Abstract
Aims
This study analyses the capability of contrast-enhanced multi-detector computed tomography (MDCT) and spectrum of molecular imaging to characterize typical carcinoids (TCs) of lung and their relationship with Ki-67 index.
Materials and methods
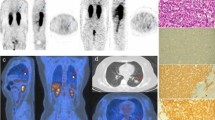
We analysed 68 patients with histological diagnosis of pulmonary TC, which underwent both MDCT and nuclear molecular imaging (somatostatin receptor scintigraphy/SPECT with 111In-pentetreotide and 18F-FDG-PET/CT) at staging evaluation before surgery. The MDCT scan was reviewed for the following features: size, margins, contrast enhancement, presence of calcifications, bronchial obstruction, lymph nodes and metastases. In 111In-pentetreotide SPECT, tumour/non-tumour ratio was measured at 4- and 24-h post-injection and the per cent difference was calculated (T/NT%). FDG uptake was measured as the ratio between lesion SUVmax and liver SUVmean (SUV ratio). All imaging features were correlated between them and with Ki-67 index.
Results
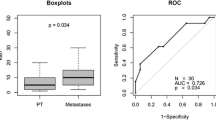
Forty-four of the 68 lesions (65%) were in the right lung. In MDCT, scan lesions appeared as a well-defined nodule in 44 patients (65%) and irregular mass in 24 patients (35%). Contrast intense enhancement was present in 53 patients (78%), calcifications in 20 patients (29%) and bronchial obstruction in 24 patients (35%). Lymph nodes and metastasis were present in 13 (19%) and 15 (22%) patients. Ki-67 index was negatively correlated with T/NT% and positively with SUV ratio; T/NT% and SUV ratio were inversely correlated. The presence of irregular margins and metastases was negatively related to T/NT%. The presence of a mass, irregular margins, bronchial obstruction, lymph nodes and metastasis was positively related to higher SUV ratio. The presence of irregular margins, bronchial obstruction, lymph nodes and metastases was significantly correlated with a higher grade of Ki-67 index.
Conclusions
MDCT and nuclear molecular imaging are important to characterize lung TCs. The majority of TCs appear as a well-defined nodule generally not associated with extra-thorax signs. We found a significant correlation between some MDCT aspects, nuclear medicine features and Ki-67 index. The association of MDCT and nuclear medicine imaging may be useful in predicting proliferative activity and prognosis of lung TCs.













Similar content being viewed by others
References
Klimstra DS (2016) Pathologic classification of neuroendocrine neoplasms. Hematol Oncol Clin North Am 30:1–19
Klöppel G (2017) Neuroendocrine neoplasms: dichotomy, origin and classification. Visc Med 33:324–330
Lloyd RV, Osamura RY, Klöppel G et al (2017) WHO classification of tumours of endocrine organs. IARC Press, Lyon
Bosman FT, Carneiro F, Hruban RH et al (eds) (2010) WHO classification of tumours of the digestive system, 4th edn, vol 3. IARC Press, Lyon
Hilal T (2017) Current understanding and approach to well differentiated lung neuroendocrine tumors: an update on classification and management. Ther Adv Med Oncol 9:189–199
Travis WD, Brambilla E, Nicholson AG et al (2015) The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 10:1243–1260
Pelosi G, Sonzogni A, Harari S et al (2017) Classification of pulmonary neuroendocrine tumors: new insights. Transl Lung Cancer Res 6:513–529
Kim JY, Hong SM, Ro JY (2017) Recent updates on grading and classification of neuroendocrine tumors. Ann Diagn Pathol 29:11–16
Raphael MJ, Chan DL, Law C et al (2017) Principles of diagnosis and management of neuroendocrine tumours. CMAJ 189:E398–E404
Caplin ME, Baudin E, Ferolla P et al (2015) Pulmonary neuroendocrine (Carcinoid) tumors: European neuroendocrine tumor society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoid. Ann Oncol 26:1604–1620
Danti G, Addeo G, Cozzi D et al (2019) Relationship between diagnostic imaging features and prognostic outcomes in gastrointestinal stromal tumors (GIST). Acta Biomed 90:9–19
Cozzi D, Dini C, Mungai F et al (2019) Primary pulmonary lymphoma: imaging findings in 30 cases. Radiol Med 124:1262–1269
Jeung MY, Gasser B, Gangi A et al (2002) Bronchial carcinoid tumors of the thorax: spectrum of radiologic findings. Radiographics 22:351–365
Detterbeck FC (2010) Management of carcinoid tumors. Ann Thorac Surg 89:998–1005
Grazzini G, Danti G, Cozzi D (2019) Diagnostic imaging of gastrointestinal neuroendocrine tumours (GI-NETs): relationship between MDCT features and 2010 WHO classification. Radiol Med 124(94):102
Tabaksblat EM, Langer SW, Knigge U et al (2016) Diagnosis and treatment of bronchopulmonary neuroendocrine tumours: state of the art. Acta Oncol 55:3–14
Ramirez RA, Chauhan A, Gimenez J et al (2017) Management of pulmonary neuroendocrine tumors. Rev Endocr Metab Disord 18:433–442
Wolin EM (2016) Advances in the diagnosis and management of well-differentiated and intermediate-differentiated neuroendocrine tumors of the lung. Chest 151:1141–1146
Lococo F, Treglia G, Cesario A et al (2014) Functional imaging evaluation in the detection, diagnosis, and histologic differentiation of pulmonary neuroendocrine tumors. Thorac Surg Clin 24:285–292
Cattoni M, Vallières E, Brown LM et al (2017) Is there a role for traditional nuclear medicine imaging in the management of pulmonary carcinoid tumours? Eur J Cardiothorac Surg 51:874–879
Stefani A, Franceschetto A, Nesci J et al (2013) Integrated FDG-PET/CT imaging is useful in the approach to carcinoid tumors of the lung. J Cardiothorac Surg 8:223
Squires MH 3rd, Volkan Adsay N, Schuster DM et al (2015) Octreoscan versus FDG-PET for neuroendocrine tumor staging: a biological approach. Ann Surg Oncol 22:2295–2301
Öberg K, Sundin A (2016) Imaging of neuroendocrine tumors. Front Horm Res 45:142–151
Chansky K, Detterbeck FC, Nicholson AG et al (2017) The IASLC lung cancer staging project: external validation of the revision of the TNM stage groupings in the eighth edition of the TNM classification of lung cancer. J Thorac Oncol 12:1109–1121
Hendifar AE, Marchevsky AM, Tuli R (2017) Neuroendocrine tumors of the lung: current challenges and advances in the diagnosis and management of well-differentiated disease. J Thorac Oncol 12:425–436
Pusceddu S, Lo Russo G, Macerelli M et al (2016) Diagnosis and management of typical and atypical lung carcinoids. Crit Rev Oncol Hematol 100:167–176
Phan AT, Oberg K, Harrison LH Jr et al (2010) NANETS consensus guideline for the diagnosis and management of neuroendocrine tumors: well-differentiated neuroendocrine tumors of the thorax (includes lung and thymus). Pancreas 39:784–798
Briganti V, Matteini M, Ferri P et al (2001) Octreoscan SPET evaluation in the diagnosis of pancreas neuroendocrine tumors. Cancer Biother Radiopharm 16:515–524
Cascini GL, Cuccurullo V, Tamburrini O et al (2013) Peptide imaging with somatostatin analogues: more than cancer probes. Curr Radiopharm 6:36–40
Cuccurullo V, Faggiano A, Scialpi M et al (2012) Questions and answers: what can be said by diagnostic imaging in neuroendocrine tumors? Minerva Endocrinol 37:367–377
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest related to the publication of this article. No funding was received for this study.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study was approved by the ethics committee on 30 May 2019. The protocol number is 14776_oss.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Danti, G., Berti, V., Abenavoli, E. et al. Diagnostic imaging of typical lung carcinoids: relationship between MDCT, 111In-Octreoscan and 18F-FDG-PET imaging features with Ki-67 index. Radiol med 125, 715–729 (2020). https://doi.org/10.1007/s11547-020-01172-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-020-01172-4