Abstract
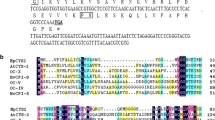
Plant peroxidases (PODs) have been ascribed a variety of biological functions, including hydrogen peroxide detoxification, lignin biosynthesis, hormonal signaling, and stress response. In the present study, ten POD genes, including three ascorbate peroxidases (class I PODs) and seven secretory peroxidases (class III PODs), were cloned from Tamarix hispida. The roles of the ten POD genes were addressed under different abiotic stress conditions, and gene expression profiles in roots, stems, and leaves were evaluated using real-time quantitative reverse-transcribed polymerase chain reaction. Our results showed that the relative abundance of the PODs was markedly different in roots, stems, and leaves, indicating that POD activity differs in these three organs. ThPOD1 and ThPOD8 were the most and least abundant, respectively, in all organs. The expression profiles in response to abiotic stresses were organ specific. All of the genes were highly induced by drought, salt, salt–alkaline, CdCl2, and abscisic acid (ABA) treatments in at least one organ. Five ThPOD genes were induced in roots, stems, and leaves under all of the studied stress conditions, indicating that they are closely associated with abiotic stress. Our results demonstrate that the ten plant peroxidases are all expressed in leaves, stems, and roots, that they are involved in different abiotic stress responses, and that they are controlled by an ABA-dependent stress signaling pathway.








Similar content being viewed by others
References
Azevedo Neto AD, Prisco JT, Eneas-Filho J, de Abreu CEB, Gomes-Filho E (2006) Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ Exp Bot 56:87–94. doi:10.1016/j.envexpbot.2005.01.008
Drazkiewicz M, Skórzyńska-Polit E, Krupa Z (2004) Copper-induced oxidative stress and antioxidant defence in Arabidopsis thaliana. Biometals 17:379–387. doi:10.1023/B:BIOM.0000029417.18154.22
Gao C, Wang Y, Liu G, Yang C, Jiang J, Li H (2008) Expression profiling of salinity-alkali stress responses by large-scale expressed sequence tag analysis in Tamarix hispid. Plant Mol Biol 66:245–258. doi:10.100710.1007/s11103-007-9266-4
Jiang Y, Deyholos MK (2006) Comprehensive transcriptional profiling of NaCl-stressed Arabidopsis roots reveals novel classes of responsive genes. BMC Plant Biol 6:25. doi:10.1186/1471-2229-6-25
Kim YH, Kim CY, Song WK, Park DS, Kwon SY, Lee HS, Bang JW, Kwak SS (2008) Overexpression of sweet potato swpa4 peroxidase results in increased hydrogen peroxide production and enhances stress tolerance in tobacco. Planta 227:867–881. doi:10.1007/s00425-007-0663-3
Koussevitzky S, Suzuki N, Huntington S, Armijo L, Sha W, Cortes D, Shulaev V, Mittler R (2008) Ascorbate peroxidase 1 plays a key role in the response of Arabidopsis thaliana to stress combination. J Biol Chem 283:34197–34203. doi:10.1074/jbc.M806337200
Li H, Wang Y, Jiang J, Liu G, Gao C, Yang C (2009) Identification of genes responsive to salt stress on Tamarix hispida roots. Gene 433:65–71. doi:10.1016/j.gene.2008.12.007
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Maksymiec W, Krupa Z (2006) The effects of short-term exposition to Cd, excess Cu ions and jasmonate on oxidative stress appearing in Arabidopsis thaliana. Environ Exp Bot 57:187–194. doi:10.1016/j.envexpbot.2005.05.006
Menezes-Benavente L, Teixeira FK, Kamei CLA, Margis-Pinheiro M (2004) Salt stress induces altered expression of genes encoding antioxidant enzymes in seedlings of a Brazilian indica rice (Oryza sativa L.). Plant Sci 166:323–331. doi:10.1016/j.plantsci.2003.10.001
Passardi F, Longet D, Penel C, Dunand C (2004) The class III peroxidase multigenic family in rice and its evolution in land plants. Phytochemistry. 65:1879–1893. doi:10.1016/j.phytochem.2004.06.023
Rau S, Miersch J, Neumann D, Weber E, Krauss GJ (2007) Biochemical responses of the aquatic moss Fontinalis antipyretica to Cd, Cu, Pb and Zn determined by chlorophyll fluorescence and protein levels. Environ Exp Bot 59:299–306. doi:10.1016/j.envexpbot.2006.03.001
Rossel JB, Walter PB, Hendrickson L, Chow WS, Poole A, Mulineaux PM, Pogson BJ (2006) A mutation affecting ASCORBATE PEROXIDASE 2 gene expression reveals a link between responses to high light and drought tolerance. Plant Cell Environ 29:269–281. doi:10.1111/j.1365-3040.2005.01419.x
Sharma P, Dubey RS (2004) Ascorbate peroxidase from rice seedlings: properties of enzyme isoforms, effects of stresses and protective roles of osmolytes. Plant Sci 167:541–550. doi:10.1016/j.plantsci.2004.04.028
Sreenivasulu N, Grimm B, Wobus U, Weschke W (2000) Differential response of antioxidant compounds to salinity stress in salt tolerant and salt sensitive seedlings of foxtail millet (Setaria italica). Physiol Plant 109:435–442. doi:10.1034/j.1399-3054.2000.100410.x
Unyayar S, Celik A, Cekic FO, Gozel A (2006) Cadmium-induced genotoxicity, cytotoxicity and lipid peroxidation in Allium sativum and Vicia faba. Mutagenesis 21:77–81. doi:10.1093/mutage/gel001
Xu W, Shi W, Ueda A, Takabe T (2008) Mechanisms of salt tolerance in transgenic Arabidopsis thaliana carrying a peroxisomal ascorbate peroxidase gene from barley. Pedosphere 18:480–495. doi:10.1016/S1002-0160(08)60039-9
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, C., Wang, Y., Liu, G. et al. Cloning of Ten Peroxidase (POD) Genes from Tamarix Hispida and Characterization of their Responses to Abiotic Stress. Plant Mol Biol Rep 28, 77–89 (2010). https://doi.org/10.1007/s11105-009-0129-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-009-0129-9