Abstract
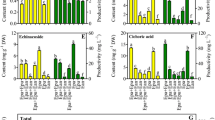
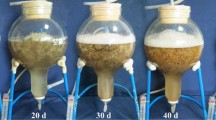
Oplopanax elatus Nakai is a high-value medicinal plant, but the availability of this resource is rare under natural conditions. To provide a novel source of O. elatus products, we used balloon-type airlift bioreactor systems to investigate a culture method. This method was developed to obtain adventitious roots (ARs), which contain a high yield of bioactive compounds. Antioxidant and anticancer properties were also investigated to evaluate the bioactivity of AR extracts. The bioreactor culture more efficiently promoted AR growth and bioactive compound accumulation (polysaccharides, phenolics, and flavonoids) than the agitated flask and solid cultures. During bioreactor culture, the maximum AR fresh (114.6 g L−1) and dry (12.7 g L−1) weights were found in the medium supplemented with 50 g L−1 sucrose and 3 mg L−1 IBA. The highest bioactive compound content was also found in the same medium; the productivities of polysaccharides, phenolics, and flavonoids reached 1913.9, 459.7, and 713.7 g L−1, respectively. The polysaccharide, phenolic, and flavonoid content of ARs from the bioreactor culture were compared with those of roots from field-grown plants. The polysaccharide content reached the maximum levels in ARs (152.5 mg g−1 DW) and in roots of 3-year-old plants (147.5 mg g−1 DW), with no evident difference. The phenolic (32.1 mg g−1 DW) and flavonoid (50.5 mg g−1 DW) contents in ARs were higher than those in the roots of plants grown in the field for 2 and 3 years. The kinetic study showed that the highest AR biomass and bioactive compound productivity were found at 40 days. Therefore, 40 days of culture is appropriate for the mass production of bioactive compounds from O. elatus by AR culture in bioreactor systems. Polysaccharide, phenolic, and flavonoid contents were higher in bioreactor-cultured ARs than in the roots of 2-and 3-year-old field-grown plants. ARs exhibited an effective antioxidant activity; the rate of DPPH radical scavenging in the AR extract was higher than that in the root extracts of field-grown plants (2 and 3 years); IC50 of AR extract was only 33.5 μg mL−1. The AR extract also elicited anticancer effect on human cancer cell lines, particularly the gastric cancer cell line AGS. AGS cell proliferation was reduced by 50 % after 24 h of these cells were exposed to 78.3 μg mL−1 AR extracts. Our results suggested that the bioreactor culture of O. elatus AR can be used to produce bioactive compounds (polysaccharides, phenolics, and flavonoids). ARs are also a potential resource to develop O. elatus products.






Similar content being viewed by others
References
Baque MA, Hahn EJ, Paek KY (2010) Growth, secondary metabolite production and antioxidant enzyme response of Morinda citrifolia adventitious root as affected by auxin and cytokinin. Plant Biotechnol Rep 4:109–116
Baque MA, Elgirban A, Lee EJ, Paek KY (2012) Sucrose regulated enhanced induction of anthraquinone, phenolics, flavonoids biosynthesis and activities of antioxidant enzymes in adventitious root suspension cultures of Morinda citrifolia (L.). Acta Physiol Plant 34:405–415
Baque MA, Shiragi MHK, Moh SH, Paek KY (2013) Production of biomass and bioactive compounds by adventitious root suspension cultures of M. citrifolia (L.) in a liquid-phase airlift balloon-type bioreactor. In Vitro Cell Dev Biol-Plant 49:739–749
Baque MA, Murthy HN, Paek KY (2014) Adventitious root culture of Morinda citrifolia in bioreactors for production of bioactive compounds. In: Paek KY, Murthy HN, Zhong JJ (eds) Production of biomass and bioactive compounds using bioreactor technology. Springer, Dordrecht, pp 185–222
Chang C, Yang M, Wen H, Chem J (2002) Estimation of total flavonoids content in propolis by two complementary colorimetric methods. J Food Drug Anal 10:178–182
Coste A, Vlase L, Halmagyi A, Deliu C, Coldea G (2011) Effects of plant growth regulators and elicitors on production of secondary metabolites in shoot cultures of Hypericum hirsutum and Hypericum maculatum. Plant Cell Tissue Organ Cult 106:279–288
Cui XH, Murthy HN, Wu CH, Paek KY (2010) Sucrose-induced osmotic stress affects biomass, metabolite, and antioxidant levels in root suspension cultures of Hypericum perforatum L. Plant Cell Tiss Organ Cult 103:7–14
Cui XH, Murthy HN, Jin YX, Yim YH, Kim JY, Paek KY (2011) Production of adventitious root biomass and secondary metabolites of Hypericum perforatum L. in a balloon type airlift reactor. Bioresour Technol 102:10072–10079
Cui HY, Baque MA, Lee EJ, Paek KY (2013) Scale-up of adventitious root cultures of Echinacea angustifolia in a pilot-scale bioreactor for the production of biomass and caffeic acid derivatives. Plant Biotechnol Rep 7:297–308
Dou DQ, Hu XY, Zhao YR, Kang TG, Liu FY, Kuang HX, Smith DC (2009) Studies on the anti-psoriasis constituents of Oplopanax elatus Nakai. Nat Prod Res 23:334–342
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Georgiev MI, Weber J, Maciuk A (2009) Bioprocessing of plant cell cultures for mass production of targeted compounds. Appl Microbiol Biotechnol 83:809–823
Hahlbrock K, Kuhlen E (1972) Relationship between growth of parsley and soybean cells in suspension cultures and changes in the conductivity of the culture medium. Planta 108:271–278
Hseu YC, Chang WH, Chen CS, Liao JW, Huang CJ, Lu FJ, Chia YC, Hsu HK, Wu JJ, Yang HL (2008) Antioxidant activities of Toona sinensis leaves extracts using different antioxidant models. Food Chem Toxicol 46:105–114
Jeong CS, Chakrabarty D, Hahn EJ, Lee LH, Paek KY (2006) Effects of oxygen, carbon dioxide and ethylene on growth and bioactive compound production in bioreactor culture of ginseng adventitious roots. Biochem Eng J 27:252–263
Jeong JA, Wu CH, Murthy HN, Hahn EJ, Paek KY (2009) Application of an airlift bioreactor system for the production of adventitious root biomass and caffeic acid derivatives of Echinacea purpurea. Biotechnol Bioprocess Eng 14:91–98
Kim JH, Eom SH, Lee HS, Kim KJ, Yu YC, Kwon YS, Lee JK, Kim MJ (2007) Assessment on antioxidant properties of Oplopanax elatus Nakai in vitro. Kor J Med Crop Sci 15:112–119
Lee SW, Kim YM, Kim WW, Chung JM (2002) Genetic variation of I-SSR markers in the natural populations of a rare and endangered tree species, Oplopanax elatus in Korea. J Kor For Soc 91:565–573
Lee EJ, Park SY, Paek KY (2015) Enhancement strategies of bioactive compound production in adventitious root cultures of Eleutherococcus koreanum Nakai subjected to methyl jasmonate and salicylic acid elicitation through airlift bioreactors. Plant Cell Tiss Organ Cult 120:1–10
Li M, Hirata Y, Xu G, Niwa M (1990) Determination of polysaccharide contents in the drugs of Dendrobium. Chinese Tradit Herb Drugs 21:10–12
Li HJ, Lian ML, Gao R, Yu XK, Piao XC (2012) Effect of salt concentration, carbon sources and phosphate concentration on adventitious root growth of Oplopanax elatus Nakai in suspension culture. J Anhui Agri Sci 40(11229–11230):11273
Liu PP, Qu Y, Dou DQ, Kang TG, Smith D (2012) Determination of anti-cancer constituents in Oplopanax horridus and Oplopanax elatus. J Chem Soc Pak 34:417–423
Moon HK, Kim JA, Park SY, Kim YW, Kang HD (2006) Somatic embryogenesis and plantlet formation from a rare and endangered tree species, Oplopanax elatus. J Plant Biol 49:320–325
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco cultures. Physiol Plant 15:473–497
Murthy HN, Hahn EJ, Paek KY (2008) Adventitious roots secondary metabolism. Chin J Biotech 24:711–716
Park SY, Moon HK, Murthy HN, Kim YW (2011) Improved growth and acclimatization of somatic embryo-derived Oplopanax elatus plantlets by ventilated photoautotrophic culture. Biol Plantarum 55:559–562
Peyman PG (2015) The extraction process optimization of antioxidant polysaccharides from Marshmallow (Althaea officinalis L.) roots. Int J Biol Macromol 75:51–57
Prasanna DB, Floyd LI, Leonard DH (2013) Determination of specific oxygen uptake rate of Photorhabdus luminescens during submerged culture in lab scale bioreactor. Biocontrol Sci Technol 23:1458–1468
Rajesh M, Sivanandhan G, Arun M, Vasudevan V, Theboral J, Girija S, Manickavasagam M, Selvaraj N, Ganapathi A (2014) Factors influencing podophyllotoxin production in adventitious root culture of Podophyllum hexandrum Royle. Acta Physiol Plant 36:1009–1021
Rao SR, Ravishankar GA (2002) Plant cell cultures: chemical factories of secondary metabolites. Biotechnol Adv 20:101–153
Ryu DDY, Lee SO, Romani RJ (1990) Determination of growth rate for plant cell cultures: comparative studies. Biotechnol Bioeng 35:305–311
Sarikurkcu C, Tepe B, Daferera D, Polissiou M, Harmandar M (2008) Studies on the antioxidant activity of the essential oil and methanol extract of Marrubium globosum subsp. globosum (Lamiaceae) by three different chemical assays. Bioresource Technol 99:4239–4246
Scheck AC, Perry K, Hank NC, Clark WD (2006) Anticancer activity of extracts derived from the mature roots of Scutellaria baicalensis on human malignant brain tumor cells. BMC Complement Altern Med 6:27–35
Shohael AM, Ali MB, Yu KW, Hahn EJ, Paek KY (2006) Effect of temperature on secondary metabolites production and antioxidant enzyme activities in Eleutherococcus senticosus somatic embryos. Plant Cell Tiss Organ Cult 85:219–228
Singleton V, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol 299:152–158
Sivakumar G, Yu KW, Paek KY (2005) Production of biomass and ginsenosides from adventitious roots of Panax ginseng in bioreactor cultures. Eng Life Sci 5:333–342
Thorpe T, Stasolla C, Yeung EC, de Klerk GJ, Roberts A, George EF (2008) The components of plant tissue culture media II: organic additions, osmotic and pH effects, and support systems. In: George EF, Hall MA, de Klerk GJ (eds) Plant propagation by tissue culture, vol 1, 3rd edn. Springer, Dordrecht, pp 115–174
Vinterhalter B, Krstić-Milošević D, Janković T, Pljevljakušić D, Ninković S, Smigocki A, Vinterhalter D (2015) Gentiana dinarica Beck. hairy root cultures and evaluation of factors affecting growth and xanthone production. Plant Cell Tiss Organ Cult 121:667–679
Wang J, Man SL, Gao WY, Zhang LM, Huang LQ (2013a) Cluster analysis of ginseng tissue cultures, dynamic change of growth, total saponins, specific oxygen uptake rate in bioreactor and immuno-regulative effect of ginseng adventitious root. Ind Crop Prod 41:57–63
Wang J, Zhang J, Gao WY, Wang Q, Yin SS, Liu H, Man SL (2013b) Identification of triterpenoids and flavonoids, step-wise aeration treatment as well as antioxidant capacity of Glycyrrhiza uralensis Fisch. cell. Ind Crop Prod 49:675–681
Wu CH, Dewir YH, Hahn EJ, Paek KY (2006) Optimization of culturing conditions for the production of biomass and phenolics from adventitious roots of Echinacea angustifolia. J Plant Biol 49:193–199
Wu SQ, Lian ML, Gao R, Park SY, Piao XC (2011) Bioreactor application on adventitious root culture of Astragalus membranaceus. In Vitro Cell Dev Biol Plant 47:719–724
Wu SQ, Yu XK, Lian ML, Park SY, Piao XC (2014) Several factors affecting hypericin production of Hypericum perforatum during adventitious root culture in airlift bioreactors. Acta Physiol Plant 36:975–981
Yang JF, Piao XC, Sun D, Lian ML (2010a) Production of protocorm-like bodies with bioreactor and regeneration in vitro of oncidium ‘Sugar Sweet’. Sci Hortic 125:712–717
Yang MC, Kwon HC, Kim YJ, Lee KR, Ok YH (2010b) Oploxynes A and B, polyacetylenes from the stems of Oplopanax elatus. J Nat Prod 73:801–805
Yin S, Gao W, Liang Y, Wang J, Liu H, Wei C, Zuo B (2013) Influence of sucrose concentration and phosphate source on biomass and metabolite accumulation in adventitious roots of Pseudostellaria heterophylla. Acta Physiol Plant 35:1579–1585
Yu KW, Murthy HN, Jeong CS, Hahn EJ, Paek KY (2005) Organic germanium stimulates the growth of ginseng adventitious roots and ginsenoside production. Process Biochem 40:2959–2961
Yu D, Piao XC, Li Y, Wei NN, Lian ML (2014) Mass production of adventitious roots of Oplopanax elatus in bioreactor. Chin Agric Sci Bull 30:252–255
Zhang J, Gao WY, Wang J (2012) Effects of sucrose concentration and exogenous hormones on growth and periplocin accumulation in adventitious roots of Periploca sepium Bunge. Acta Physiol Plant 34:1345–1351
Zobayed S, Saxena PK (2004) Production of St. John’s wort plants under controlled environment for maximizing biomass and secondary metabolites. In Vitro Cell Dev Biol-Plant 40:108–114
Zou Y, Liao S, Shen W, Liu F, Tang C, Chen C (2012) Phenolics and antioxidant activity of mulberry leaves depend on cultivar and harvest month in Southern China. Mol Sci 13:16544–16553
Acknowledgments
This research was supported by the National Science Foundation of China (31260182).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, Y.J., Piao, X.C., Liu, J.S. et al. Bioactive compound production by adventitious root culture of Oplopanax elatus in balloon-type airlift bioreactor systems and bioactivity property. Plant Cell Tiss Organ Cult 123, 413–425 (2015). https://doi.org/10.1007/s11240-015-0845-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-015-0845-9