Abstract
Mesopredators including coyotes (Canis latrans) and red foxes (Vulpes vulpes) often co-occur in urban environments, but how niche partitioning facilitates their coexistence remains unclear. Highly urbanized areas can be spatial refuges for smaller mesopredators (i.e., spatial human shield effect), however these species also may coexist through temporal niche partitioning. We used camera traps (n = 110 sites) across an urbanization gradient in Chicago to examine coyote-fox interactions from 2011 to 2018. We analyzed spatial partitioning through multi-season occupancy models and structural equation modeling (SEM), and quantified temporal overlap between canids and with humans. Coyotes most often occurred in natural areas, and urbanization reduced their colonization rates and increased their extinction rates. Initial occupancy for red foxes was negatively impacted by urbanization, but their extinction rates depended on a surprising interaction between coyotes and humans. When coyotes were rare, fox extinction was related positively to human activity; but when coyotes were more common, fox extinction was related negatively to human activity. This outcome may reflect a human shield effect at a within-site scale. The SEM further supported the negative impact of urbanization on both canids, and lack of an effect of coyotes on the distribution of foxes. Diel activity of coyotes and red foxes indicated temporal niche partitioning intensified at more urbanized sites. Our results suggest the spatial human shield effect is not operating across sites in Chicago. Instead, coyotes and red foxes may share green spaces, especially in highly urbanized areas, where species coexistence is promoted by temporal niche partitioning.
Similar content being viewed by others
Introduction
Urbanization acts as a strong filter that can cause local extinctions of wildlife species while also providing opportunities for urban adapters and exploiters (McKinney 2002). Urbanization decreases and fragments habitat, which can restrict wildlife movements (Crooks 2002; Ordeñana et al. 2010; Magle et al. 2012) and increase vehicle collisions (Tigas et al. 2002). However, remaining green spaces can be suitable habitat and serve as movement corridors (Magle et al. 2010; Gallo et al. 2017), and maintaining a variety of green spaces can promote habitat heterogeneity and gamma diversity for urban areas (Gallo et al. 2017; Tryjanowski et al. 2017). Anthropogenic foods may be alternative resources for urban wildlife, especially if natural foods are limited (Newsome and Eeden 2017). Reliance on human foods or use of human structures can lead to human-wildlife conflicts, however, including property destruction, wildlife attacks, and wildlife mortality (Hadidian et al. 2010; Cusa et al. 2015; Poessel et al. 2017).
Urbanization also alters predator distributions and mediates species interactions (Fischer et al. 2012; Wang et al. 2015; Greenspan et al. 2018). Some mammalian mesopredators have increased in urban settings leading to excessive predation on native prey species, increased interference competition, and intraguild predation (Crooks and Soule 1999; Crooks et al. 2010). Fundamentally, intraguild predation consists of a dominant predator excluding and killing a competing, subordinate predator that shares common prey (Polis and Myers 1989; Prugh and Sivy 2020). Theory suggests the two species can coexist when resource levels are intermediate, whereas scarce resources favor the subordinate predator (assuming it is a better exploitative competitor), and abundant resources favor the dominant predator (Holt and Polis 1997; Robinson et al. 2014; Lonsinger et al. 2017). However, interactions between subordinate and dominant predators may be altered in urban areas (Sévêque et al. 2020). For instance, bobcats (Lynx rufus) avoided areas recently used by pumas (Puma concolor) in less urban environments but did not show the same avoidance in more developed areas, even though pumas kill bobcats (Lewis et al. 2015). Higher levels of urbanization may also offer relief to subordinate predators due to the “spatial human shield effect”. This hypothesis suggests that subordinate predators will use areas with increased human activity due to dominant predators avoiding these spaces (Moll et al. 2018; Parsons et al. 2019). For instance, in east-central Illinois, red foxes (Vulpes vulpes) were killed by coyotes (Canis latrans) in rural areas, but predation was much lower in urban areas where they were able to avoid coyotes (Gosselink et al. 2007).
Land conversion and the extirpation of wolves (Canis lupus) have allowed coyotes to expand their geographic range across eastern North America (Hody and Kays 2018). In Illinois, an increase in coyote populations started in rural areas around the 1970s and expanded into major cities, like Chicago, in the 1990s (Gosselink et al. 2007; Gehrt et al. 2009). Coyotes are a generalist species that have adjusted well to urban areas and become the top predator in many of these systems (Gompper 2002; Gehrt and Riley 2010; Greenspan et al. 2018). Although vehicle collisions can contribute to >50% of coyote mortalities in cities (Gehrt and Riley 2010), shifting activity to nocturnal hours by coyotes can decrease risk (Murray and St. Clair 2015). Even though coyotes have successfully colonized urban environments, they still usually prefer natural habitats within them (Crooks 2002; Gehrt and Riley 2010; Gese et al. 2012). For example, coyotes in Chicago often select areas with forest preserves and golf courses away from higher levels of human activity and development (Gehrt et al. 2009; Gallo et al. 2017; Wurth et al. 2020).
Harvest reports in Illinois, which mainly track trends in rural areas, indicate a strong decline of red foxes as coyote populations have increased over time (Gosselink et al. 2007; Bauder et al. 2022). Coyotes and red foxes are intraguild competitors that share similar prey resources, such as small mammals and lagomorphs (Gese et al. 1996; Gosselink et al. 2007; Peterson et al. 2021). However, red foxes may find refuge from coyotes in moderate levels of urbanization (Gosselink et al. 2003, 2007). Red foxes have acclimated to urbanization, inhabiting many cities in North America and Europe (Soulsbury et al. 2010; Mueller et al. 2018; Scholz et al. 2020). Furthermore, red foxes are positively associated with urbanization in cities with sufficient green spaces (Fidino et al. 2021). As urbanization increases, however, suitable habitat may become limited and restrict competing species to using the same green spaces (LeFlore et al. 2019; Parsons et al. 2019). To decrease potential negative interactions, red foxes might shift their diel activity to reduce overlap with coyotes (LeFlore et al. 2019; Malhotra et al. 2022; Parsons et al. 2022). Such temporal niche partitioning may facilitate species coexistence in areas where habitat is limited.
We explored coyote and red fox interactions across an urbanization gradient in Chicago, Illinois. We asked whether spatial partitioning and the human shield effect was operating and determined if temporal niche partitioning reduced the risk of intraguild predation, especially in highly urban areas with limited green spaces. Due to their preference for including natural habitat within their territory and a desire to avoid humans (Riley et al. 2003; Gallo et al. 2017; Mueller et al. 2018), we predicted that coyotes would mainly inhabit natural areas and lower levels of urbanization. We expected coyotes to restrict red foxes spatially and therefore predicted foxes would use moderate levels of urbanization to avoid coyotes (Lesmeister et al. 2015). We also expected that both species would avoid high levels of urbanization, when possible, reflecting a limit to their tolerance of humans (Gehrt and Riley 2010; Soulsbury et al. 2010). If coyotes and red foxes were forced to use the same areas, we predicted foxes would shift their temporal activity to avoid coyotes (LeFlore et al. 2019; Parsons et al. 2022). Furthermore, we expected both species to have low temporal overlap in activity with humans (Riley et al. 2003; Moll et al. 2018).
Methods
Study area
We conducted our study across the Chicago metropolitan area in Illinois, USA. Chicago is the third largest city in the United States with a population of 2.7 million people and 9.5 million people throughout the entire metropolitan area (U.S. Census Bureau 2020). Although highly urbanized, Chicago also includes fragmented natural habitat in part due to >81,000 ha of land protected under the Chicago Wilderness coalition (Wang and Moskovits 2001). The southwest portion of Lake Michigan is to the east of the city, whereas natural areas encompass parts of the west, north, and south sides of the city and metropolitan area. Natural areas contain forests, oak (Quercus spp.) savannahs, tallgrass prairies, wetlands, and the lakeshore (Wang and Moskovits 2001). However, toward the fringes of the metropolitan area the landscape becomes highly modified with row-crop agriculture (Lehrer et al. 2021) with limited natural habitat despite being the rural component of the urbanization gradient.
Study design and sampling
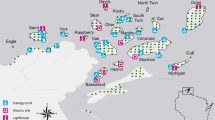
We collected data using cameras traps (Bushnell, Trophy Cam, Overland Park, Kansas) along three 50-km transects (Fig. 1; Magle et al. 2014; Fidino et al. 2016). Transects started from the city center and extended to the northwest, west, and southwest. We subdivided each transect into 10 5-km stretches and randomly placed camera traps within 2 km of each transect within a green space (Gallo et al. 2017). No more than four green spaces were chosen within each 5-km stretch (Fidino et al. 2016; Gallo et al. 2017). All camera traps were located ≥1 km from other camera traps. We strapped cameras to trees ~1.5-2.0 m above the ground and placed a synthetic fatty acid tablet (USDA, Pocatello Supply Depot, Idaho) as lure within 3-5 m, in line with the camera. We sampled a total of 110 sites throughout our study.
The green spaces that we sampled included four categories: city parks (n = 43), cemeteries (n = 12), golf courses (n = 10), and natural areas (n = 45; Magle et al. 2014). Smaller city parks (≤2 ha) included native and non-native trees spread among turfgrass (Fidino et al. 2016; Gallo et al. 2017), whereas larger parks (>2 ha) had native plants dispersed throughout (Fidino et al. 2016). Cemeteries were variable with larger cemeteries (>2 ha) resembling small city parks, and smaller cemeteries containing or bordering forested areas (Fidino et al. 2016). Golf courses were large (mean = 83 ha) and heavily landscaped but included natural features and human-made water structures (Fidino et al. 2016; Gallo et al. 2017). Natural areas mainly consisted of protected forest preserves plus other native vegetation (e.g., grasslands, prairies) but with varying levels of non-native species (Fidino et al. 2016; Gallo et al. 2017; Greenspan et al. 2018).
We used camera trap data from the fall season (Oct-Nov) of each year between 2011 and 2018. Our sampling occurred over four weeks usually starting the first week of October. We considered each survey to last one week, which created four occupancy surveys per sampling season.
Model covariates and urbanization gradient
We estimated detection, initial occupancy, colonization, and extinction separately for coyotes and red foxes using single-species, multi-season occupancy models (MacKenzie et al. 2003). We did not use a multispecies model for interacting species because our data for red foxes were too sparse to estimate the additional parameters. For both species, our only detection covariate was sampling effort (number of working camera days/seven), which was a survey-specific covariate. Our occupancy, colonization, and extinction covariates included coyote photographic rate (for fox models), human photographic rate, habitat type, and urbanization. Coyote and human photographic rates (number of independent photographs/number of days sampled each year) were treated as site-specific covariates that could vary among years. We considered sequential photos of species at a site independent if separated by ≥30 minutes (Farris et al. 2015, Moll et al. 2018). Human photographic rate was a measure of human activity at the local scale and complemented our urbanization metric measured at a broader spatial scale (see below). We acknowledge that human photographic rate only captures activity near the camera location. Thus, this measure could be conservative for sites with high human activity if cameras were placed to avoid areas with the most activity to minimize camera disturbance. Our habitat covariate included the four types of green spaces where camera traps were located (city parks, cemeteries, golf courses, natural areas).
To quantify our urbanization gradient, we used principal component analysis (PCA) to combine housing density, impervious land cover, and tree canopy cover into one urbanization metric (Gallo et al. 2017). We measured these three variables within 1-km buffers centered on the camera trap locations. We used the Illinois block housing density from the U.S. Census Bureau (2010) to measure housing density using ArcGIS 10.7.1 (ESRI 2019). We then extracted impervious cover and tree canopy cover using the Chicago Metropolitan Agency for Planning 10-County Region Land Cover (2016) in R ver. 4.0.3 (R Core Team 2020). We extracted data using the ‘ClassStat’ function in the ‘SDMTools’ package (v. 1.1-221.2; VanDerWal et al. 2019). We used the first principal component from the PCA for urbanization, which accounted for 71% of the variation. The factor loadings for the first principal component (housing density = 0.56, impervious cover = 0.62, tree canopy cover = -0.54) indicated that a positive PCA score for urbanization characterized a site with high housing density and impervious land cover but low tree canopy cover.
Occupancy models
We created single-species, multi-season occupancy models for coyotes and red foxes using the ‘unmarked’ package (v. 1.1.0; Fiske and Chandler 2011). For coyotes, we considered three covariates that could affect initial occupancy, colonization, and extinction: human photographic rate, habitat type, and urbanization. For red foxes, we evaluated the same three covariates plus coyote photographic rate, which indexed pressure from the intraguild predator. Because photographic rates were treated as yearly site covariates, we could not run models with missing values. Thus, if a site was not sampled within a year, we interpolated the mean value for that site across other years for coyote and human photographic rates. We then centered and scaled each continuous covariate to have a mean of 0 and SD of 1 in R ver. 4.0.3.
We examined eight potential models (single covariate, additive, null) for initial occupancy, colonization, and extinction for coyotes (Tables S1-S3), and eighteen potential models for red foxes (single covariate, additive, interactive, null; Tables S4-S6). The interaction models for red foxes included an interaction between coyote photographic rate and human photographic rate, and a model with an interaction between coyote photographic rate and urbanization. These interaction models reflected expectations that red foxes might tolerate different levels of coyotes at a site depending on its level of local human activity or location along the urbanization gradient. Specifically, foxes might be able to find safe patches within sites avoided by coyotes due their own avoidance of humans (i.e., spatial human shield effect at a within-site scale).
To identify important covariates for initial occupancy and turnover, we used a sequential selection process (Cosentino et al. 2010; Ahlers et al. 2016). Sampling effort was included as a detection covariate throughout the sequential process. First, we evaluated initial occupancy while holding colonization and extinction constant using intercept-only models. Then, we carried the supported occupancy covariate(s) over as we analyzed colonization, while holding extinction constant. Finally, we analyzed extinction while including the supported covariates for occupancy and colonization. At each step, we ranked our models using AICc (Akaike’s Information Criterion adjusted for small sample size) and considered models with ΔAICc ≤ 2 as competitive (Burnham and Anderson 2002).
Structural equation model
We used structural equation modeling (SEM; Grace 2006) to further explore relationships among variables and to test the human shield effect hypothesis directly. We chose SEM because of its ability to measure direct and indirect effects through model pathways (Grace et al. 2010; Sivy et al. 2017). We used a subset of our data (2011-2014) because naïve occupancy for red foxes was greater for those years and declined to low levels in subsequent years. Thus, we tested whether humans mediated negative effects of coyotes when foxes were not uncommon overall. For each year, we estimated occupancy probability for coyotes and red foxes using a multi-season occupancy model and the recursive equation (MacKenzie et al. 2003). We included sampling effort as our detection covariate but did not include any occupancy covariates because the variables of interest were included in the SEM. We then averaged our occupancy probabilities across the four years.
We used human photographic rate (averaged for 2011-2014) and our urbanization metric as described above. We included these variables as direct pathways to both coyote and red fox occupancy (Fig. 2). We also included a direct pathway from coyote occupancy to red fox occupancy, which created indirect pathways from human photographic rate and urbanization to red fox occupancy (Fig. 2). These indirect pathways allowed us to assess any mediation coyote occupancy may have on red fox occupancy in relation to human activity and urbanization.
Structural equation model for coyotes and red foxes. We included indirect pathways from human photographic rate and urbanization, through coyote occupancy, to red fox occupancy to test the human shield effect hypothesis. Bold, solid black lines indicate p ≤ 0.05, solid black lines indicate 0.05 < p < 0.10, and dashed lines indicate p > 0.10. Unstandardized path coefficients are within the parentheses next to the standardized coefficients
We used a global estimation approach for our model that reflected the human shield effect hypothesis (Fig. 2). This estimation created a variance-covariance matrix from our pathways and used maximum likelihood to estimate parameterization values (Grace et al. 2015; Wagnon et al. 2020). We report both standardized and unstandardized coefficients and used standardized estimates to evaluate the strength of our direct and indirect pathways (Grace and Bollen 2005; Wagnon et al. 2020). We considered pathways to have strong support if p ≤ 0.05, and moderate support if 0.05 > p ≤ 0.10. We used the ‘lavaan’ package (Rosseel 2012) in R for our SEM.
Because our final model was a saturated model, our df = 0 and model fit could not be assessed directly. However, prior to choosing our final model, we analyzed the same variables but created different direct and indirect pathways that resulted in unsaturated models (Fig. S1). We compared the overall model fit values between our saturated model and unsaturated models, which were considered good fits (Chi-squared test, p > 0.05; Grace et al. 2015), by using the ‘anova’ function in ‘lavaan’ and found no strong differences. We chose to move forward with the saturated model as it matched our human shield effect hypothesis.
Diel activity and temporal niche partitioning
We assessed temporal overlap in diel activity between coyotes and red foxes, coyotes and humans, and red foxes and humans using time stamps from independent photos for fall between 2011 and 2018. We used the ‘overlap’ package (v.0.3.3; Meredith and Ridout 2014) in R to fit kernel density functions and estimate the coefficient of overlap (Δ, Rideout and Linkie 2009; Moll et al. 2018). The coefficient of overlap is on a scale of 0-1. No temporal overlap between species produces a coefficient of 0, and complete overlap produces a coefficient of 1 (Rideout and Linkie 2009). We used the appropriate nonparametric estimator based on sample size (Schmid and Schmidt 2006): \(\widehat{\Delta }\)1 for smaller samples, and \(\widehat{\Delta }\)4 for larger samples (Rideout and Linkie 2009).
We next tested whether temporal overlap between species differed between two levels of urbanization. We used our urbanization metric and defined sites above the median as “high urbanization” and sites below the median as “low urbanization”. We split independent photos into these two categories and then used time stamps to estimate temporal overlap for each species pairing based on urbanization level. To complement the coefficient of overlap, we tested for differences in diel activity patterns between species using the Mardia-Watson-Wheeler test (Batschelet 1981; Kovach 2009). As before, we compared activity between species across all levels of urbanization and for high versus low levels of urbanization.
Results
During fall between 2011 and 2018, we documented 1,295 independent photos for coyotes, 119 for red foxes, and 11,092 for humans. Coyotes were recorded at 90 of the 110 (82%) sites overall and occupied 17 sites (15%) once, 17 sites (15%) twice, 17 sites (15%) thrice, and 39 sites (35%) for four or more years. Red foxes were recorded at 27 of the 110 (25%) sites overall and occupied 11 sites (10%) once, 4 sites (4%) twice, 3 sites (3%) thrice, and 9 (8%) sites for four or more years. Accounting for sampling effort, average occupancy per year was 0.54 for coyotes (range = 0.44 – 0.61) and 0.12 for red foxes (range = 0.07 – 0.22) with occupancy for foxes declining during the study (Fig. S2).
For coyotes, when holding other parameters constant, weekly detection probability was 0.44 (95% CI = 0.40 - 0.47), colonization probability was 0.26 (95% CI = 0.20 - 0.32), and extinction probability was 0.25 (95% CI = 0.19 - 0.32). For red foxes, weekly detection probability was 0.25 (95% CI = 0.18 - 0.33), colonization probability was 0.03 (95% CI = 0.01 - 0.06), and extinction probability was 0.36 (95% CI = 0.20 - 0.56).
Occupancy models
For coyotes, habitat type was the most supported covariate for explaining initial occupancy (Tables 1, S1). Natural areas were our reference habitat and had the highest probability of occupancy, followed by golf courses (β = -0.28, SE = 1.30), cemeteries (β = -1.34, SE = 1.05), and city parks (β = -2.91, SE = 0.85; Fig. 3). The second-ranked model for initial occupancy that included human photographic rate did not explain substantially more variation, and models with only human photographic rate or urbanization were not competitive (Table S1). The only competitive model for coyote colonization included the additive effects of habitat type and urbanization (β = -0.55, SE = 0.26; Tables 1, S2). When compared to natural areas, coyotes were more likely to colonize golf courses (β = 1.01, SE = 0.86) but not cemeteries (β = -0.94, SE = 0.56) and city parks (β = -1.21, SE = 0.48). Coyotes were more likely to colonize less urbanized areas (Fig. 3). Urbanization (β = 0.70, SE = 0.22) was also in the only competitive model for extinction probability (Tables 1, S3). Extinction of coyotes was related positively to urbanization (Fig. 3). Sampling effort was held as a detection covariate throughout our modeling, but it was not strongly associated with coyote detection probability (β = 0.09, SE = 0.78).
(A) Initial occupancy probability (±SE) for coyotes in each habitat type in 2011, (B) effects of urbanization and habitat type on coyote colonization from 2011-2018 (for 95% CIs for habitat types see Fig. S4), and (C) effects of urbanization on coyote extinction probability across the Chicago metropolitan area, 2011-2018. Shaded area is the 95% CI
For red foxes, two models were competitive for initial occupancy (Tables 1, S4). The top-ranked model included human photographic rate (β = 0.55, SE = 0.34) and urbanization (β = -1.00, SE = 0.44), and the second-ranked model included only urbanization (β = -0.657, SE = 0.35). We selected the urbanization model for inference because human photographic rate did not substantially improve model fit (log likelihood) for the top model or when occurring as a single covariate (Tables 1, S4). Red foxes were more likely to occupy less urbanized sites (Fig. 4). The top-ranked model for colonization was our null model (Tables 1, S5) indicating covariates were not strongly supported. Extinction probability for red foxes was best explained by an interaction (β = -24.25, SE = 14.43) between coyote photographic rate (β = -12.04, SE = 7.33) and human photographic rate (β = -11.79, SE = 7.63). No other models were competitive (Tables 1, S6). At sites where coyotes were uncommon, extinction rate for foxes was positively related to human activity (Fig. 4). In contrast, at sites where coyotes were more common, extinction rate for foxes was negatively related to human activity (Fig. 4). Again, sampling effort was kept as a detection covariate throughout our modeling but had a weak association (β = 1.09, SE = 1.89).
(A) Effects of urbanization on initial occupancy probability of red foxes across the Chicago metropolitan area in 2011 (shaded area is the 95% CI), and (B) extinction rate for red foxes from 2011-2018 depended on an interaction between coyote and human photographic rates. Effects of human activity on local extinctions for foxes is shown for sites with low (25th percentile), medium (50th percentile), and high (75th percentile) coyote photographic rates
Structural equation model
Urbanization had a direct negative effect on mean site occupancy for coyotes and red foxes (Fig. 2). Support for this relationship was strong for coyotes (p = 0.003) and moderate for red foxes (p = 0.072). Human photographic rate had a direct negative effect for coyote occupancy (p = 0.021) but not for red fox occupancy (p = 0.189). We did not detect an effect of coyote occupancy on red fox occupancy (p = 0.354), however, so the indirect pathway from urbanization to foxes representing the human shield effect was not supported (Fig. 2).
Diel activity and temporal niche partitioning
Overall, coyotes and red foxes had fairly high temporal overlap (\(\widehat{\Delta }\) = 0.88) but still differed in their diel activity patterns (p = 0.001, W = 13.10; Fig. 5). In high levels of urbanization, temporal overlap decreased (\(\widehat{\Delta }\) = 0.79, Fig. 6) and diel activity differed between the two species (p = 0.007, W = 9.82). In low levels of urbanization, temporal overlap increased (\(\widehat{\Delta }\) = 0.90, Fig. 6) and support for differences in diel activity was weaker (p = 0.096, W = 4.82). These results indicate urbanization drives greater divergence in temporal niches of coyotes and red foxes.
Overlap in temporal activity between (A) coyotes and red foxes, (B) coyotes and humans, and (C) red foxes and humans across the Chicago metropolitan area, 2011-2018. Overlap is shown in gray. \(\widehat{\Delta }\) is the coefficient of overlap between the two species. Dashed black lines represent the averaged sunrise and sunset times across sampling periods
Coyotes and human had low temporal overlap (\(\widehat{\Delta }\) = 0.37, Fig. 5) and differed in diel activity (p < 0.001, W = 1738.89). Coyotes and humans retained low overlap both in high levels of urbanization (\(\widehat{\Delta }\) = 0.37; p < 0.001, W = 1370.12) and in low levels of urbanization (\(\widehat{\Delta }\) = 0.26; p < 0.001, W = 479.95; Fig. S3). Red foxes and humans also had low overlap (\(\widehat{\Delta }\) = 0.29; Fig. 5) and differed in diel activity (p < 0.001, W = 188.37). Foxes and humans had similar low overlap in high levels of urbanization (\(\widehat{\Delta }\) = 0.23; p < 0.001, W = 136.03) and in low levels of urbanization (\(\widehat{\Delta }\) = 0.24; p < 0.001, W = 74.05; Fig. S3).
Discussion
Our study suggests strong spatial partitioning does not occur between coyotes and red foxes in the Chicago metropolitan area, but temporal niche partitioning may promote their coexistence. Urbanization negatively impacted both coyotes and red foxes spatially, and human activity likely affected diel activity patterns for both species. We did not find evidence for the spatial human shield effect across our broad urbanization gradient. Instead, both species may be forced to use the same green spaces, especially in highly urban areas, but with red foxes displaying more nocturnal activity compared to coyotes. Our work extended investigations of coyotes and red foxes to a larger urban ecosystem compared to past research (e.g., Gosselink et al. 2003; Moll et al. 2018; Mueller et al. 2018; Parsons et al. 2019). We also examined these predator interactions over a longer period that enabled us to assess covariates associated with site turnover and document the decline of red foxes.
The occupancy modeling supported our expectations that initial site occupancy by coyotes would be higher at more natural habitats and be negatively related to urbanization. Coyotes were most likely to occupy natural areas and golf courses, and least likely to occupy city parks. These results are consistent with previous research on habitat use by coyotes in Chicago. Radio-tracked coyotes used natural areas and undeveloped lands while avoiding urbanization (Gehrt et al. 2009), and research from our study area spanning fewer years indicates high use of natural areas and golf courses by coyotes (Gallo et al. 2017). Natural areas are important for urban coyotes because they can provide connected habitat, prey, and concealment from humans (Gese et al. 2012; Greenspan et al. 2018). Golf courses can benefit coyotes due to their large extent and the forested areas and water features usually found within courses (Fidino et al. 2016; Gallo et al. 2017). Coyotes can exploit golf courses during the night because humans are usually only active during the day within these spaces (Gallo et al. 2017; Wurth et al. 2020).
Coyotes also were most likely to colonize golf courses and natural areas, as well as sites in less urban areas. Likewise, Gallo et al. (2017) observed high colonization for natural areas, with coyotes also persisting across years at high rates on golf courses. Urbanization also played a role in the distribution of coyotes by increasing the likelihood of local extinctions at sites. Coyotes can tolerate urbanized environments, but they still prefer patches of natural habitat within their home ranges (Gehrt and Riley 2010; Ordeñana et al. 2010; Mueller et al. 2018). Crooks (2002) observed a decrease in coyote occurrence when habitat patches become too isolated and fragmented in urban areas, and Moll et al. (2018) also documented a decrease in coyote occupancy with increased development.
The occupancy models for red foxes provided partial support for our predictions. Urbanization had a negative effect on initial occupancy, and we expected that foxes would only tolerate moderate urbanization. In places without coyotes as the top predator, such as England, red foxes prefer moderate levels of urbanization and avoid highly urbanized areas (Soulsbury et al. 2010). None of our covariates strongly explained colonization of sites by red foxes. In general, we rarely documented red foxes and their population appeared to be declining during our study (Fig. S2), which probably contributed to model uncertainty.
We predicted coyotes would have a negative effect on red foxes due to the threat of intraguild predation. However, we found no evidence that photographic rate of coyotes was a predictor for initial occupancy or colonization of sites by red foxes. Likewise, our SEM indicated urbanization had a direct negative effect on coyotes and red foxes, but there was no effect of coyote occupancy on fox occupancy. Collectively, these results do not support the “spatial human shield effect” across the Chicago metropolitan area. However, we cannot rule out that such an effect could have operated across Chicago in the past before coyotes became widespread and red foxes rare. Our monitoring started a decade or more after coyotes increased in Chicago, and we could be witnessing the spatial outcome of past intraguild predation.
Species distributions and interactions in different urban settings can vary making it challenging to generalize (Sévêque et al. 2020; Fidino et al. 2021). Spatial analyses of natural areas in Cleveland, Ohio revealed red foxes positively responded to development, whereas coyotes were negatively impacted, indicating the “spatial human shield effect” occurs there (Moll et al. 2018). Unlike Chicago, Cleveland has varied terrain with slopes and this difference in topography might influence how coyotes and red foxes interact in urban areas. Moll et al. (2018) suggested red foxes may use steeper slopes to avoid coyotes, as more energy is required when using sloped areas. In Madison, Wisconsin, spatial partitioning also occurred between the two species, as coyotes used more natural areas while red foxes avoided these habitats (Mueller et al. 2018). Gosselink et al. (2003) also observed red foxes using urban areas as spatial refuges from coyotes in Champaign, Illinois, which is a moderate-sized city that lacks forest preserves large enough to maintain coyote territories. Similar to our study, however, Parsons et al. (2019) found no evidence for the “spatial human shield effect” between coyotes and red foxes across Washington, DC, and Raleigh, North Carolina. Using species interaction models, Parsons et al. (2019) noted a positive interaction between the two canids, although this relationship was weak.
Surprisingly, an interaction between human activity and coyote photographic rate best explained local extinctions by red foxes (Fig. 4). At sites where coyotes were rare, fox extinction was related positively to human activity. In contrast, where coyotes were more common, red foxes were less likely to go extinct at sites with higher human activity. One hypothesis for this pattern is that the two species may co-occur at certain high-quality sites, with enough suitable habitat for both canids, but coyotes typically use areas with less human activity while red foxes tolerate areas closer to humans. Such a scenario would represent a spatial human shield effect at a finer, within-site level.
Our study focused on top-down interactions, but prey resources can affect expected outcomes of intraguild predation (Holt and Polis 1997; Robinson et al. 2014), perhaps between coyotes and foxes in urban settings (Mueller et al. 2018). However, only larger prey such as white-tailed deer (Odocoileus virginianus) and eastern cottontails (Sylvilagus floridanus) can be reliably monitored with our camera traps, but rodents also are important prey for coyotes and red foxes in suburban areas (Randa et al. 2009; Peterson et al. 2021). Future studies may consider adding small mammal trapping at a subset of camera sites (DaVanon et al. 2016) to understand how prey abundance influences urban predator interactions.
Another caveat is that our inferences are restricted to spatial distributions across our camera trap sites, which were limited to natural areas, golf courses, city parks, and cemeteries. Thus, we could not analyze how coyotes and red foxes use the urban landscape matrix outside of green spaces (Gese et al. 2012). For example, we may be missing residential backyard use by these species. Nevertheless, the green spaces that we sampled represent the primary types of suitable habitats in Chicago (Fidino et al. 2016; Gallo et al. 2017) and provide valuable insights on species interactions.
Although we found no evidence of spatial partitioning between the canid species, our results suggest a role for temporal niche partitioning in species coexistence. Overall, coyotes and red foxes had statistically significant differences in diel activity patterns, but retained fairly high temporal overlap, so the divergence may not be biologically relevant. However, the degree of temporal niche partitioning changed across our urbanization gradient. Separation in diel activity for coyotes and red foxes was stronger at more urbanized sites. Coyotes were more active during the crepuscular hours (i.e., one hour before and after sunrise and sunset), whereas red fox activity peaked around midnight. This shift toward nocturnal activity in more developed areas (Wang et al. 2015; Gaynor et al. 2018) by red foxes could partly explain why they were less likely to go extinct at sites with high human and coyote activity. At more rural sites, both species increased diurnal activity and did not display strong differences in peak activity.
As predicted, both canid species had low temporal overlap in activity with humans (see also Moll et al. 2018). Human diurnal activity increased in more suburban areas, whereas crepuscular and nocturnal activity was higher in more urban areas. These changes in human behavior across the gradient could contribute to shifts in activity by urban wildlife (Gaynor et al. 2018) and the likelihood of direct human-wildlife interactions.
Understanding how predator interactions change across urban landscapes is important as urbanization continues to expand and transform ecosystems (Sévêque et al. 2020). Furthermore, it is essential to incorporate both spatial and temporal analyses because partitioning between dominant and subordinate predators can occur in space, time, or both. Finally, continuing to compare occupancy dynamics and activity patterns for species in Chicago and other smaller cities (Magle et al. 2019) will establish which predator interactions are consistent across urban systems or how they change with scale (Uchida et al. 2021). Identifying generalizations that hold across cities, and drivers of differences among cities, will inform urban planning that can benefit biodiversity (Fidino et al. 2021).
Data availability
Not applicable.
Code availability
Not applicable.
References
Ahlers AA, Heske EJ, Schooley RL (2016) Prey distribution, potential landscape supplementation, and urbanization affect occupancy dynamics of American mink in streams. Landsc Ecol 31:1601–1613. https://doi.org/10.1007/s10980-016-0350-5
Batschelet E (1981) Circular statistics in biology. Academic Press, London
Bauder JM, Allen ML, Ahlers AA, Benson TJ, Miller CA, Stodola KW (2022) Long-term data reveal equivocal evidence for intraguild suppression among sympatric canids. Biodivers Conserv 31:2965–2979
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer-Verlag, New York
Chicago Metropolitan Agency for Planning (2016) 10-county region. CMAP Data Hub. https://datahub.cmap.illinois.gov/dataset/high-resolution-land-cover-ne-illinois-and-nw-indiana-2010. Accessed Nov 2019
Cosentino BJ, Schooley RL, Phillips CA (2010) Wetland hydrology, area, and isolation influence occupancy and spatial turnover of the painted turtle, Chrysemys picta. Landsc Ecol 25:1589–1600. https://doi.org/10.1007/s10980-010-9529-3
Crooks KR (2002) Relative sensitivities of mammalian carnivores to habitat fragmentation. Conserv Biol 16:488–502. https://doi.org/10.1046/j.1523-1739.2002.00386.x
Crooks KR, Soule ME (1999) Mesopredator release and avifaunal extinctions in a fragmented system. Nature 400:563–566. https://doi.org/10.1038/23028
Crooks KR, Riley SPD, Gehrt SD, Gosselink TE, Van Deelen TR (2010) Community ecology of urban carnivores. In: Gehrt SD, Riley SPD, Cypher BL (eds) Urban carnivores: ecology, conflict, and conservation, 1st edn. Baltimore, Maryland, pp 185–196
Cusa M, Jackson DA, Mesure M (2015) Window collisions by migratory bird species: urban geographical patterns and habitat associations. Urban Ecosyst 18:1427–1446. https://doi.org/10.1007/s11252-015-0459-3
DaVanon KA, Howard LK, Mabry KE, Schooley RL, Bestelmeyer BT (2016) Effects of exurban development on trophic interactions in a desert landscape. Landsc Ecol 31:2343–2354
ESRI (2019) Environmental Systems Research Institute. Redlands, California
Farris ZJ, Kelly MJ, Karpanty S, Ratelolahy F (2015) Patterns of spatial co-occurrence among native and exotic carnivores in north-eastern Madagascar. Anim Conserv 19:189–198. https://doi.org/10.1111/acv.12233
Fidino M, Gallo T, Lehrer EW, Murray MH, Kay CAM, Sander HA, MacDougall B, Salsbury CM, Ryan TJ, Angstmann JL, Belaire JA, Dugelby B, Schell CJ, Stankowich T, Amaya M, Drake D, Hursh SH, Ahlers AA, Williamson J, Hartley LM, Zellmer AJ, Simon K, Magle SB (2021) Landscape-scale differences among cities alter common species’ responses to urbanization. Ecol Appl 31:1–12. https://doi.org/10.1002/eap.2253
Fidino MA, Lehrer EW, Magle SB (2016) Habitat dynamics of the Virginia opossum in a highly urban environment. Am Midl Nat 175:155–167. https://doi.org/10.1674/0003-0031-175.2.155
Fischer JD, Cleeton SH, Lyons TP, Miller JR (2012) Urbanization and the predation paradox: the role of trophic dynamics in structuring vertebrate communities. BioScience 62:809–818. https://doi.org/10.1525/bio.2012.62.9.6
Fiske I, Chandler R (2011). unmarked: An R package for fitting hierarchical models of wildlife occurrence and abundance. J Stat Softw 43:1–23. https://www.jstatsoft.org/v43/i10/
Gallo T, Fidino M, Lehrer EW, Magle S (2017) Mammal diversity and metacommunity dynamics in urban green spaces: implications for urban wildlife conservation. Ecol Appl 27:2330–2341. https://doi.org/10.1002/eap.1611
Gaynor KM, Hojnowski CE, Carter NH, Brashares JS (2018) The influence of human disturbance on wildlife nocturnality. Science 360:1232–1235
Gehrt SD, Anchor C, White LA (2009) Home range and landscape use of coyotes in a metropolitan landscape: conflict or coexistence? J Mammal 90:1045–1057. https://doi.org/10.1644/08-MAMM-A-277.1
Gehrt SD, Riley SPD (2010) Coyotes (Canis latrans). In: Gehrt SD, Riley SPD, Cypher BL (eds) Urban carnivores: ecology, conflict, and conservation, 1st edn. Baltimore, Maryland, pp 78–95
Gese EM, Morey PS, Gehrt SD (2012) Influence of the urban matrix on space use of coyotes in the Chicago metropolitan area. J Ethol 30:413–425. https://doi.org/10.1007/s10164-012-0339-8
Gese EM, Stotts TE, Grothe S (1996) Interactions between coyotes and red foxes in Yellowstone National Park, Wyoming. J Mammal 77:377–382. https://doi.org/10.2307/1382811
Gompper ME (2002) Top carnivores in the suburbs? Ecological and conservation issues raised by colonization of north-eastern North America by coyotes. BioScience 52:185–190. https://doi.org/10.1641/0006-3568(2002)052[0185:TCITSE]2.0.CO;2
Gosselink TE, Van Deelen TR, Warner RE, Joselyn MG (2003) Temporal habitat partitioning and spatial use of coyotes and red foxes in East-Central Illinois. J Wildl Manag 67:90–103. https://doi.org/10.2307/3803065
Gosselink TE, Van Deelen TR, Warner RE, Mankin PC (2007) Survival and cause-specific mortality of red foxes in agricultural and urban areas of Illinois. J Wildl Manag 71:1862–1873. https://doi.org/10.2193/2006-020
Grace JB (2006) Structural equation modeling and natural systems. Cambridge University Press, Cambridge
Grace JB, Bollen KA (2005) Interpreting the results from multiple regression and structural equation models. Bull Ecol Soc Am 86:283–295. https://doi.org/10.1890/0012-9623(2005)86[283:ITRFMR]2.0.CO;2
Grace JB, Anderson TM, Olff H, Scheiner SM (2010) On the specification of structural equation models for ecological systems. Ecol Monogr 80:67–87. https://doi.org/10.1890/09-0464.1
Grace JB, Scheiner SM, Schoolmaster DR (2015) Structural equation modeling: building and evaluating causal models. In: Fox G, Negrete-Yankelevich S, Sosa VJ (eds) Ecological statistics: contemporary theory and application, 1st edn. Oxford, UK, pp 168–199
Greenspan E, Nielsen CK, Cassel KW (2018) Potential distribution of coyotes (Canis latrans), Virginia opossums (Didelphis virginiana), striped skunks (Mephitis mephitis), and raccoons (Procyon lotor) in the Chicago Metropolitan Area. Urb Ecosyst 21:983–997. https://doi.org/10.1007/s11252-018-0778-2
Hadidian J, Prange S, Rosatte R, Riley SPD, Gehrt SD (2010) Raccoons (Procyon lotor). In: Gehrt SD, Riley SPD, Cypher BL (eds) Urban carnivores: ecology, conflict, and conservation, 1st edn. Baltimore, Maryland, pp 35–47
Hody JW, Kays R (2018) Mapping the expansion of coyotes (Canis latrans) across North and Central America. ZooKeys 759:81–97. https://doi.org/10.3897/zookeys.759.15149
Holt RD, Polis GA (1997) A theoretical framework for intraguild predation. Am Nat 149:745–764. https://doi.org/10.1086/286018
Kovach WL (2009) Oriana—circular statistics for windows, version 3. Kovack Computing Services, Pentraeth
LeFlore EG, Fuller TK, Finn JT, DeStefano S, Organ JF (2019) Wild canid distribution and co-existence in a natural-urban matrix of the pioneer valley of Western Massachusetts. Northeast Nat 26:325–342. https://doi.org/10.1656/045.026.0208
Lehrer EW, Gallo T, Fidino M, Kilgour RJ, Wolff PJ, Magle SB (2021) Urban bat occupancy is highly influenced by noise and the location of water: considerations for nature-based urban planning. Landsc Urban Plan 210:1–9. https://doi.org/10.1016/j.landurbplan.2021.104063
Lesmeister DB, Nielsen CK, Schauber EM, Hellgren EC (2015) Spatial and temporal structure of a mesocarnivore guild in Midwestern North America. Wildl Monogr 191:1–61. https://doi.org/10.1002/wmon.101
Lewis JS, Bailey LL, VandeWoude S, Crooks KR (2015) Interspecific interactions between wild felids vary across scales and levels of urbanization. Ecol Evol 5:5946–5961. https://doi.org/10.1002/ece3.1812
Lonsinger RC, Gese EM, Bailey LL, Waits LP (2017) The roles of habitat and intraguild predation by coyotes on the spatial dynamics of kit foxes. Ecosphere 8(3):e01749. https://doi.org/10.1002/ecs2.1749
MacKenzie DI, Nichols JD, Hines JE, Knutson MG, Franklin AB (2003) Estimating site occupancy, colonization, and local extinction when a species is detected imperfectly. Ecol 84:2200–2207. https://doi.org/10.1890/02-3090
Magle SB, Fidino M, Lehrer EW, Gallo T, Mulligan MP, María Jazmín Ríos MJ, Ahlers AA, Angstmann J, Belaire A, Dugelby B, Gramza A, Hartley L, MacDougall B, Ryan T, Salsbury C, Sander H, Schell C, Simon K, St Onge S, Drake D (2019) Advancing urban wildlife research through a multi-city collaboration. Front Ecol Environ 17:232–239. https://doi.org/10.1002/fee.2030
Magle SB, Hunt VM, Vernon M, Crooks KR (2012) Urban wildlife research: past, present, and future. Biol Conserv 155:23–32. https://doi.org/10.1016/j.biocon.2012.06.018
Magle SB, Poessel SA, Crooks KR, Beck SW (2014) More dogs less bite: the relationship between human-coyote conflict and prairie dog colonies in an urban landscape. Landsc Urban Plan 127:146–153. https://doi.org/10.1016/j.landurbplan.2014.04.013
Magle SM, Reye P, Zhu J, Crooks KR (2010) Extirpation, colonization, and habitat dynamics of a keystone species along an urban gradient. Biol Conserv 143:2146–2155. https://doi.org/10.1016/j.biocon.2010.05.027
Malhotra R, Lima S, Harris NC (2022) Temporal refuges of a subordinate carnivore vary across rural-urban gradient. Ecol Evol 12:e9310
McKinney M (2002) Urbanization, biodiversity, and conservation. BioScience 52:883–890. https://doi.org/10.1641/0006-3568(2002)052[0883:UBAC]2.0.CO;2
Meredith M, Ridout M (2014) Overlap: estimates of coefficient of overlapping for animal activity patterns. https://cran.r-project.org/web/packages/overlap/index.html
Moll RJ, Cepek JD, Lorch PD, Dennis PM, Robison T, Millspaugh JJ, Montgomery RA (2018) Humans and urban development mediate the sympatry of competing carnivores. Urban Ecosyst 21:765–778. https://doi.org/10.1007/s11252-018-0758-6
Mueller MA, Drake D, Allen ML (2018) Coexistence of coyotes (Canis latrans) and red foxes (Vulpes vulpes) in an urban landscape. PLoS ONE 13(1):e0190971. https://doi.org/10.1371/journal.pone.0190971
Murray MH, St. Clair CC (2015) Individual flexibility in nocturnal activity reduces risk of road mortality for an urban carnivore. Behav Ecol 26:1520–1527. https://doi.org/10.1093/beheco/arv102
Newsome TM, van Eeden LM (2017) The effects of food wastes on wildlife and humans. Sustainability 9(7):1269. https://doi.org/10.3390/su9071269
Ordeñana MA, Crooks KR, Boydston EE, Fisher RN, Lyren LM, Siudyla S, Haas CD, Harris S, Hathaway SA, Turschak GM, Miles AK, Van Vuren DH (2010) Effects of urbanization on carnivore species distribution and richness. J Mammal 91:1322–1331. https://doi.org/10.1644/09-MAMM-A-312.1
Parsons AW, Rota CT, Forrester T, Baker-Whatton MC, McShea WJ, Schuttler SG, Millspaugh JJ, Kays R (2019) Urbanization focuses carnivore activity in remaining natural habitats, increasing species interactions. J Appl Ecol 56:1894–1904. https://doi.org/10.1111/1365-2664.13385
Parsons AW, Kellner KF, Rota CT, Schuttler SG, Millspaugh JJ, Kays RW (2022) The effect of urbanization on spatiotemporal interactions between gray foxes and coyotes. Ecosphere 13:e3993
Peterson M, Baglieri M, Mahon K, Ries Sarno RJ., L, Burman P, Grigione MM, (2021) The diet of coyotes and red foxes in Southern New York. Urban Ecosyst 24:1–10. https://doi.org/10.1007/s11252-020-01010-5
Poessel SA, Gese EM, Young JK (2017) Environmental factors influencing the occurrence of coyotes and conflicts in urban areas. Landsc Urban Plan 157:259–269. https://doi.org/10.1016/j.landurbplan.2016.05.022
Polis GA, Meyers CA (1989) The ecology and evolution of intraguild predation: potential competitors that eat each other. Annu Rev Ecol Syst 20:297–330. https://doi.org/10.1016/j.landurbplan.2016.05.022
Prugh LR, Sivy KJ (2020) Enemies with benefits: integrating positive and negative interactions among terrestrial carnivores. Ecol Lett 23:902–918. https://doi.org/10.1111/ele.13489
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Randa LA, Cooper DM, Meserve PI, Yunger JA (2009) Prey switching of sympatric canids in response to variable prey abundance. J Mammal 90:594–603. https://doi.org/10.1644/08-MAMM-A-092R1.1
Ridout MS, Linkie M (2009) Estimating overlap of daily activity patterns from camera trap data. J Agric Biol Environ Stat 14:322–337. https://doi.org/10.1198/jabes.2009.08038
Riley SPD, Sauvajot RM, Fuller TK, York EC, Kamradt DA, Bromley C, Wayne RK (2003) Effects of urbanization and habitat fragmentation on bobcats and coyotes in Southern California. Conserv Biol 17:566–576. https://doi.org/10.1046/j.1523-1739.2003.01458.x
Robinson QH, Bustos D, Roemer GW (2014) The application of occupancy modeling to evaluate intraguild predation in a model carnivore system. Ecol 95:3112–3123. https://doi.org/10.1890/13-1546.1
Rosseel Y (2012) Lavaan: an R package for structural equation modeling. J Stat Softw 48:1–36
Schmid F, Schmidt A (2006) Nonparametric estimation of the coefficient of overlapping-theory and empirical application. Comput Stat Data Anal 50:1583–1596. https://doi.org/10.1016/j.csda.2005.01.014
Scholz C, Firozpoor J, Kramer-Schadt S, Gras P, Schulze C, Kimming SE, Voigt CC, Ortmann S (2020) Individual dietary specialization in a generalist predator: a stable isotope analysis of urban and rural red foxes. Ecol Evol 10:8855–8870. https://doi.org/10.1002/ece3.6584
Sivy KJ, Pozzanghera CB, Grace JB, Prugh LR (2017) Fatal attraction? Intraguild facilitation and suppression among predators. Am Nat 190:663–679. https://doi.org/10.1086/693996
Sévêque A, Gentle LK, López-Bao JV, Yarnell RW, Uzal A (2020) Human disturbance has contrasting effects on niche partitioning within carnivore communities. Biol Rev 95:1689–1705
Soulsbury CD, Baker PJ, Iossa G, Harris S (2010) Red Foxes (Vulpes vulpes). In: Gehrt SD, Riley SPD, Cypher BL (eds) Urban carnivores: ecology, conflict, and conservation, 1st edn. Baltimore, Maryland, pp 62–77
Tigas LA, Van Vuren DH, Sauvajot RM (2002) Behavioral responses of bobcats and coyotes to habitat fragmentation and corridors in an urban environment. Biol Conserv 108:299–306. https://doi.org/10.1016/S0006-3207(02)00120-9
Tryjanowski P, Morelli F, Mikula P, Krištíne A, Indykiewicz P, Grzywaczewski G, Kronenberg J, Jerzak L (2017) Bird diversity in urban green space: a large-scale analysis of differences between parks and cemeteries in Central Europe. Urban For Urban Green 27:264–271. https://doi.org/10.1016/j.ufug.2017.08.014
U.S. Census Bureau (2010) U.S. Census TIGER/Line 210 block shapefiles. https://www.census.gov/geographies/mapping-files/time-series/geo/tiger-line-file.html. Accessed Nov 2019
U.S. Census Bureau (2020) City and town populations totals: 2010-2020. https://www.census.gov/programs-surveys/popest/technical-documentation/research/evaluation-estimates/2020-evaluation-estimates/2010s-cities-and-towns-total.html. Accessed September 2021
Uchida K, Blakey RV, Burger JR, Cooper DS, Niesner CA, Blumstein DT (2021) Urban biodiversity and the importance of scale. Trends Ecol Evol 36:123–131
VanDerWal J, Falconi L, Januchowski S, Shoo L, Storlie C (2019) SDMTools: Species distribution modelling tools: Tools for processing data associated with species distribution modelling exercises, In R package version 1.1-221.2. http://CRAN.R-project.org/package=SDMTools
Wagnon CJ, Schooley RL, Cosentino BJ (2020) Shrub encroachment creates a dynamic landscape of fear for desert lagomorphs via multiple pathways. Ecosphere 11(9):e03240. https://doi.org/10.1002/ecs2.3240
Wang Y, Moskovits DK (2001) Tracking fragmentation of natural communities and changes in land cover: applications of Landsat data for conservation in an urban landscape (Chicago wilderness). Conserv Biol 15:835–843. https://doi.org/10.1046/j.1523-1739.2001.015004835.x
Wang Y, Allen ML, Wilmers CC (2015) Mesopredator spatial and temporal responses to large predators and human development in the Santa Cruz Mountains of California. Biol Conserv 190:23–33. https://doi.org/10.1016/j.biocon.2015.05.007
Wurth AM, Ellington EH, Gehrt SD (2020) Golf courses as potential habitat for urban coyotes. Wild Soc Bull 44:333–341. https://doi.org/10.1002/wsb.1081
Acknowledgments
We thank the interns and volunteers who assisted with fieldwork and management of the photographic data including R. Hagan, K. McBride, and A. Vrioni. We also thank N. Alexander and C. Wagnon for their input throughout the project. We are grateful to the many landowners who allowed us access to their property, including the forest preserve districts of Cook, DuPage, Lake, and Will counties, the Illinois Department of Natural Resources’ Nature Preserve Commission, the Chicago Park District, and the Archdiocese of Chicago.
Funding
A. Cervantes was supported by a fellowship from the Graduate College at the University of Illinois and by the USDA National Institute of Food and Agriculture (Project 1020529). Additional funding was provided by the Abra Prentice Wilkin Foundation and the Davee Foundation.
Author information
Authors and Affiliations
Contributions
A. Cervantes, T. Gallo, E. Lehrer, S. Magle, and R. Schooley contributed to the study conception and design. E. Lehrer, S. Magle, and R. Schooley coordinated funding and resources. Data analyses were performed by A. Cervantes, M. Fidino, T. Gallo, M. Allen, and R. Schooley. The first draft of the manuscript was written by A. Cervantes and all authors reviewed, edited, and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable; no animals were handled for this research.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflicts of interest
The authors have no relevant financial interests to disclose. T. Gallo and S. Magle serve on the Editorial Board.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cervantes, A.M., Schooley, R.L., Lehrer, E.W. et al. Carnivore coexistence in Chicago: niche partitioning of coyotes and red foxes. Urban Ecosyst 26, 1293–1307 (2023). https://doi.org/10.1007/s11252-023-01368-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-023-01368-2