Abstract
Corrosion inhibitors have offered new opportunities to bring positive impacts on our society, especially when it has helped in protecting metals against corrosion in an aqueous solution. Unfortunately, the commonly known corrosion inhibitors used to protect metals or alloys against corrosion are invariably related to one or more drawbacks such as the employment of hazardous anti-corrosion agents, leakage of anti-corrosion agents in aqueous solution, and high solubility of anti-corrosion agents in water. Over the years, using food additives as anti-corrosion agents have drawn interest as it offers biocompatibility, less toxic, and promising applications. In general, food additives are considered safe for human consumption worldwide, and it was rigorously tested and approved by the US Food and Drug Administration. Nowadays, researchers are more interested in innovating and using green, less toxic, and economical corrosion inhibitors in metal and alloy protection. As such, we have reviewed the use of food additives to protect metals and alloys against corrosion. The current review is significant and differs from the previous review articles made on corrosion inhibitors, in which the new role of food additives is highlighted as green and environmental-friendly substances in the protection of metals and alloys against corrosion. It is anticipated that the next generation will be utilizing non-toxic and sustainable anti-corrosion agents, in which food additives might be the potential to fulfill the green chemistry goals.
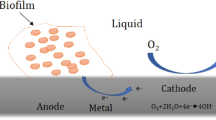
Graphical Abstract









Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- AA:
-
Ascorbic acid
- Cu:
-
Copper
- EIS:
-
Electrochemical impedance spectroscopy
- FDA:
-
Food and drug administration
- HCl:
-
Hydrochloric acid
- H2SO4 :
-
Sulfuric acid
- IR:
-
Infrared
- K:
-
Kelvin
- KCl:
-
Potassium chloride
- M:
-
Molarity
- Min:
-
Minute
- NaCl:
-
Sodium chloride
- Na2CO3 :
-
Sodium carbonate
- nm:
-
Nanometer
- NPs:
-
Nanoparticles
- OPCW:
-
Organization for the prohibition of chemical weapons
- PDP:
-
Potentiodynamic polarization
- pH:
-
Potential of hydrogen
- SEM:
-
Scanning electron microscopy (SEM)
- SHMP:
-
Sodium hexametaphosphate
References
Abdel-Karim AM, El-Shamy AM (2022) A review on green corrosion inhibitors for protection of archeological metal artifacts. J Bio- Tribo-Corros 8:1–21. https://doi.org/10.1007/s40735-022-00636-6
Abdelwedoud BO, Damej M, Tassaoui K, Berisha A, Tachallait H, Bougrin K, Mehmeti V, Benmessaoud M (2022) Inhibition effect of N-propargyl saccharin as corrosion inhibitor of C38 steel in 1 M HCl, experimental and theoretical study. J Mol Liq. 354:118784. https://doi.org/10.1016/j.molliq.2022.118784
Abdulhessein BA, Ali AM (2021) Green corrosion inhibitor of low carbon steel in acidic media by pectin nanoparticles using potentiostatic and weight losses method. Mater Sci Forum 1021. Trans Tech Publications Ltd. https://doi.org/10.4028/www.scientific.net/msf.1021.241
Abou-Elseoud WS, Abdel-karim AM, Hassan EA, Hassan ML (2021) Enzyme-and acid-extracted sugar beet pectin as green corrosion inhibitors for mild steel in hydrochloric acid solution. Carbohydr Polym Technol App 2:100072. https://doi.org/10.1016/j.carpta.2021.100072
Ahmad Ruslan NAA, Kan SY, Hamzah AS, Chia PW (2021) Natural food additives as green catalysts in organic synthesis: a review. Environ Chem Lett 19:3359–3380. https://doi.org/10.1007/s10311-021-01209-8
AL-Amouri A, Dwivedi PB (2019) Experimental study on ascorbic acid aditive as green inhibitor against corrosion of mild steel. Green Chem Technol Lett 5:1–9. https://doi.org/10.18510/gctl.2019.511
Al-Nami SY, Al-Qahtani SD, Snari RM, Ibarhiam SF, Alfi AA, Aldawsari AM, El-Metwaly NM (2022) Preparation of photoluminescent and anticorrosive epoxy paints immobilized with nanoscale graphene from sugarcane bagasse agricultural waste. Environ Sci Pollut Res 29:60173–60188. https://doi.org/10.1007/s11356-022-20111-6
Alrefaee SH, Rhee KY, Verma C, Quraishi MA, Ebenso EE (2021) Challenges and advantages of using plant extract as inhibitors in modern corrosion inhibition systems: Recent advancements. J Mol Liq 321:114666. https://doi.org/10.1016/j.molliq.2020.114666
Anastassiadis S, Rehm HJ (2006) Citric acid production from glucose by yeast Candida oleophila ATCC 20177 under batch, continuous and repeated batch cultivation. Electron J Biotechnol 9:26–39. https://doi.org/10.2225/vol9-issue1-fulltext-5
Anejjar A, El Mouden OI, Batah A, Bouskri A, Rjoub A (2017) Corrosion inhibition potential of ascorbic acid on carbon steel in acid media. Appl J Environ Eng 3:3–1. https://doi.org/10.48422/IMIST.PRSM/ajees-v3i1.7432
Asiya SI, Pal K, Kralj S, El-Sayyad GS, de Souza FG, Narayanan T (2020) Sustainable preparation of gold nanoparticles via green chemistry approach for biogenic applications. Mater Today Chem 17:100327
Asseri SNARM, Tan SH, Mohamad WNKW, Poh SC, Chia PW, Kan SY, Chuah TS (2017) MgCl2 as efficient and inexpensive catalyst for the synthesis of 1, 4-dihydropyridine derivatives. Malay J Anal Sci 21:13–19. https://doi.org/10.17576/mjas-2017-2101-02
Balakrishnan A, Jena G, Pongachira George R, Philip J (2021) Polydimethylsiloxane–graphene oxide nanocomposite coatings with improved anti-corrosion and anti-biofouling properties. Environ Sci Pollut Res 28:7404–7422. https://doi.org/10.1007/s11356-020-11068-5
Barrodi MR, Mirzaee A, Kafashan A, Zahedifard S, Majidi HJ, Davoodi A, Hosseinpour S (2023) Synergistic effect in tragacanth gum-ceftriaxone hybrid system as an environmentally friendly corrosion inhibitor for mild steel in acidic solutions. Mater Today Commun 105390. https://doi.org/10.2139/ssrn.4236211
Bhola SM, Gurmeet S, Brajendra M (2013) Flavin mononucleotide as a corrosion inhibitor for hot rolled steel in hydrochloric acid. Int J Electrochem Sci 8(4):5635–5642
Charitha BP, Rao P (2020) Pectin as a potential green inhibitor for corrosion control of 6061Al–15%(V) SiC(P) composite in acid medium: electrochemical and surface studies. J Fail Anal Preven 20:1684–1696. https://doi.org/10.1007/s11668-020-00973-z
Chidiebere MA, Oguzie EE, Liu L, Li Y, Wang F (2015) Ascorbic acid as corrosion inhibitor for Q235 mild steel in acidic environments. J Ind Eng Chem 26:182–192. https://doi.org/10.1016/j.jiec.2014.11.029
Dang N, Wei YH, Hou LF, Li YG, Guo CL (2015) Investigation of the inhibition effect of the environmentally friendly inhibitor sodium alginate on magnesium alloy in sodium chloride solution. Mater Corros 66:1354–1362. https://doi.org/10.1002/maco.201408141
David J, Helen Z, Zhao XJ (2014) Corrosion inhibition of sodium hexametaphosphate for carbon steel in KCl Solution. Adv Mater Res 886:12–15. https://doi.org/10.4028/www.scientific.net/AMR.886.12
Elhousni L, Galai M, Elkamraoui FZ, Dkhireche N, Touhami ME, Touir R, Chebabe D, Sfaira M, Zarrouk A (2016) Study of sodium gluconate and cetyltrimethyl ammonium bromide as inhibitor for copper in Moroccan industrial cooling water systems. J Mater Environ Sci 7:2513–2525
Elsharif AM, Abubshait SA, Abdulazeez I, Abubshait HA (2020) Synthesis of a new class of corrosion inhibitors derived from natural fatty acid: 13-Docosenoic acid amide derivatives for oil and gas industry. Arab J Chem 13:5363–5376. https://doi.org/10.1016/j.arabjc.2020.03.015
Ferreira ES, Giacomelli C, Giacomelli FC, Spinelli A (2004) Evaluation of the inhibitor effect of L-ascorbic acid on the corrosion of mild steel. Mater Chem Phys 83:129–134. https://doi.org/10.1016/j.matchemphys.2003.09.020
Fuadi A (2019) Investigation of ascorbic acid as environment-friendly corrosion inhibitor of low carbon steel in marine environment. IOP Conf Ser Mater Sci Eng 536:012108. https://doi.org/10.1088/1757-899X/536/1/012108
Ganasan E, Mohd Yusoff H, Azmi AA, Chia PW, Lam SS, Kan SY, Liew RK, Venkateswarlu K, Teo CK (2023) Food additives for the synthesis of metal nanoparticles: a review. Environ Chem Lett 21:525–538. https://doi.org/10.1007/s10311-022-01473-2
Golonka RM, Yeoh BS, Vijay-Kumar M (2019) Dietary additives and supplements revisited: the fewer, the safer for gut and liver health. Curr Pharmacol Rep 5:303–316. https://doi.org/10.1007/s40495-019-00187-4
Hanoon M, Zinad DS, Resen AM, Al-Amiery AA (2020) Gravimetrical and surface morphology studies of corrosion inhibition effects of a 4-aminoantipyrine derivative on mild steel in a corrosive solution. Int J Corros Scale Inhibit 9:953–966. https://doi.org/10.17675/2305-6894-2020-9-3-10
Irwan, Fuadi A, Suhendrayatna (2019) Investigation of ascorbic acid as environment-friendly corrosion inhibitor of low carbon steel in marine environment. IOP Conf Ser: Mater Sci Eng 536:012108. https://doi.org/10.1088/1757-899X/536/1/012108
Ivušić F, Lahodny-Šarc O, Stojanović I (2014) Corrosion inhibition of carbon steel in saline solutions by gluconate, zinc sulphate and green clay eluate. Tehn Vjesn 21:107–114
Jmiai A, Ibrahim BE, Tara A et al (2019) The effect of the two biopolymers “sodium alginate and chitosan” on the inhibition of copper corrosion in 1 M hydrochloric acid. Mater Today: Proc 22:12–15. https://doi.org/10.1016/j.matpr.2019.08.057
Kaya AI (2022) Determination of chemical structure, mechanical properties and combustion resistance of polyurethane doped with boric acid. J Thermoplas Compos Mater 35:720–739. https://doi.org/10.1177/08927057211019444
Kobotiatis L, Tsikrikas C, Koutsoukos PG (1997) Enhanced pitting corrosion resistance of aluminum alloy 7075 in the presence of oxalate anions. Corrosion 53(1997):562–570
Kumar A, Kumar J (2022) Natural gums as corrosion inhibitor: a review. Mater Today: Proc 64:141–146. https://doi.org/10.1016/j.matpr.2022.04.073
Kusmierek E, Chrzescijanska E (2013) Tannic acid as corrosion inhibitor for metals and alloys. Mater Corros 66:169–174. https://doi.org/10.1002/maco.201307277
Kyohei O, Masatoshi S (2021) Synergistic effects of metal cations and sodium gluconate on the inhibition of freshwater corrosion of mild steel. Mater Trans 62:815–820. https://doi.org/10.2320/matertrans.MT-M2021002
Laudisi F, Stolfi C, Monteleone G (2019) Impact of food additives on gut homeostasis. Nutrients 11:2334. https://doi.org/10.3390/nu11102334
Lee SH, Md Tahir P, Lum WC, Tan LP, Bawon P, Park BD, Al Edrus SSO, Abdullah UH (2020) A review on citric acid as green modifying agent and binder for wood. Polymers 12:1692. https://doi.org/10.3390/polym12081692
Loto RT, Loto CA (2019) Data on the corrosion inhibition effect of non-toxic organic derivatives on high carbon steel in dilute acid media. Chem Data Collect 20:100214. https://doi.org/10.1016/j.cdc.2019.100214
Malarvizhi M, Jagananthan M, Sivakumar D (2017) Gum tragacanth powder as A green corrosion inhibitor for mild steel in 1N sulphuric acid solution. Int J Inno Sci Res Technol 2:132–140
Marzorati S, Verotta L, Trasatti SP (2018) Green corrosion inhibitors from natural sources and biomass wastes. Molecules 24:48. https://doi.org/10.3390/molecules24010048
Miralrio A, Espinoza Vázquez A (2020) Plant extracts as green corrosion inhibitors for different metal surfaces and corrosive media: a review. Processes 8:942. https://doi.org/10.3390/pr8080942
Mobin M, Marziya R, Lukman O, Ebenso E (2017) Biopolymer from tragacanth gum as a green corrosion inhibitor for carbon steel in 1 M HCl solution. ACS Omega 2:3997–4008. https://doi.org/10.1021/acsomega.7b00436
Nezha KB, Latefa S, Abdelillah B (2022) Application of ascorbic acid as green corrosion inhibitor of reinforced steel in concrete pore solutions contaminated with chlorides. J Adhes Sci Technol 36:1176–1199. https://doi.org/10.1080/01694243.2021.1962090
Obot IB, Onyeachu I, Madhan AK (2017) Sodium alginate: a promising biopolymer for corrosion protection of API X60 high strength carbon steel in saline medium. J Carbohydr Polym 178:200–208. https://doi.org/10.1016/j.carbpol.2017.09.049
Pal K, Asthana N, Aljabali AA, Bhardwaj SK, Kralj S, Penkova A, Thomas S, Zaheer T, Gomes de Souza F (2022a) A critical review on multifunctional smart materials nanographene emerging avenue: nano-imaging and biosensor applications. Crit Rev Solid State Mater Sci 47:691–707. https://doi.org/10.1080/10408436.2021.1935717
Pal K, Chakroborty S, Panda P, Nath N, Soren S (2022b) Environmental assessment of wastewater management via hybrid nanocomposite matrix implications—an organized review. Environ Sci Pollut Res 29:76626–76643. https://doi.org/10.1016/j.nanoso.2017.12.003
Park TJ, Jang H (2021) Effects of inhibitors on corrosion resistance of steel in CaCl2 solution based on response surface analysis. Corros Sci Technol 20:129–142. https://doi.org/10.14773/cst.2021.20.3.129
Partridge D, Lloyd KA, Rhodes JM, Walker AW, Johnstone AM, Campbell BJ (2019) Food additives: assessing the impact of exposure to permitted emulsifiers on bowel and metabolic health–introducing the FADiets study. Nutr Bull 44:329–349. https://doi.org/10.1111/nbu.12408
Qian B, Hou B, Zheng M (2013) The inhibition effect of tannic acid on mild steel corrosion in seawater wet/dry cyclic conditions. Corros Sci 72:1–9. https://doi.org/10.1016/j.corsci.2013.01.040
Qu JE, Luo H, Liu Z, Wang H, Chen Y, Yang L (2022) Effect of sodium-zinc EDTA and sodium gluconate as electrolyte additives on corrosion and discharge behavior of Mg as anode for air battery. Mater Corros 73:1776–1787. https://doi.org/10.1002/maco.202213322
Rani BE, Basu BBJ (2012) Green inhibitors for corrosion protection of metals and alloys: an overview. Int J Corros 2012:1–16. https://doi.org/10.1155/2012/380217
Rico-Yuson CA, Hornyak GL, Bora T (2021) Cyanide-free environment-friendly alternative to copper electroplating for zinc die-cast alloys. Environ Sci Pollut Res 28:38065–38073. https://doi.org/10.1007/s11356-021-13398-4
Sanni O, Popoola AP (2017) Gluconates as corrosion inhibitor of aluminum in various corrosive media. https://doi.org/10.5772/intechopen.71467
Schweiggert RM (2018) Perspective on the ongoing replacement of artificial and animal-based dyes with alternative natural pigments in foods and beverages. J Agric Food Chem 66:3074–3081. https://doi.org/10.1021/acs.jafc.7b05930
Shilova OA, Tsvetkova IN, Vlasov DY, Ryabusheva YV, Sokolov GS, Kychkin AK, Nguyên CV, Khoroshavina YV (2022) Microbiologically induced deterioration and environmentally friendly protection of wood products. In Biodegradation and biodeterioration at the nanoscale. Elsevier, pp. 283–321. https://doi.org/10.1016/B978-0-12-823970-4.00013-0
Soraya N, Rayenne D, Boulanouar M, Rabah O (2018) Structurecorrosion inhibition performance relationship: application to some natural free acids and antioxidants. Port Electrochim 36:23–34
Tang H, Chen X, Gao Y, Hong L, Chen Y (2020) Alteration in root morphological and physiological traits of two maize cultivars in response to phosphorus deficiency. Rhizosphere 14:100201. https://doi.org/10.1016/j.rhisph.2020.100201
Umoren SA, Obot IB, Madhankumar A, Gasem ZM (2015) Performance evaluation of pectin as ecofriendly corrosion inhibitor for X60 pipeline steel in acid medium: experimental and theoretical approaches. Carbohyd Polym 124:280–291. https://doi.org/10.1016/j.carbpol.2015.02.036
Vu VT, Dinh T, Pham NT, Nguyen T, Nguyen PT, To HT (2018). Evaluation of the corrosion inhibiting capacity of silica/polypyrrole-oxalate nanocomposite in epoxy coatings. Int J Corros 2018. https://doi.org/10.1155/2018/6395803
Wang Z, Zhou Z, Xu W, Yang D, Xu Y, Yang L, Ren J, Li Y, Huang Y (2021) Research status and development trends in the field of marine environment corrosion: a new perspective. Environ Sci Pollut Res 28:54403–54428. https://doi.org/10.1007/s11356-021-15974-0
Wang X, Ren J, Li Z, Li Y (2022) Research progress of vapor phase corrosion inhibitors in marine environment. Environ Sci Pollut Res 29:88432–88439. https://doi.org/10.1007/s11356-022-23400-2
Xhanari K, Finšgar M (2016) Organic corrosion inhibitors for aluminium and its alloys in acid solutions: a review. RSC Adv 6:62833–62857. https://doi.org/10.1039/C6RA11818F
Zhang WW, Nie B, Li H-J, Li QY, Li CY, Wu Y-C (2021) Inhibition of mild steel corrosion in 1 M HCl by chondroitin sulfate and its synergistic effect with sodium alginate. J Carbohydr Polym 260:117842. https://doi.org/10.1016/j.carbpol.2021.117842
Zin IM, Pokhmurskii VI, Korniy SA, Karpenko OV, Lyon SB, Khlopyk OP, Tymus MB (2018) Corrosion inhibition of aluminium alloy by rhamnolipid biosurfactant derived from Pseudomonas sp PS-17. Anti-Corros Method M 65:517–527. https://doi.org/10.1108/ACMM-03-2017-1775
Funding
We would like to express our sincere gratitude to the Organization For The Prohibition of Chemical Weapons (OPCW) for providing the research grant (project Account No: L/ICA/ICB-115/21) and (UMT/PPP/2–2/25 Jld. 9 (94), No. Vot: 53481).
Author information
Authors and Affiliations
Contributions
Low Ee Jian, Hanis Mohd Yusoff, Nurhanna Batar, Intan Nur Zulayqha Nor Azmi, Katta Venkateswarlu, Mohammad Fakhratul Ridwan Zulkifli, Poh Wai Chia: conceptualization, methodology, investigation. Su Shiung Lam, Su-Yin Kan, Rock Keey Liew, Gaik Ee Lee, Katta Venkateswarlu, Mohammad Fakhratul Ridwan Zulkifli: data curation, writing—original draft preparation: Poh Wai Chia: supervision.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
The corresponding author on behalf of all co-authors agreed to publish in Environmental Science and Pollution Research.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Low, E.J., Yusoff, H.M., Batar, N. et al. The use of food additives as green and environmental-friendly anti-corrosion inhibitors for protection of metals and alloys: a review. Environ Sci Pollut Res 30, 76297–76307 (2023). https://doi.org/10.1007/s11356-023-27823-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-27823-3