Abstract
Purpose
We assessed the incidence rate and management impact of oligometastatic disease detected on 18F-fluciclovine (Axumin™) PET/CT in men with first biochemical recurrence (BCR) of prostate cancer (PCA) after definitive primary therapy.
Methods and Materials
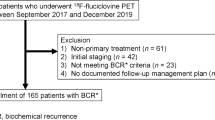
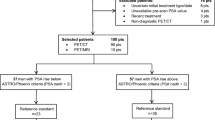
We retrospectively reviewed our clinical database for men with PCA who underwent 18F-fluciclovine PET/CT for imaging evaluation of BCR with negative or equivocal findings on conventional imaging. We included patients with up to and including 5 metastases (oligometastases) regardless of imaging evidence for local recurrence in the treated prostate bed. We examined the association between mean serum prostate specific antigen (PSA) levels with the number of oligometastases (non-parametric ANOVA) and between patients with or without local recurrence (Student t-test). The management impact of oligometastatic disease was tabulated.
Results
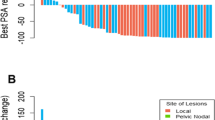
We identified 21 patients with oligometastases upon first BCR (PSA 0.2–56.8 ng/mL) out of 89 eligible patients. There was a significant difference (p = 0.04) in the mean PSA levels between patients with local recurrence (n = 12) and those without local recurrence (n = 9). In the subgroup of analysis of patients without local recurrence, there was no significant association between mean PSA level and number of oligometastases (p = 0.83). Distribution of oligometastases included 66.7% isolated nodal disease and 33.3% bone only. Twelve (57.1%) patients had change in management to include change in ADT, salvage therapy, or both. Treatment change was initiated in 62.5%, 28.6%, 66.7%, 100%, and 100% of patients with 1, 2, 3, 4, and 5 oligometastatic lesions, respectively.
Conclusion
The incidence rate of oligometastatic disease in men with first BCR of PCA undergoing 18F-fluciclovine PET/CT for imaging evaluation of BCR was 23.6% in our eligible patient population. There was no significant association between serum PSA level and the number of oligometastases. Treatment management was affected in 57.1% of patients with oligometastases.




Similar content being viewed by others
References
Artibani W, Porcaro AB, De Marco V, Cerruto MA, Siracusano S (2018) Management of biochemical recurrence after primary curative treatment for prostate cancer: a review. Urol Int 100(3):251–262
Cookson MS, Aus G, Burnett AL, Canby-Hagino ED, D’Amico AV, Dmochowski RR et al (2007) Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol 177(2):540–545
Roach M 3rd, Hanks G, Thames HJ, Schellhammer P, Shipley WU, Sokol GH et al (2006) Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int J Radiat Oncol Biol Phys 65(4):965–974
Vargas HA, Martin-Malburet AG, Takeda T, Corradi RB, Eastham J, Wibmer A et al (2016) Localizing sites of disease in patients with rising serum prostate-specific antigen up to 1ng/ml following prostatectomy: how much information can conventional imaging provide? Urol Oncol 34(11):482.e5-482.e10
Kane CJ, Amling CL, Johnstone PAS, Pak N, Lance RS, Thrasher JB et al (2003) Limited value of bone scintigraphy and computed tomography in assessing biochemical failure after radical prostatectomy. Urology 61(3):607–611
Johnstone PA, Tarman GJ, Riffenburgh R, Rohde DC, Puckett ML, Kane CJ (1997) Yield of imaging and scintigraphy assessing biochemical failure in prostate cancer patients. Urol Oncol 3(4):108–112
Jadvar H, Ballas LK, Choyke PL, Fanti S, Gulley JL, Herrmann K et al (2020) Appropriate use criteria for imaging evaluation of biochemical recurrence of prostate cancer after definitive primary treatment. J Nucl Med 61(4):552–562
Wallitt KL, Khan SR, Dubash S, Tam HH, Khan S, Barwick TD (2017) Clinical PET imaging in prostate cancer. Radiographics 37(5):1512–1536
Schwarzenboeck SM, Rauscher I, Bluemel C, Fendler WP, Rowe SP, Pomper MG et al (2017) PSMA ligands for PET imaging of prostate cancer. J Nucl Med 58(10):1545–1552
Tosoian JJ, Gorin MA, Ross AE, Pienta KJ, Tran PT, Schaeffer EM (2017) Oligometastatic prostate cancer: definitions, clinical outcomes, and treatment considerations. Nat Rev Urol 14(1):15–25
Amorim BJ, Prabhu V, Marco SS, Gervais D, Palmer WE, Heidari P et al (2020) Performance of (18)F-fluciclovine PET/MR in the evaluation of osseous metastases from castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging 47(1):105–114
Battaglia A, De Meerleer G, Tosco L, Moris L, Van den Broeck T, Devos G et al (2019) Novel insights into the management of oligometastatic prostate cancer: a comprehensive review. Eur Urol Oncol 2(2):174–188
Ahmed KA, Barney BM, Davis BJ, Park SS, Kwon ED, Olivier KR (2012) Stereotactic body radiation therapy in the treatment of oligometastatic prostate cancer. Front Oncol 2:215
Crawford ED, Koo PJ, Shore N, Slovin SF, Concepcion RS, Freedland SJ et al (2019) A clinician’s guide to next generation imaging in patients with advanced prostate cancer (RADAR III). J Urol 201(4):682–692
Hellman S, Weichselbaum RR (1995) Oligometastases. J Clin Oncol 13:8–10
Ost P, Bossi A, Decaestecker K, De Meerleer G, Giannarini G, Karnes RJ et al (2015) Metastasis-directed therapy of regional and distant recurrences after curative treatment of prostate cancer: a systematic review of the literature. Eur Urol 67(5):852–863
Tabata K-I, Niibe Y, Satoh T, Tsumura H, Ikeda M, Minamida S et al (2012) Radiotherapy for oligometastases and oligo-recurrence of bone in prostate cancer. Pulm Med 2012:541656
Schick U, Jorcano S, Nouet P, Rouzaud M, Vees H, Zilli T et al (2013) Androgen deprivation and high-dose radiotherapy for oligometastatic prostate cancer patients with less than five regional and/or distant metastases. Acta Oncol 52(8):1622–1628
Decaestecker K, De Meerleer G, Lambert B, Delrue L, Fonteyne V, Claeys T et al (2014) Repeated stereotactic body radiotherapy for oligometastatic prostate cancer recurrence. Radiat Oncol 9:135
Odewole OA, Tade FI, Nieh PT, Savir-Baruch B, Jani AB, Master VA et al (2016) Recurrent prostate cancer detection with anti-3-[(18)F]FACBC PET/CT: comparison with CT. Eur J Nucl Med Mol Imaging 43(10):1773–1783
Savir-Baruch B, Lovrec P, Solanki AA, Adams WH, Yonover PM, Gupta G et al (2019) Fluorine-18-labeled fluciclovine PET/CT in clinical practice: factors affecting the rate of detection of recurrent prostate cancer. AJR Am J Roentgenol 213(4):851–858
El BJ, Grybowski D, Lovrec P, Solanki AA, Gabriel MS, Wagner RH et al (2022) Positivity rate of [(18)F]fluciclovine PET/CT in patients with suspected prostate cancer recurrence at PSA levels below 1 ng/mL. Mol imaging Biol 24(1):42–49
Salavati A, Gencturk M, Koksel Y, Schik AN, Carroll PR, Feng FY et al (2021) A bicentric retrospective analysis of clinical utility of (18)F-fluciclovine PET in biochemically recurrent prostate cancer following primary radiation therapy: is it helpful in patients with a PSA rise less than the Phoenix criteria? Eur J Nucl Med Mol Imaging 48(13):4463–4471
England JR, Paluch J, Ballas LK, Jadvar H (2019) 18F-fluciclovine PET/CT detection of recurrent prostate carcinoma in patients with serum PSA ≤ 1 ng/mL after definitive primary treatment. Clin Nucl Med 44(3):e128–e132
Calais J, Ceci F, Eiber M, Hope TA, Hofman MS, Rischpler C et al (2019) (18)F-fluciclovine PET-CT and (68)Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: a prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol 20(9):1286–1294
Abiodun-Ojo OA, Jani AB, Akintayo AA, Akin-Akintayo OO, Odewole OA, Tade FI et al (2021) Salvage radiotherapy management decisions in postprostatectomy patients with recurrent prostate cancer based on (18)F-fluciclovine PET/CT guidance. J Nucl Med 62(8):1089–1096
Michael J, Khandani AH, Basak R, Tan H-J, Royce TJ, Wallen E et al (2021) Patterns of recurrence, detection rates, and impact of 18-F fluciclovine PET/CT on the management of men with recurrent prostate cancer. Urology 155:192–198
Savir-Baruch B, Choyke PL, Rowe SP, Schuster DM, Subramaniam RM, Jadvar H (2021) Role of (18)F-fluciclovine and prostate-specific membrane antigen PET/CT in guiding management of oligometastatic prostate cancer: AJR expert panel narrative review. AJR Am J Roentgenol 216(4):851–859
Jadvar H (2018) Oligometastatic prostate cancer: molecular imaging and clinical management implications in the era of precision oncology. J Nucl Med 59(9):1338–1339
Andriole GL, Kostakoglu L, Chau A, Duan F, Mahmood U, Mankoff DA et al (2019) The impact of positron emission tomography with 18F-fluciclovine on the treatment of biochemical recurrence of prostate cancer: results from the LOCATE trial. J Urol 201(2):322–330
Vapiwala N, Chen Y-H, Cho SY, Duan F, Kyriakopoulos C, Shevrin DH et al (2021) PET-directed local or systemic therapy intensification in prostate cancer patients with post-prostatectomy biochemical recurrence: a trial of the ECOG-ACRIN Cancer Research Group (EA8191). J Clin Oncol 39(6suppl):TPS267–TPS267
Galgano SJ, McDonald AM, Rais-Bahrami S, Porter KK, Choudhary G, Burgan C et al (2021) Utility of (18)F-fluciclovine PET/MRI for staging newly diagnosed high-risk prostate cancer and evaluating response to initial androgen deprivation therapy: a prospective single-arm pilot study. AJR Am J Roentgenol 217(3):720–729
Scarsbrook AF, Bottomley D, Teoh EJ, Bradley KM, Payne H, Afaq A et al (2020) Effect of (18)F-fluciclovine positron emission tomography on the management of patients with recurrence of prostate cancer: results from the FALCON trial. Int J Radiat Oncol Biol Phys 107(2):316–324
Funding
This work was supported in part by grants R21-EB017568 and P30‐CA014089 from the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Hossein Jadvar declare that he is on the advisory board of Radiomedix, a consultant to Bayer and Blue Earth Diagnostics, and on the speaker’s bureau for Lantheus, all unrelated to this submission. Dr. Redmond-Craig Anderson and Dr. Erik M. Velez declare no conflict of interests or competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Anderson, RC., Velez, E.M. & Jadvar, H. Management Impact of Metachronous Oligometastatic Disease Identified on 18F-Fluciclovine (Axumin™) PET/CT in Biochemically Recurrent Prostate Cancer. Mol Imaging Biol 24, 920–927 (2022). https://doi.org/10.1007/s11307-022-01742-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-022-01742-0