Abstract
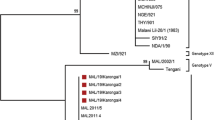
This study investigates suspected African swine fever (ASF) outbreaks in two villages of Kannur district in Kerala, India, with the aim of identifying the causative agent and its genotype, the source of infection, and estimating the economic losses due to the outbreaks. Clinically, the disease was acute with high mortality, while gross pathology was characterized by widespread haemorrhages in various organs, especially the spleen, which was dark, enlarged and had friable cut surfaces with diffuse haemorrhages. Notably, histopathological examination revealed multifocal, diffuse haemorrhages in the splenic parenchyma and lymphoid depletion accompanied by lymphoid cell necrosis. The clinico-pathological observations were suggestive of ASF, which was confirmed by PCR. The source of outbreak was identified as swill and it was a likely point source infection as revealed by epidemic curve analysis. The phylogenetic analysis of p72 gene identified the ASFV in the current outbreak as genotype-II and IGR II variant consistent with ASFVs detected in India thus far. However, the sequence analysis of the Central Variable Region (CVR) of the B602L gene showed that the ASFVs circulating in Kerala (South India) formed a separate clade along with those found in Mizoram (North East India), while ASFVs circulating in Arunachal Pradesh and Assam states of India grouped in to different clade. This study represents the first investigation of ASF outbreak in South India, establishing the genetic relatedness of the ASFV circulating in this region with that in other parts of the country. The study also underscores the utility of the CVR of the B602L gene in genetically characterizing highly similar Genotype II ASFVs to understand the spread of ASF within the country.






Similar content being viewed by others
Data availability
The sequences are deposited in the NCBI Nucleotide database under accession numbers OR502005-OR502009 (Intergenic region between I73R and I329L), OR490939-OR490943 (B602L) and OR472614-OR472617 (B646L). Any other data if required will be made available on request.
References
Agüero M, Fernández J, Romero L et al (2003) Highly sensitive PCR assay for routine diagnosis of African swine fever virus in clinical samples. J Clin Microbiol 41:4431–4434. https://doi.org/10.1128/JCM.41.9.4431-4434.2003
Alonso C, Borca M, Dixon L et al (2018) ICTV virus taxonomy profile: asfarviridae. J Gen Virol 99:613–614. https://doi.org/10.1099/jgv.0.001049
Amar S, De Boni L, de Voux A et al (2021) An outbreak of African swine fever in small-scale pigs, Gauteng, South Africa, July 2020. Int J Infect Dis 110:S44–S49. https://doi.org/10.1016/j.ijid.2021.04.003
Bastos ADS, Penrith ML, Crucière C et al (2003) Genotyping field strains of African swine fever virus by partial p72 gene characterisation. Arch Virol 148:693–706. https://doi.org/10.1007/s00705-002-0946-8
Buragohain L, Barman NN, Sen S et al (2023) Transmission of African swine fever virus to the wild boars of Northeast India. Vet Q 43:1–10. https://doi.org/10.1080/01652176.2023.2178689
Carrasco L, Chacón-M De Lara F, De Las M, Mulas J et al (1997) Ultrastructural changes related to the lymph node haemorrhages in acute African swine fever. Res Vet Sci 62:199–204. https://doi.org/10.1016/S0034-5288(97)90190-9
CDC (2022) Using an Epicurve to determine mode of spread. In: CD2. https://www.cdc.gov/training/quicklearns/epimode/. Accessed 1 June 2023
Cheng J, Ward MP (2022) Risk factors for the spread of African Swine Fever in China: a systematic review of Chinese-language literature. Transbound Emerg Dis 69:e1289–e1298
Costard S, Zagmutt FJ, Porphyre T, Pfeiffer DU (2015) Small-scale pig farmers’ behavior, silent release of African swine fever virus and consequences for disease spread. Sci Rep 5. https://doi.org/10.1038/srep17074
Cwynar P, Stojkov J, Wlazlak K (2019) African swine fever status in europe. Viruses 11(4):310. https://doi.org/10.3390/v11040310
Dame-Korevaar A, Boumans IJMM, Antonis AFG et al (2021) Microbial health hazards of recycling food waste as animal feed. Futur Foods 4:100062. https://doi.org/10.1016/j.fufo.2021.100062
Deka RP (2015) Pig system in Northeast India: an untapped opportunity for entrepreneurs. Presented at a seminar on enhancing the export of meat and meat products from Northeast India, Guwahati, India. Nairobi, Kenya
Dijkhuizen AA, Huirne RBM, Jalvingh AW (1995) Economic analysis of animal diseases and their control. Prev Vet Med 25:135–149. https://doi.org/10.1016/0167-5877(95)00535-8
Dixon LK, Chapman DAG, Netherton CL, Upton C (2013) African swine fever virus replication and genomics. Virus Res 173:3–14
Dixon LK, Stahl K, Jori F et al (2020) African swine fever epidemiology and control. Annu Rev Anim Biosci 8:221–246
FAO-EMPRES-AH (2023) FAO-Emergency Prevention System for Animal Health (EMPRES-AH). African swine fever (ASF) situation updates in Asia & Pacific. In: FAO. https://www.fao.org/animal-health/situation-updates/asf-in-asia-pacific/en. Accessed 6 July 2023
Gallardo C, Fernández-Pinero J, Pelayo V et al (2014) Genetic variation among african swine fever genotype II viruses, Eastern and Central Europe. Emerg Infect Dis 20:1544–1547. https://doi.org/10.3201/eid2009.140554
Gallardo C, Casado N, Soler A et al (2023) A multi gene-approach genotyping method identifies 24 genetic clusters within the genotype II-European African swine fever viruses circulating from 2007 to 2022. Front Vet Sci 10. https://doi.org/10.3389/fvets.2023.1112850
Gao Y, Nielsen LH, Boklund AE et al (2023) SWOT analysis of risk factors associated with introduction of African Swine Fever through vehicles returning after export of pigs. Front Vet Sci 9. https://doi.org/10.3389/fvets.2022.1049940
Kannan A, Xavier F, Raja TV, Murugan M (2008) Awareness and Needs of Pig Farmers in Kerala. J Vet Med Anim Sci 39:36–39
Kerala Forest Statstics (2021) Kerala Forest Statistics. Thiruvananthapuram
Konno S, Taylor WD, Hess WR, Heuschele WP (1972) Spleen pathology in African swine fever. Cornell Vet 62:486–506
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Matsumoto N, Siengsanan-Lamont J, Halasa T et al (2021) The impact of African swine fever virus on smallholder village pig production: an outbreak investigation in Lao PDR. Transbound Emerg Dis 68:2897–2908. https://doi.org/10.1111/tbed.14193
Mazloum A, van Schalkwyk A, Chernyshev R et al (2023) A Guide to Molecular Characterization of Genotype II African Swine Fever Virus: Essential and Alternative Genome Markers. Microorganisms 11(3):642. https://doi.org/10.3390/microorganisms11030642
Mohan NH, Misha MM, Gupta VK (2021) Consequences of African swine fever in India: beyond economic implications. Transbound Emerg Dis 68:3009–3011. https://doi.org/10.1111/tbed.14318
Nantima N, Ocaido M, Ouma E et al (2015) Risk factors associated with occurrence of African swine fever outbreaks in smallholder pig farms in four districts along the Uganda-Kenya border. Trop Anim Health Prod 47:589–595. https://doi.org/10.1007/s11250-015-0768-9
Nga BTT, Tran Anh Dao B, Nguyen Thi L et al (2020) Clinical and pathological study of the first outbreak cases of African swine fever in Vietnam, 2019. Front Vet Sci 7. https://doi.org/10.3389/fvets.2020.00392
Nguyen TTH, Nguyen VT, Le PN et al (2023) Pathological characteristics of domestic pigs orally infected with the virus strain causing the first reported african swine fever outbreaks in Vietnam. Pathogens 12. https://doi.org/10.3390/pathogens12030393
Nix RJ, Gallardo C, Hutchings G et al (2006) Molecular epidemiology of African swine fever virus studied by analysis of four variable genome regions. Arch Virol 151:2475–2494. https://doi.org/10.1007/s00705-006-0794-z
Njau EP, Domelevo Entfellner JB, Machuka EM et al (2021) The first genotype II African swine fever virus isolated in Africa provides insight into the current Eurasian pandemic. Sci Rep 11. https://doi.org/10.1038/s41598-021-92593-2
Patil SS, Hemadri D, Shankar BP et al (2010) Genetic typing of recent classical swine fever isolates from India. Vet Microbiol 141:367–373. https://doi.org/10.1016/j.vetmic.2009.09.021
Penrith ML, Bastos AD, Etter EMC, Beltrán-Alcrudo D (2019) Epidemiology of African swine fever in Africa today: sylvatic cycle versus socio-economic imperatives. Transbound Emerg Dis 66:672–686
Penrith ML (2020) Current status of African swine fever. CABI Agric Biosci 1:11. https://doi.org/10.1186/s43170-020-00011-w
Rajkhowa TK, Jagan Mohanarao G, Gogoi A et al (2015) Porcine reproductive and respiratory syndrome virus (PRRSV) from the first outbreak of India shows close relationship with the highly pathogenic variant of China. Vet Q 35:186–193. https://doi.org/10.1080/01652176.2015.1066043
Rajkhowa TK, Kiran J, Hauhnar L et al (2022) Molecular detection and characterization of African swine fever virus from field outbreaks in domestic pigs, Mizoram, India. Transbound Emerg Dis 69:e1028–e1036. https://doi.org/10.1111/tbed.14384
Rajukumar K, Senthilkumar D, Venkatesh G et al (2021) Genetic characterization of African swine fever virus from domestic pigs in India. Transbound Emerg Dis 68:2687–2692. https://doi.org/10.1111/tbed.13986
Salguero FJ (2020) Comparative Pathology and Pathogenesis of African Swine Fever Infection in Swine. Front Vet Sci 7:282. https://doi.org/10.3389/fvets.2020.00282
Sánchez-Vizcaíno JM, Mur L, Gomez-Villamandos JC, Carrasco L (2015) An update on the epidemiology and pathology of African swine fever. J Comp Pathol 152:9–21
Schulz K, Staubach C, Blome S (2017) African and classical swine fever: Similarities, differences and epidemiological consequences. Vet Res 48(1):84. https://doi.org/10.1186/s13567-017-0490-x
Semerjyan AB, Tatoyan MR, Karalyan NY et al (2018) Cardiopathology in acute African Swine Fever. Ann Parasitol 64:253–258. https://doi.org/10.17420/ap6403.161
Senthilkumar D, Rajukumar K, Venkatesh G et al (2022) Complete genome analysis of African swine fever virus isolated from domestic pigs during the first ASF outbreaks in India. Transbound Emerg Dis 69:e2020–e2027. https://doi.org/10.1111/tbed.14536
Sharma S (2011) Negotiating transnational identities on Indo-Myanmar border. India Q A J Int Aff 67:53–64. https://doi.org/10.1177/097492841006700104
Sidhu HS, Kansal SK, Singh J, Singh P (2019) Exploration of marketing channels followed by pig farmers in Punjab, India. Int J Curr Microbiol Appl Sci 8:1471–1478
Singh B, Bardhan D, Verma MR et al (2016) Incidence of Classical Swine Fever (CSF) in pigs in India and its economic valuation with a simple mathematical model. Anim Sci Rep 10:3–9
Thomas R, Singh V, Gupta VK (2021) Current status and development prospects of India’s pig industry. Indian J Anim Sci 91:255–268
WAHIS (2015) African swine fever. EFSA J 13. https://doi.org/10.2903/j.efsa.2015.4163
WAHIS (2019) African swine fever, Myanmar. [WWW Document]. WAHIS. https://www.oie.int/wahis_2/public/wahid.php/Reviewreport/Review?page_refer=MapEventSummary. Accessed 6.5.23
WAHIS (2021) African swine fever, India [WWW Document]. WAHIS. https://wahis.woah.org/#/in-review/3190?reportId=152126&fromPage=event-dashboard-url. Accessed 6.5.23
World Organisation for Animal Health (OIE), Food and Agriculture Organization of the United Nations (FAO) (2018) A field manual for animal disease outbreak investigation and management. OIE Sub-Regional Represent. South-East Asia Dep. Livest. Dev. 69/1, Phaya Thai Road, Ratchathewi, Bangkok 10400, Thail. 1–89
Yabe J, Hamambulu P, Simulundu E et al (2015) Pathological and molecular diagnosis of the 2013 African swine fever outbreak in Lusaka, Zambia. Trop Anim Health Prod 47:459–463. https://doi.org/10.1007/s11250-014-0732-0
Acknowledgements
The authors would like to thank Dr. G. Govindaraj, Principal Scientist, ICAR-NIVEDI for providing inputs to improve the presentation of the data on economic loss due to ASF outbreak. We are highly thankful to officials of State Animal Husbandry Department and the District Animal Husbandry Office, Kannur, Kerala for their kind cooperation in carrying out the epidemiological investigation of ASF. We thank Indian Council of Agricultural Research, New Delhi for funding the project No. ANSCNIVEDISIL202300800148.
Author information
Authors and Affiliations
Contributions
Conceptualization: Jagadish Hiremath; Methodology: Jagadish Hiremath, Divakar Hemadri, Shivasharanappa Nayakvadi, Chethan Kumar , C. S. Sathish Gowda; Formal Analysis: Jagadish Hiremath, Divakar Hemadri, Shivasharanappa Nayakvadi, Chethan Kumar , C. S. Sathish Gowda; Investigation: Jagadish Hiremath, Shivasharanappa Nayakvadi, Chethan Kumar , C. S. Sathish Gowda, Ranjini A.R, Jayamohanan T. V, Swapna Susan Abraham; Writing- original draft preparation: Jagadish Hiremath; Writing- review and editing: Divakar Hemadri, Shivasharanappa Nayakvadi, Chethan Kumar , C. S. Sathish Gowda, Damini Sharma, Ramamoorthy. R., Mamatha S S, Sharanagouda Patil, Ranjini A.R, Jayamohanan T. V, Swapna Susan Abraham, Baldev Raj Gulati: Funding Acquisition: Baldev Raj Gulati; Resources: Baldev Raj Gulati.
Corresponding author
Ethics declarations
Ethical approval
Not Applicable, as research involved collection of clinical samples from suffering animal.
Consent to participate
Not applicable as the research did not involve humans or client-owned animals.
Consent for publication
Not applicable as the research did not involve humans or client-owned animals.
Competing interest
The authors declare no competing interests.
Conflict of interest
Authors expresses no conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hiremath, J., Hemadri, D., Nayakvadi, S. et al. Epidemiological investigation of ASF outbreaks in Kerala (India): detection, source tracing and economic implications. Vet Res Commun 48, 827–837 (2024). https://doi.org/10.1007/s11259-023-10254-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10254-3