Abstract
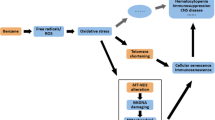
Exposure to benzene is a common occupational hazard as well as a hematopoietic system intoxicant, but the entire picture of its molecular pathogenesis is still hazy. Its leukemogenic effect could be attributed to DNA damage, decreased repair capacity, altered methylation patterns, and defective apoptosis. Poly ADP-ribose polymerase1, DNA methyltransferase1, and CCCTC-binding factor (PARP1-DNMT1-CTCF) complex play an essential role in methylation maintenance and DNA damage repair response. This study aimed to assess the expression of PARP1, PAR glycohydrolases (PARG), DNMT1, CTCF, and apoptosis-inducing factor (AIF) in subjects occupationally exposed to benzene. A total of 200 subjects were enrolled in this study: 100 workers occupationally exposed to benzene (painters and decorators) and 100 unexposed office workers. Occupational exposure data were obtained. The biochemical and hematological evaluations were done. Quantitative reverse transcription polymerase chain reaction (RT-PCR) was used to assess mRNA expression of PARP1, PARG, DNMT1, CTCF, and AIF. Both biochemical and hematological parameters were within normal limits; workplace benzene air concentration was significantly higher in exposed workers than the levels among controls (P < 0.001). Significant decrease in mRNA levels of PARP1, DNMT1, CTCF, and AIF was noticed among the exposed group (P = 0.01, P < 0.001, P = 0.004, P < 0.001, respectively) in comparison with the control group, while PARG showed non-significant difference (P = 0.16). There was a significant negative correlation between workplace benzene air concentration and expression levels of PARP1, DNMT1, and AIF. The reduced expression of PARP1, DNMT1, CTCF, and AIF observed in exposed workers may represent one of the first benzene-induced changes that might threaten erythropoiesis.







Similar content being viewed by others
References
Ame´ JC, Spenlehauer C, de Murcia G (2004) The PARP superfamily. BioEssays 26:882–893. https://doi.org/10.1002/bies.20085
Bai P (2015) Biology of poly(ADP-ribose) polymerases: the factotums of cell maintenance. Mol Cell 58(6):947–958. https://doi.org/10.1016/j.molcel.2015.01.034
Blagitko-Dorfs N, Schlosser P, Greve G, Pfeifer D, Meier R, Baude A, Brocks D, Plass C, Lübbert M (2019) Combination treatment of acute myeloid leukemia cells with DNMT and HDAC inhibitors: predominant synergistic gene downregulation associated with gene body demethylation. Leukemia 33(4):945–956. https://doi.org/10.1038/s41375-018-0293-8
Bollati V, Baccarelli A, Hou L, Bonzini M, Fustinoni S, Cavallo D, Byun HM, Jiang J, Marinelli B, Pesatori AC, Bertazzi PA, Yang AS (2007) Changes in DNA methylation patterns in subjects exposed to low-dose benzene. Cancer Res 67(3):876–880. https://doi.org/10.1158/0008-5472.CAN-06-2995
Caiafa P, Guastafierro T, Zampieri M (2009) Epigenetics: poly(ADP-ribosyl)ation of PARP-1 regulates genomic methylation patterns. FASEB J 23:672–678. https://doi.org/10.1096/fj.08-123265
Ciccaronea F, Zampieri M, Caiafa P (2017) PARP1 orchestrates epigenetic events setting up chromatin domains. Semin Cell Dev Biol 63:123–134. https://doi.org/10.1016/j.semcdb.2016.11.010
Cregan SP, Dawson VL, Slack RS (2004) Role of AIF in caspase-dependent and caspase-independent cell death. Oncogene. 23(16):2785–2796. https://doi.org/10.1038/sj.onc.1207517
De Vos M, Schreiber V, Dantzer F (2012) The diverse roles and clinical relevance of PARPs in DNA damage repair: current state of the art. Biochem Pharmacol 84:137–146. https://doi.org/10.1016/j.bcp.2012.03.018
Dorak M (2006) Real-time PCR. Clin Chem 50:1680–1693 Taylor & Francis Group. ISBN: 0–4153-7734-X
Fathallah H, Weinberg RS, Galperin Y, Sutton M, Atweh GF (2007) Role of epigenetic modifications in normal globin gene regulation and butyrate-mediated induction of fetal hemoglobin. Blood 110(9):3391–3397. https://doi.org/10.1182/blood-2007-02-076091
Fustinoni S, Rossella F, Polledri E, Bollati V, Campo L, Byun HM, Agnello L, Consonni D, Pesatori AC, Baccarelli A, Bertazzi PA (2012) Global DNA methylation and low-level exposure to benzene. Med Lav 103(2):84–95
Gao A, Zuo X, Liu Q, Lu X, Guo W, Tian L (2010) Methylation of PARP-1 promoter involved in the regulation of benzene-induced decrease of PARP-1 mRNA expression. Toxicol Lett 195(2–3):114–118. https://doi.org/10.1016/j.toxlet.2010.03.005
Gao A, Zuo X, Song S, Guo W, Tian L (2011) Epigenetic modification involved in benzene-induced apoptosis through regulating apoptosis-related genes expression. Cell Biol Int 35(4):391–396. https://doi.org/10.1042/CBI20100256
Guastafierro T, Catizone A, Calabrese R, Zampieri M, Martella O, Bacalini MG, Reale A, Di Girolamo M, Miccheli M, Farrar D, Klenova E, Ciccarone F, Caiafa P (2013) ADP-ribose polymer depletion leads to nuclear Ctcf re-localization and chromatin rearrangement (1). Biochem J 449(3):623–630. https://doi.org/10.1042/BJ20121429
Guastafierro T, Cecchinelli B, Zampieri M, Reale A, Riggio G, Sthandier O, Zupi G, Calabrese L, Caiafa P (2008) CCCTC binding factor activates PARP1 affecting DNA methylation machinery. J Biol Chem 283(32):21873–21880. https://doi.org/10.1074/jbc.M801170200
Gui Z, Zhang H, Tan Q, Ling X, Liu Z, Peng J, Shao J, Wu M, Yuan Q, Li J, Pan Z, Zhong B, Liu L (2019) Poly(ADP-ribose) polymerase-1 promotes expression of miR-155 by the upregulation of methyl-CpG binding domain protein 2 in TK6 cells exposed to hydroquinone. Toxicol in Vitro 55:51–57. https://doi.org/10.1016/j.tiv.2018.11.007
Hakme A, Wong HK, Dantzer F, Schreiber V (2008) The expanding field of poly(ADP-ribosyl)ation reactions. EMBO Rep 9(11):1094–1100. https://doi.org/10.1038/embor.2008.191
Han D, Chen Q, Shi J, Zhang F, Yu X (2017) CTCF participates in DNA damage response via poly(ADP-ribosyl)ation. Sci Rep 7:43530. https://doi.org/10.1038/srep43530
Herman JG, Baylin SB (2003) Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med 349(21):2042–2054. https://doi.org/10.1056/NEJMra023075
Hong SJ, Dawson TM, Dawson VL (2004) Nuclear and mitochondrial conversations in cell death: PARP-1 and AIF signaling. Trends Pharmacol Sci 25(5):259–264. https://doi.org/10.1016/j.tips.2004.03.005
Huang Z, Lin S, Long C, Zhou X, Fan Y, Kuang X, He J, Ning J, Zhang H, Zhang Q, Shen H (2018) Notch signaling pathway mediates doxorubicin-driven apoptosis in cancers. Cancer Manag Res 10:1439–1448. https://doi.org/10.2147/CMAR.S160315
Ishihama M, Toyooka T, Ibuki Y (2008) Generation of phosphorylated histone H2AX by benzene metabolites. Toxicol in Vitro 22(8):1861–1868. https://doi.org/10.1016/j.tiv.2008.09.005
Ji Z, Zhang L, Peng V, Ren X, McHale CM, Smith MT (2010) A comparison of the cytogenetic alterations and global DNA hypomethylation induced by the benzene metabolite, hydroquinone, with those induced by melphalan and etoposide. Leukemia 24(5):986–991. https://doi.org/10.1038/leu.2010.43
Kemp CJ, Moore JM, Moser R, Bernard B, Teater M, Smith LE, Rabaia NA, Gurley KE, Guinney J, Busch SE, Shaknovich R, Lobanenkov VV, Liggitt D, Shmulevich I, Melnick A, Filippova GN (2014) CTCF haploinsufficiency destabilizes DNA methylation and predisposes to cancer. Cell Rep 7(4):1020–1029. https://doi.org/10.1016/j.celrep.2014.04.004
Krishnakumar R, Kraus WL (2010) The PARP side of the nucleus: molecular actions, physiological outcomes, and clinical targets. Mol Cell 39(1):8–24. https://doi.org/10.1016/j.molcel.2010.06.017
Li Y, Wu XR, Li XF, Suriguga YCH, Li YR, Yi ZC (2013) Changes in DNA methylation of erythroid-specific genes in K562 cells exposed to phenol and hydroquinone. Toxicology 312:108–114. https://doi.org/10.1016/j.tox.2013.08.007
Liu L, Ling X, Liang H, Gao Y, Yang H, Shao J, Tang H (2012) Hypomethylation mediated by decreased DNMTs involves in the activation of proto-oncogene MPL in TK6 cells treated with hydroquinone. Toxicol Lett 209(3):239–245. https://doi.org/10.1016/j.toxlet.2011.12.020
Peng C, Ng JC (2016) The role of epigenetic changes in benzene-induced acute myeloid leukaemia. J Clin Epigenet 2:2–19. https://doi.org/10.21767/2472-1158.100019
Razin A, Kantor B (2005) DNA methylation in epigenetic control of gene expression. Prog Mol Subcell Biol 38:151–167. https://doi.org/10.1007/3-540-27310-7_6
Schuhwerk H, Bruhn C, Siniuk K, Min W, Erener S, Grigaraviciu P, Kruger A, Ferrari E, Zubel T, Lazaro D, Monajembashi S, Kiesow K, Kroll T, Burkle A, Mangerich A, Hottiger M, Wang ZQ (2017) Kinetics of poly(ADP-ribosyl)ation, but not PARP1 itself, determines the cell fate in response to DNA damage in vitro and in vivo. Nucleic Acids Res 45(19):11174–11192. https://doi.org/10.1093/nar/gkx717
Seow WJ, Pesatori AC, Dimont E, Farmer PB, Albetti B, Ettinger ES, Bollati V, Bolognesi C, Roggieri P, Panev TI, Georgiva T, Merlo DF, Bertazzi PA, Baccarilli AA (2012) Urinary benzene biomarkers and DNA methylation in Bulgarian petrochemical workers: study findings and comparison of linear and beta regression models. PLoS one 7(12):e50471. https://doi.org/10.1371/journal.pone.0050471
Sha Y, Zhou W, Yang Z, Zhu X, Xiang Y, Li T, Zhu D, Yang X (2014) Changes in poly(ADP-ribosyl)ation patterns in workers exposed to BTX. PLoS One 9(9):e106146. https://doi.org/10.1371/journal.pone.0106146
Sha Y, Zhou W, Yang Z, Zhu X, Yang X, Li TD, Zhu D (2015) Down-regulation of poly (ADP-ribose) polymerase 1 leads to change of hydroquinone cytotoxicity in TK6 cells. Toxicol Mech Methods 25(6):467–477. https://doi.org/10.3109/15376516.2015.1070222
Sun B, Shi Y, Yang X, Zhao T, Duan J, Sun Z (2018) DNA methylation: a critical epigenetic mechanism underlying the detrimental effects of airborne particulate matter. Ecotoxicol Environ Saf 161:173–183. https://doi.org/10.1016/j.ecoenv.2018.05.083
Tabish AM, Poels K, Hoet P, Godderis L (2012) Epigenetic factors in cancer risk: effect of chemical carcinogens on global DNA methylation pattern in human TK6 cells. PLoS One 7(4):e34674. https://doi.org/10.1371/journal.pone.0034674
The International Agency for Research on Cancer (IARC). A review of human carcinogens. F. Chemical agents and related occupations. Lyon, International Agency for Research on Cancer (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 100) [summary in Baan R et al. (2009). A review of human carcinogens—Part F: Chemical agents and related occupations. The Lancet Oncology, 10(12):1143–1144]. https://doi.org/10.1016/S1470-2045(09)70358-4
Tisdale MJ (1986) Antitumour imidazotetrazines-X, effect of 8-carbamoyl-3- methylimidazo [5,1-d]-1,2,3,5-tetrazin-4-(3H)-one (CCRG 81045; M & B 39831; NSC 362856) on DNA methylation during induction of haemoglobin synthesis in human leukaemia cell line K562. Biochem Pharmacol 35(2):311–316. https://doi.org/10.1016/0006-2952(86)90531-9
Wang E, Miller L, Ohnmacht G, Liu E, Marincola F (2000) High-fidelity mRNA amplification for gene profiling. Nat Biotechnol 18(4):457–459. https://doi.org/10.1038/74546
Xing C, Wang QF, Li B, Tian H, Ni Y, Yin S, Li G (2010) Methylation and expression analysis of tumor suppressor genes p15 and p16 in benzene poisoning. Chem Biol Interact 184(1–2):306–309. https://doi.org/10.1016/j.cbi.2009.12.028
Zampieri M, Guastafierro T, Calabrese R, Ciccarone F, Bacalini MG, Reale A, Perilli M, Passananti C, Caiafa P (2012) ADP-ribose polymers localized on Ctcf–Parp1–Dnmt1 complex prevent methylation of Ctcf target sites. Biochem J 441(2):645–652. https://doi.org/10.1042/BJ20111417
Zampieri M, Passananti C, Calabrese R, Perilli M, Corbi N, De Cave F, Guastafierro T, Bacalini MG, Reale A, Amicosante G, Calabrese L, Zlatanova J, Caiafa P (2009) Parp1 localizes within the Dnmt1 promoter and protects its unmethylated state by its enzymatic activity. PLoS One 4(3):e4717. https://doi.org/10.1371/journal.pone.0004717
Zhang L, McHale CM, Rothman N, Li G, Ji Z, Vermeulen R, Hubbard AE, Ren X, Shen M, Rappaport SM, North M, Skibola CF, Yin S, Vulpe C, Chanock SJ, Smith MT, Lan Q (2010) Systems biology of human benzene exposure. Chem Biol Interact 184(1–2):86–93. https://doi.org/10.1016/j.cbi.2009.12.011
Acknowledgments
The authors wish to acknowledge the participants who volunteered to be involved in this research, and Dr. Gaafar Abd-El Rasoul and Dr. Dian Rholman, who gave the opportunity for collaboration between the authors and the funding bodies.
Funding
This research was funded (in part) by a grant from the Fogarty Institute and the National Institute of Environmental Health Sciences (R01ES022163).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed written consent was obtained from the study subjects after illustrating the purpose of this study. The Medical Research Ethics Committee at Menoufia University had approved the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Disclaimer
The contents are the responsibility of the authors and do not represent the official views of the NIH.
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Habieb, M.S.Ed., Younis, F.E., Safan, M. et al. PARP1-DNMT1-CTCF complex and the apoptotic-induced factor mRNA expressions in workers occupationally exposed to benzene. Environ Sci Pollut Res 27, 22648–22657 (2020). https://doi.org/10.1007/s11356-020-08614-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08614-6