JH Jr, Watanabe M, Simon P, Angell CA (2014) Energy applications of ionic liquids. Energy Environ Sci 7:232–250. doi:10.1039/C3EE42099J
Huddleston JG, Visser AE, Reichert WM, Willauer HD, Broker GA, Rogers RD (2001) Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation. Green Chem 3:156–164. doi:10.1039/B103275P
Appetecchi GB, Scaccia S, Tizzani C, Alessandrini F, Passerini S (2006) Synthesis of hydrophobic ionic liquids for electrochemical applications. J Electrochem Soc 153:A1685-A1691b. doi: 10.1149/1.2213420
Cardiano P, Mineo PG, Neri F, Lo Schiavo S, Piraino P (2008) A new application of ionic liquids: hydrophobic properties of tetraalkylammonium-based poly(ionic)liquids. J Mat Chem 18:1253–1260. doi:10.1039/B801227J
Bates ED, Mayton RD, Ntai I, Davis JH Jr (2002) CO2 capture by a task-specific ionic liquid. J Am Chem Soc 124:926–927. doi:10.1021/ja017593d
Bara JE, Camper DE, Gin DL, Noble RD (2010) Room-temperature ionic liquids and composite materials: platform technologies for CO2 capture. Acc Chem Res 43:152–159. doi:10.1021/ar9001747
Mineo PG, Livoti L, Giannetto M, Gulino A, Lo Schiavo S, Cardiano P (2009) Very fast CO2 response and hydrophobic properties of novel poly(ionic liquid)s. J Mater Chem 19:8861–8870. doi:10.1039/B912379B
Mineo PG, Livoti L, Lo Schiavo S, Cardiano P (2012) Fast and reversible CO2 quartz crystal microbalance response of vinylimidazolium-based poly(ionic liquid)s. Pol Adv Technol 23:1511–1519. doi:10.1002/pat.2076
Zhu S, Wu Y, Chen Q, Yu Z, Wang C, Jin S, Ding Y, Wu G (2006) Dissolution of cellulose with ionic liquids and its application: a mini-review. Green Chem 8:325–327 . doi:10.1039/B601395Cand references therein
Yuan J, Mecerreyes D, Antonietti M (2013) Poly(ionic liquid)s: an update. Prog Polym Sci 38:1009–1036. doi:10.1016/j.progpolymsci.2013.04.002
Shaplov AS, Marcilla R, Mecerreys D (2015) Recent advances in innovative polymer electolytes based on poly(ionic liquid)s. Electrochim Acta 175:18–34 . doi:10.1016/j.electacta.2015.03.038and references therein
Cardiano P, Lazzara G, Manickam S, Mineo P, Milioto S, Lo Schiavo S (2012) POSS-tetraalkylammonium salts: a new class of ionic liquids. Eur J Inorg Chem 34:5668–5676. doi:10.1002/ejic.201200874
Castriciano MA, Leone N, Cardiano P, Manickam S, Monsù Scolaro L, Lo Schiavo S (2013) A new supramolecular polyhedral oligomeric silsesquioxanes (POSS)-porphyrin nanohybrid: synthesis and spectroscopic characterization. J Mater Chem C 1:4746–4753. doi:10.1039/c3tc30532e
Cardiano P, Fazio E, Lazzara G, Manickam S, Milioto S, Neri F, Mineo PG, Piperno A, Lo Schiavo S (2015) Highly untangled multiwalled carbon nanotubes@polyhedral oligomeric silsesquioxane ionic hybrids: synthesis, characterization and nonlinear optical properties. Carbon 86:325–337. doi:10.1016/j.carbon.2015.01.047
Zailani NAM, Latif FA, Ali AMM, Zainuddin LW, Kamaruddin R, Yahya MZA (2016) Effect of ionic liquid incarceration during free radical polymerization of PMMA on its structural and electrical properties. Ionics. doi:10.1007/s11581-016-1827-6
Francis KA, Liew CW, Ramesh S, Ramesh K, Ramesh S (2016) Ionic liquid enhanced magnesium-based polymer electrolytes for electrical double-layer capacitors. Ionics 22:919. doi:10.1007/s11581-015-1619-4
Kimura K, Hassoun J, Panero S, Scrosati B, Tominaga Y (2015) Electrochemical properties of a poly(ethylene carbonate)-LiTFSI electrolyte containing a pyrrolidinium-based ionic liquid. Ionics 21:895. doi:10.1007/s11581-015-1370-x
Dong Z, Zhang Q, Yu C, Peng J, Ma J, Ju X, Zhai M (2013) Effect of ionic liquid on the properties of poly(vinylidene fluoride)-based gel polymer electrolytes. Ionics 19:1587. doi:10.1007/s11581-013-0905-2
Xu W, Ledin PA, Shevchenko VV, Tsukruk VV (2015) Architecture, assembly, and emergency applications of branched functional polyelectrolytes and poly(ionic liquid)s. ACS Appl Mater Interfaces 7:12570–12596. doi:10.1021/acsami.5b01833
Tiruye GA, Munoz-Torrero D, Palma J, Anderson M, Marcilla R (2015) All-solid state supercapacitors operating at 3.5 V by using ionic liquids based polymer electrolytes. J Power Sources 279:472–480. doi:10.1016/j.jpowsour.2015.01.039
Pandey GP, Hashmi SA (2013) Ionic liquid 1-ethyl-3-methylimidazolium tetracyanoborate-based gel polymer electrolyte for electrochemical capacitors. J Mater Chem A 1:3372–3378. doi:10.1039/c2ta01347a
Misra R, McCarthy M, Hebard AF (2007) Electric field gating with ionic liquids. App Phys Lett 90:052905. doi:10.1063/1.2437663
Wang G, Zhang L, Zhang J (2012) A review of electrode materials for electrochemical supercapacitors. Chem Soc Rev 41:797–828. doi:10.1039/C1CS15060J
Orazem ME, Tribollet B (2008) Electrochemical impedance spectroscopy. John Wiley & Sons, Inc., Hoboken
Lee KH, Zhang S, Lodge TP, Frisbie CD (2011) Electrical impedance of spin-coatable ion gel films. J Phys Chem B 115:3315–3321. doi:10.1021/jp110166u
Liew C-W, Ramesh S, Arof AK (2014) Good prospect of ionic liquid based-poly(vinyl alcohol) polymer electrolytes for supercapacitors with excellent electrical, electrochemical and thermal properties. Int J Hydrog Energ 39:2953–2963. doi:10.1016/j.ijhydene.2013.06.061
Choi JH, Gu Y, Hong K, Xie W, Frisbie CD, Lodge TP (2014) High capacitance, photo-patternable ion gel gate insulators compatible with vapor deposition of metal gate electrodes. ACS Appl Mater Interfaces 6:19275–19281. doi:10.1021/am505298q
Rafik F, Gualous H, Gallay R, Crausaz A, Berthon A (2007) Frequency, thermal and voltage supercapacitor characterization and modeling. J Power Sources 165:928–934. doi:10.1016/j.jpowsour.2006.12.021
Cho JH, Lee J, He Y, Kim BS, Lodge TP, Frisbie CD (2008) High-capacitance ion gel gate dielectrics with faster polarization response times for organic thin film transistors. Adv Mater 20:686–690. doi:10.1002/adma.200701069
Panzer MJ, Frisbie CD (2006) High carrier density and metallic conductivity in poly(3-hexylthiophene) achieved by electrostatic charge injection. Adv Funct Mater 16:1051–1056. doi:10.1002/adfm.200600111
Fujimoto T, Awaga K (2013) Electric-double-layer field-effect transistors with ionic liquids. Phys Chem Chem Phys 15:8983–9006. doi:10.1039/C3CP50755F
Zhang S, Lee KH, Frisbie CD, Lodge TP (2011) Ionic conductivity, capacitance, and viscoelastic properties of block copolymer-based ion gels. Macromolecules 44:940–949. doi:10.1021/ma102435a
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patanè, S., Triolo, C., Cardiano, P. et al. Capacitive properties of the hydrophobic [2-(methacryloyloxy)ethyl]-trimethyl ammonium nonafluoro-1-butanesulfonate poly(ionic liquid) thin film. Ionics 23, 1481–1487 (2017). https://doi.org/10.1007/s11581-017-1971-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-1971-7