Abstract
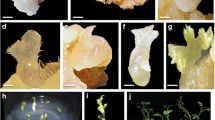
An efficient in vitro plant regeneration system was established through somatic embryogenesis for Anoectochilus elatus Lindley, an endangered jewel orchid. Direct somatic embryogenesis was achieved from nodal explants (17.4 embryos per explant with 63.4% response) on Mitra medium supplemented with Morel vitamins, thidiazuron (4.54 µM) and ∞-naphthaleneacetic acid (2.69 µM). Simultaneously, a protocol was developed for indirect somatic embryogenesis from internodal explant, produced embryogenic calli and embryos (31.3 embryos with 76.4% response) on same medium amended with 50 mg/L peptone and 5% coconut water. Both types of embryogenic pathways, produced morphologically similar globular embryos in the form of protocorm like bodies and successfully germinated on hormone free Mitra medium supplemented with Morel vitamins. Morpho-histological investigation of the embryo revealed the initiation and developmental features of somatic embryos. In vitro regenerated plantlets were successfully established from heterotrophic to a photoautotrophic stage by reducing the nutrient content in culture media, adjusting temperature and humidity through three step method. During the process, no morphological and physiological abnormalities were observed. Hardened plantlets were successfully acclimatized at poly tunnel chamber with 95% of survival rate. Further, inter simple sequence repeats (ISSRs) molecular markers were used to analyse the genetic homogeneity of regenerated plants. Analysis with this method showed that the homogeneity is comparatively higher in direct somatic embryo regenerated plants (94.22%) as compared to plants elevated from an indirect somatic embryo (93.05%). The present study provides morpho-histological and genetically stable plants for germplasm conservation and further utility of this endangered jewel orchid.





Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-diclorophenoxyacetic acid
- 2iP:
-
N6-(2-isopentenyl) adenine
- BA:
-
N6-benzyleadenine
- CW:
-
Coconut water
- DSE:
-
Direct somatic embryogenesis
- IDSE:
-
Indirect somatic embryogenesis
- ISSRs:
-
Inter simple sequence repeats
- NAA:
-
∞-naphthaleneacetic acid
- PEP:
-
Peptone
- PGRs:
-
Plant growth regulators
- PLBs:
-
Protocorm like bodies
- SEM:
-
Scanning electron microscopy
- TDZ:
-
Thidiazuron
References
Abrahamian P, Kantharajah A (2011) Effect of vitamins on in vitro organogenesis of plant. Am J Plant Sci 2:669–674
Alatar AA (2015) Thidiazuron induced efficient in vitro multiplication and ex vitro conservation of Rauvolfia serpentine—a potent antihypertensive drug producing plant. Biotechnol Biotechnol Equip 29:489–497
Al-Khayri JM (2001) Optimization of biotin and thiamine requirements for somatic embryogenesis of date palm (Phoenix dactylifera L.). In Vitro Cell Dev Biol Plant 37:453–456
Baskaran P, Kumari A, Van Staden J (2015) Embryogenesis and synthetic seed production in Mondia whitei. Plant Cell Tissue Organ C 121:205–214
Bhattacharjee A, Chowdhery HJ (2012) Jewel Orchids of India- an overview. International Seminar on “Multidisciplinary approaches in Angiosperm Systematics”. pp 554–556
Bhattacharyya P, Kumaria S (2014) Molecular characterization of Dendrobium nobile Lindl., an endangered medicinal orchid, based on randomly amplified polymorphic DNA. Plant Syst Evol 301:201–210
Bhattacharyya P, Kumaria S, Diengdoh R, Tandon P (2014) Genetic stability and phytochemical analysis of the in vitro regenerate plants of Dendrobium nobile Lindl., an endangered medicinal orchid. Meta Gene 2:489–504
Brearley TA, Vaidya BN, Joshee N (2014) Cytokinin, carbon source, and acclimatization requirements for in vitro propagation of Scutellaria barbata D. Don and Scutellaria racemosa Pers. Am J Plant Sci 5:36–62
Chen JT, Chang WC (2000) Efficient plant regeneration through somatic embryogenesis from callus cultures of Oncidium (Orchidaceae). Plant Sci 160:87–93
Chen JT, Chang WC (2001) Effects of auxins and cytokinins on direct somatic embryogenesis on leaf explants of Oncidium ‘Gower Ramesy’. Plant Growth Regul 34:229–232
Christenhusz MJ, Byng JW (2016) The number of known plants species in the world and its annual increase. Phytotaxa 261:201–217
Dangi B, Khurana-Kaul V, Kothari SL, Kachhwaha S (2014) Micropropagation of Terminalia bellerica from nodal explants of mature tree and assessment of genetic fidelity using ISSR and RAPD markers. Physiol Mol Biol Plants 20:509–516
Dey T, Saha S, Ghosh PD (2015) Somaclonal variation among somatic embryo derived plants, evaluation of agronomically important somaclones and detection of genetic changes by RAPD in Cymbopogon winterianus. S Afr J Bot 96:112–121
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Feng JH, Chen JT (2014) A novel in vitro protocol for inducing direct somatic embryogenesis in Phalaenopsis aphrodite without taking explants. Scientific World J. https://doi.org/10.1155/2014/263642
Gangaprasad A, Latha PG, Seeni S (2000) Micropropagation of terrestrial orchids, Anoectochilus sikkimensis and Anoectochilus regalis. Indian J Exp Biol 38:149–154
George EF (1996) Plant tissue culture procedure—background. In: George EF, Hall MA, De Klerk G (eds) Plant propagation by tissue culture. (3rd edn) Springer, Basingstoke
Gray DJ (1996) Non-zygotic embryogenesis. In: Trigiano RN, Gray DJ (eds) Plant tissue culture concepts and laboratory exercise. CRC Press, New York, pp 133–147
Guo B, Abbasi BH, Zeb A, Xu LL, Wei YH (2011) Thidiazuron a multi-dimensional plant growth regulator. Afr J Biotechnol 10:8984–9000
Hossain MM, Kant R, Van PT, Winarto B, Zeng S, Teixeira da Silva JA (2013) The application of biotechnology to orchids. Crit Rev Plant Sci 32:69–139
Huetteman CA, Preece JE (1993) Thidiazuron a potent cytokinin for woody plant tissue culture. Plant Cell Tissue Organ C 33:105–119
Isah T (2015) Adjustments to in vitro culture conditions and associated anomalies in plants. Acta Biol Cracoviensia Ser Bot 57:9–28
Ishii Y, Takamura T, Goi M, Tanaka M (1998) Callus induction and somatic embryogenesis of Phalaenopsis. Plant Cell Rep 17:446–450
Jalal JS, Jayanthi J (2012) Endemic orchids of peninsular India: a review. J Threatened Taxa 4:3415–3425
Ji A, Geng X, Zhang Y, Yang H, Wu G (2011) Advances in somatic embryogenesis research of horticultural plants. Am J Plant Sci 2:727–732
Juntada K, Taboonmee S, Meetum P, Poomjae S, Chiangmai (2015) Somatic embryogenesis induction from protocorm-like bodies and leaf segments of Dendrobium Sonia ‘Earsakul’. Silpakorn U Sci Tech J 9:9–19
Kaur RP (2015) Photoautotrophic micropropagation an emerging new vista in micropropagation—a review. Agric Rev 36:198–207
Ket NV, Hahn EJ, Park SY, Chakrabarty D, Paek KY (2004) Micropropagation of an endangered orchid Anoectochilus formosanus. Biol Plant 48:339–344
Khilwani B, Kaur A, Ranjan R, Kumar A (2016) Direct somatic embryogenesis and encapsulation of somatic embryos for in vitro conservation of Bacopa monnieri (L.) Wettst. Plant Cell Tissue Organ C 127:433–442
Kodja H, Noirot M, Khoyratty SS, Limbada H, Verpoorte R, Palama LT (2015) Biochemical characterization of embryogenic calli of Vanilla planifolia in response to two years of thidiazuron treatment. Plant Physiol Biochem 96:337–344
Lee YI, Hsu ST, Yeung EC (2013) Orchid protocorm-like bodies are somatic embryos. Am J Bot 100:2121–2131
Ma SS (1991) Somatic embryogenesis and plant regeneration from cell suspension culture of banana. In: Proceedings of symposium on tissue culture of horticultural crops, Taipei, Taiwan, 8–9 March, pp 181–188
Mahendran G, Bai VN (2015) Direct somatic embryogenesis of Malaxis densiflora (A. Rich.) Kuntze. J Genet Eng Biotechnol 14:77–81
Maridass M, Hussain MI, Raju G (2008) Phytochemical survey of orchids in the Tirunelveli hills of South India. Ethnobot Leafl 12:705–712
Masanori T (2002) The cytokinin preference for immature embryo culture of some terrestrial orchids. Comb Proc Int Plant Propag Soc 52:331–334
McAdam SA, Brodribb TJ (2014) Separating active and passive influences on stomatal control of transpiration. Plant Physiol 164:1578–1586
Milella L, Martelli G, Salava J, Fernandez E, Ovesna J, Greco I (2011) Total phenolic content, RAPDs, AFLPs and morphological traits for the analysis of variability in Smallanthus sonchifolius. Genet Resour Crop Evol 58:545–551
Misra S (2007) Orchids of India—a glimpse. Bishen Singh Mahendra Pal Singh, Dehradun (ISBN: 978-81-211-0618-4)
Mitra GC, Prasad RN, Choudhury RA (1976) Inorganic salts and differentiation of protocorms in seed callus of orchid correlative changes in its free amino acid content. Indian J Exp Biol 14:350–351
Mokhtarzadeh S, Hajyzadeh M, Ahmad HA, Khawar KM (2013) The problems in acclimatisation of in vitro multiplied plants of Lavandula angustifolia Miller under field conditions. Acta Hortic (ISHS) 988:71–76
Morel G, Wetmore RH (1951) Tissue culture of monocotyledons. Am J Bot 38:138–140
Moyo M, Aremu AO, Van Staden J (2015) Insights into the multifaceted application of microscopic techniques in plant tissue culture systems. Planta 242:773–790
Mukerji KG, Chamola BP, Sharma AK (2000) Glimpse in Botany. APH Publishing Corporation, New Delhi. (ISBN: 81-7648-204-8)
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Naing AH, Chung JD, Lim KB (2011) Plant regeneration through indirect somatic embryogenesis in Coelogyne cristata Orchid. Am J Plant Sci 2:262–267
Parthibhan S, Rao MV, Kumar TS (2015) In vitro regeneration from protocorms in Dendrobium aqueum Lindley—an imperiled orchid. J Genet Eng Biotechnol 13:227–233
Pikulthong V, Teerakathiti T, Thamchaipenet A, Peyachoknagul S (2016) Development of somatic embryos for genetic transformation in Curcuma longa L. and Curcuma manga Valeton & Zijp. Agric Nat Resour 50:276–285
Rachmawati F, Winarto B, Mattjik AN, Wiendi AMN, Furwito A (2015) Shoot tips derived-somatic embryogenesis in mass propagation of Dendrobium Indonesia Raya ‘Ina’. Emir J Food Agric 27:1–10
Saha S, Adhikari S, Dey T, Ghosh P (2016) RAPD and ISSR based evaluation of genetic stability of microprpagated plantlets of Morus alba L. variety S-1. Meta Gene 7:7–15
Sarkar MK (2012) Management strategies for endemic and threatened medicinal plants in India- a geoinformatic approach with special reference to Kalakad Mundanthurai Tiger Reserve, Southern Western Ghats of Tamil Nadu, India, vol 1, Department of Environment, Government of Tamil Nadu, Chennai
Seth S, Rath CS, Rout SC, Rout GR, Panigrahi J (2016) Somatic embryogenesis in Abutilon indicum (L.) Sweet and assessment of genetic homogeneity using SCoT markers. Plant Biosyst 0:1–11
Shajahan A, Raju CS, Thilip C, Varutharaju K, Faizal K, Mehaboob VM, Aslam A (2016) Direct and indirect somatic embryogenesis in mango ginger (Curcuma amada Roxb.). In: Loyola-Vargas VM, Ochoa-Alejo N (eds) Somatic embryogenesis: fundamental aspects and applications, Springer, Cham, pp 367–379
Shekhawat MS, Manokari M (2016) In vitro regeneration frequency, micro-morphological studies and ex vitro rooting of Hemidesmus indicus (L.) R. Br.: a multi-potent endangered climber. Indian J Plant Physiol 21:151–160
Sherif NA, Franklin Benjamin JH, Muthukrishnan S, Senthil Kumar T, Rao MV (2012) Regeneration of plantlets from nodal and shoot tip explants of Anoectochilus elatus Lindley, an endangered terrestrial orchid. Afr J Biotechnol 11:7549–7553
Sherif NA, Senthil Kumar T, Rao MV (2016) In vitro regeneration by callus of Anoectochilus elatus Lindley, an endangered terrestrial jewel orchid. In Vitro Cell Dev Biol Plant 52:72–80
Shiau YJ, Nalawade SM, Hsai CN, Tsay HS (2005) Propagation of Haemaria discolor via in vitro seed germination. Biol Plant 49:341–346
Soundar Raju C, Aslam A, Shajahan A (2015) High frequency direct somatic embryogenesis and plant regeneration from leaf base explants of turmeric (Curcuma longa L.). Plant Cell Tissue Organ C 122:79–87
Strosse H, Schoofs H, Panis B, Andre E, Reyniers K, Swennen R (2006) Development of embryonic cell suspension from shoot meristematic tissue in Banana and plantains (Musa Spp.). Plant Sci 170:104–112
Takagi H, Sugawara S, Saito T, Tasaki H, Lu Y, Guan K, Han DS, Godo T, Nakano M (2011) Plant regeneration via direct and indirect adventitious shoot formation and chromosome doubled somaclonal variation in Titanotrichum oldhamii (Hemsl.) Solereder. Plant Biotechnol Rep 5:187–195
Teixeira Da Silva JA (2013) Orchids: advances in tissue culture, genetics, phytochemistry and transgenic biotechnology. Floric Ornam Biotechnol 7:1–52
Viehmannova I, Bortlova Z, Vitamvas J, Cepkova HP, Eliasova K, Svobodova E, Travnickova M (2014) Assessment of somaclonal variation in somatic embryo derived plants of yacon [Smallanthus sonchifolius (Poepp. And Endl.) H. Robinson] using inter simple sequence repeat analysis and flow cytometry. Electron J Biotechnol 17:102–106
Vinoth A, Ravindhran R (2016) Efficient plant regeneration of watermelon (Citrullus lanatus Thunb.) via somatic embryogenesis and assessment of genetic fidelity using ISSR markers. In Vitro Cell Dev Biol Plant 52:107–115
Zhang F, Lv Y, Dong H, Guo S (2010) Analysis of genetic stability through inter simple sequence repeats molecular markers in microprpagated plantlets of Anoectochilus formosanus Hayata, Medicinal Plant. Bio Pharm Bull 33:384–388
Zhang A, Wang H, Shao Q, Xu M, Zhang W, Li M (2015) Large scale in vitro propagation of Anoectochilus roxburghii for commercial application: pharmaceutical important and ornamental plant. Ind Crops Prod 70:158–162
Acknowledgements
The corresponding author is grateful to the University Grant Commission (UGC), Delhi, India for providing Emeritus Fellowship. All the authors are grateful to Dr. P. Palani, Assistant Professor, Centre for Advanced Studies in Botany, University of Madras, Guindy Campus, Chennai 600 025, Tamil Nadu, India, for valuable help in scanning electron microscopic observation.
Author information
Authors and Affiliations
Contributions
NAS, JHFB, TSK and MVR are equally contributed to bringing out the manuscript in a successful manner.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Communicated by Sergio J. Ochatt.
Rights and permissions
About this article
Cite this article
Sherif, N.A., Franklin Benjamin, J.H., Senthil Kumar, T. et al. Somatic embryogenesis, acclimatization and genetic homogeneity assessment of regenerated plantlets of Anoectochilus elatus Lindl., an endangered terrestrial jewel orchid. Plant Cell Tiss Organ Cult 132, 303–316 (2018). https://doi.org/10.1007/s11240-017-1330-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-017-1330-4