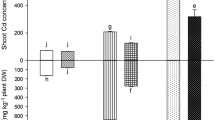
Abstract
Background and aims
Dark septate endophytes (DSE) are a group of multifunctional fungi that ubiquitously colonize the roots of numerous plants worldwide, benefiting the nutrition and adaptation of hosts to various stressed environments. How DSE benefit phosphorus absorption by their host plants remains unclear.
Methods
We established pot cultures and compartmentalized microcosms in the greenhouse and used transcriptomic analysis to examine the response of maize to inoculation with a DSE strain, Exophiala pisciphila H93, which has been found to be capable of promoting plant growth and enhancing the tolerance of maize to heavy metals.
Results
H93 promoted the biomass, phosphorus absorption and photosynthesis of inoculated maize by enhancing phosphorus enzyme activities in the rhizosphere and by decreasing the pH of the rhizosphere compared with non-inoculated controls, either in sufficient or deficient phosphorus conditions in pot cultures. H93 inoculation changed the transcriptional pattern of maize related to 8 metabolic pathways. While H93 did not induce particular phosphate transporter gene expression under deficient phosphorus conditions, it dissolved the sparingly soluble phosphates (tricalcium phosphate) in the rhizosphere.
Conclusions
Enhanced phosphorus absorption by the host plant by DSE (H93) seemed to occur independently of DSE-induced genes involved in phosphorus absorption. The greatest contribution of DSE to phosphorus utilization of hosts might be their efficiency in transferring sparingly soluble phosphorus into soluble phosphate (Pi).






Similar content being viewed by others
Abbreviations
- DSE:
-
Dark septate endophytes
- P:
-
Phosphorus
- Pi:
-
Inorganic orthophosphate
- TCP:
-
Tricalcium phosphate
- Pts:
-
Phosphate transporters
- PBRMs:
-
Plant beneficial rhizosphere microorganisms
- AMF:
-
Arbuscular mycorrhizal fungi
- PAE:
-
Phosphorus acquisition efficiency
- PUE:
-
Phosphorus use efficiency
- PVK:
-
Pikovskaya’s medium
- IR:
-
Inhibition rate
- PR:
-
Promotion rate
References
Addy HD, Piercey MM, Currah RS (2005) Microfungal endophytes in roots. Can J Bot 83:1–13
Avdalović J, Beškoski V, Gojgić-Cvijović G, Mattinen M-L, Stojanović M, Zildžović S, Vrvić MM (2015) Microbial solubilization of phosphorus from phosphate rock by iron-oxidizing Acidithiobacillus sp. B2. Miner Eng 72: 17-22. Doi: https://doi.org/10.1016/j.mineng.2014.12.010
Barrow N (2017) The effects of pH on phosphate uptake from the soil. Plant Soil 410:401–410. https://doi.org/10.1007/s11104-016-3008-9
Baum C, Hrynkiewicz K, Szymańska S, Vitow N, Hoeber S, Fransson PMA, Weih M (2018) Mixture of Salix genotypes promotes root colonization with dark septate endophytes and changes P cycling in the mycorrhizosphere. Front Microbiol 9:1–10. https://doi.org/10.3389/fmicb.2018.01012
Beech IB, Paiva M, Caus M, Coutinho C (2001) Enzymatic activity and within biofilms of sulphate-reducing bacteria. In: Gilbert PG, Allison D, Brading M, Verran J, Walker J (eds) Biofilm community interactions: change or necessity? Boiline, Cardiff, pp 231–239
Berthelot C, Chalot M, Leyval C, Blaudez D (2019) From darkness to light: emergence of the mysterious dark septate endophytes in plant growth promotion and stress alleviation. In: murphy B R, Hodkinson T, Doohan F, Saunders M (eds):Endophytes for a growing world. Cambridge University Press, Cambridge, pp 143
Bueno de Mesquita CP, Sartwell SA, Ordemann EV, Porazinska DL, Farrer EC, King AJ, Spasojevic MJ, Smith JG, Suding KN, Schmidt SK (2018) Patterns of root colonization by arbuscular mycorrhizal fungi and dark septate endophytes across a mostly-unvegetated, high-elevation landscape. Fungal Ecol 36:63–74. https://doi.org/10.1016/j.funeco.2018.07.009
Byrne ME (2009) A role for the ribosome in development. Trends Plant Sci 14:512–519. https://doi.org/10.1016/j.tplants.2009.06.009
Cameron SL (1998) Colonization of Populus tremuloides seedlings by the fungus Phialocephala fortinii in the presence of the ectomycorrhizal fungus Thelephora terrestris. M.Sc. thesis, University of Guelph, Guelph, Ont
Cao G-H, He S, Chen D, Li T, Zhao Z-W (2019) EpABC genes in the adaptive responses of Exophiala pisciphila to metal stress: functional importance and relation to metal tolerance. Appl Environ Microb 85:e01844–e01819. https://doi.org/10.1128/AEM.01844-19
Chaves MM, Pinheiro C, Flexas J (2008) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot-london 103:551–560. https://doi.org/10.1093/aob/mcn125
Chen J-H, Barber SA (1990) Soil pH and phosphorus and potassium uptake by maize evaluated with an uptake model. Soil Sci Soc Am J 54:1032–1036. https://doi.org/10.2136/sssaj1990.03615995005400040017x
Chen Y, Zhi J, Zhang H, Li J, Zhao Q, Xu J (2017) Transcriptome analysis of Phytolacca americana L. in response to cadmium stress. PLoS One 12:e0184681. https://doi.org/10.1371/journal.pone.0184681
Chiu CH, Paszkowski U (2019) Mechanisms and impact of symbiotic phosphate acquisition. Cold Spring Harb Perspect Biol 11:a034603
de Oliveira MG, Moreira de Freitas AL, Liparini Pereira O, Ribeiro da Silva I, Bojkov Vassilev N, Dutra Costa M (2014) Mechanisms of phosphate solubilization by fungal isolates when exposed to different P sources. Ann Microbiol 64:239–249. https://doi.org/10.1007/s13213-013-0656-3
Della Mónica IF, Godoy MS, Godeas AM, Scervino JM (2018) Fungal extracellular phosphatases: their role in P cycling under different pH and P sources availability. J Appl Microbiol 124:155–165. https://doi.org/10.1111/jam.13620
Della Monica IF, Saparrat MCN, Godeas AM, Scervino JM (2015) The co-existence between DSE and AMF symbionts affects plant P pools through P mineralization and solubilization processes. Fungal Ecol 17: 10-17. Doi. https://doi.org/10.1016/j.funeco.2015.04.004
Della Mónica IF, Stefanoni Rubio PJ, Cina RP, Recchi M, Godeas AM, Scervino JM (2014) Effects of the phosphate-solubilizing fungus Talaromyces flavus on the development and efficiency of the Gigaspora rosea-Triticum aestivum symbiosis. Symbiosis 64:25–32. https://doi.org/10.1007/s13199-014-0299-6
Deubel A, Merbach W (2005) Influence of microorganisms on phosphorus bioavailability in soils. In: A Varma, F Buscot (eds) Microorganisms in soils: roles in genesis and functions. Springer Berlin Heidelberg, Berlin, Heidelberg, pp177–191
Diao YH, Li T, Zhao ZW (2013) Zinc accumulation characteristics of two Exophiala strains and their antioxidant response to Zn2+ stress. J Environ Prot 4:12–19
Duan J, Tian H, Drijber RA, Gao Y (2015) Systemic and local regulation of phosphate and nitrogen transporter genes by arbuscular mycorrhizal fungi in roots of winter wheat (Triticum aestivum L.). Plant Physiol Bioch 96:199–208. https://doi.org/10.1016/j.plaphy.2015.08.006
Fijan N (1969) Systemic mycosis in channel catfish. Bull Wildl Dis Assoc 5:109–110
Franken P (2012) The plant strengthening root endophyte Piriformospora indica: potential application and the biology behind. Appl Microbiol Biot 96:1455–1464. https://doi.org/10.1007/s00253-012-4506-1
Gaind S, Gaur A (1989) Effect of pH on phosphate solubilization by microbes. Curr Sci 58:1208–1211
Gaind S, Nain L (2015) Soil–phosphorus mobilization potential of phytate mineralizing fungi. J Plant Nutr 38:2159–2175. https://doi.org/10.1080/01904167.2015.1014561
García-López AM, Aviles M, Delgado A (2015) Plant uptake of phosphorus from sparingly available P- sources as affected by Trichoderma asperellum T34. Agr Food Sci 24:249–260. https://doi.org/10.23986/afsci.49532
Garg S, Bahl GS (2008) Phosphorus availability to maize as influenced by organic manures and fertilizer P associated phosphatase activity in soils. Bioresource Technol 99:5773–5777. https://doi.org/10.1016/j.biortech.2007.10.063
Ghosh P, Rathinasabapathi B, Ma LQ (2015) Phosphorus solubilization and plant growth enhancement by arsenic-resistant bacteria. Chemosphere 134:1–6. https://doi.org/10.1016/j.chemosphere.2015.03.048
Gómez-Guiñán Y (2004) Actividad de las fosfatasas ácidas y alcalinas (extracelulares e intracelulares) en hongos de la rizosfera de Arachis hypogaea (Papiloneaceae). Rev biol trop 52: 287-295 (in Spanish, with English abstract)
Handa Y, Kawaguchi M, Takeda N, Nishide H, Suzuki Y, Saito K (2015) RNA-seq transcriptional profiling of an arbuscular mycorrhiza provides insights into regulated and coordinated gene expression in Lotus japonicus and Rhizophagus irregularis. Plant Cell Physiol 56:1490–1511. https://doi.org/10.1093/pcp/pcv071
Harrison MJ, Buuren MLV (1995) A phosphate transporter from the mycorrhizal fungus Glomus versiforme. Nature 378:626–629. https://doi.org/10.1038/378626a0
Hiruma K, Gerlach N, Sacristán S, Nakano Ryohei T, Hacquard S, Kracher B, Neumann U, Ramírez D, Bucher M, O’Connell Richard J, Schulze-Lefert P (2016) Root endophyte Colletotrichum tofieldiae confers plant fitness benefits that are phosphate status dependent. Cell 165:464–474. https://doi.org/10.1016/j.cell.2016.02.028
Javot H, Pumplin N, Harrison MJ (2007) Phosphate in the arbuscular mycorrhizal symbiosis: transport properties and regulatory roles. Plant Cell Environ 30:310–322. https://doi.org/10.1111/j.1365-3040.2006.01617.x
Julou T, Burghardt B, Gebauer G, Berveiller D, Damesin C, Selosse M (2005) Mixotrophy in orchids: insights from a comparative study of green individuals and nonphotosynthetic individuals of Cephalanthera damasonium. New Phytol 166:639–653. https://doi.org/10.1111/j.1469-8137.2005.01364.x
Jumpponen A (2001) Dark septate endophytes—are they mycorrhizal? Mycorrhiza 11:207–211. https://doi.org/10.1007/s005720100112
Jungk A, Barber SA (1975) Plant age and the phosphorus uptake characteristics of trimmed and untrimmed corn root systems. Plant Soil 42:227–239. https://doi.org/10.1007/bf02186985
Kovar JL (2009) Methods of phosphorus analysis for soils, sediments, residuals, and waters, 2nd edn. In: JL Kovar (ed) Southern Cooperative Series Bulletin. SERA-IEG 17,Virginia Tech University, pp 408
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
Leyval C, Berthelin J (1989) Interactions between Laccaria laccata, Agrobacterium radiobacter and beech roots: influence on P, K, mg, and Fe mobilization from minerals and plant growth. Plant Soil 117:103–110. https://doi.org/10.1007/bf02206262
Li T, Liu MJ, Zhang XT, Zhang HB, Sha T, Zhao ZW (2011) Improved tolerance of maize (Zea mays L.) to heavy metals by colonization of a dark septate endophyte (DSE) Exophiala pisciphila. Sci Total Environ 409:1069–1074. https://doi.org/10.1016/j.scitotenv.2010.12.012
Li X, He C, He X, Su F, Hou L, Ren Y, Hou Y (2019) Dark septate endophytes improve the growth of host and non-host plants under drought stress through altered root development. Plant Soil 439:259–272. https://doi.org/10.1007/s11104-019-04057-2
Lin J, Wang Y, Sun S, Mu C, Yan X (2017) Effects of arbuscular mycorrhizal fungi on the growth, photosynthesis and photosynthetic pigments of Leymus chinensis seedlings under salt-alkali stress and nitrogen deposition. Sci Total Environ 576:234–241. https://doi.org/10.1016/j.scitotenv.2016.10.091
Lindström MS (2009) Emerging functions of ribosomal proteins in gene-specific transcription and translation. Biochem Bioph Res Co 379:167–170. https://doi.org/10.1016/j.bbrc.2008.12.083
Liu F, Xu Y, Jiang H, Jiang C, Du Y, Gong C, Wang W, Zhu S, Han G, Cheng B (2016) Systematic identification, evolution and expression analysis of the Zea mays PHT1 gene family reveals several new members involved in root colonization by arbuscular mycorrhizal fungi. Int J Mol Sci 17:930
López-Arredondo DL, Leyva-González MA, González-Morales SI, López-Bucio J, Herrera-Estrella L (2014) Phosphate nutrition: improving low-phosphate tolerance in crops. Annu Rev Plant Biol 65:95–123. https://doi.org/10.1146/annurev-arplant-050213-035949
Ma X-F, Wright E, Ge Y, Bell J, Xi Y, Bouton JH, Wang Z-Y (2009) Improving phosphorus acquisition of white clover (Trifolium repens L.) by transgenic expression of plant-derived phytase and acid phosphatase genes. Plant Sci 176:479–488. https://doi.org/10.1016/j.plantsci.2009.01.001
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–3793. https://doi.org/10.1093/bioinformatics/bti430
Marques D, Silva E, Silva D, Martins Ferreira M, Paglis C, Souza T, Maluf W, Maciel G, Dilelis Ferreira E, Klynger A, Lobato S (2016) Differential responses of root system and gas exchange in contrasting tomato genotypes under phosphorus starvation. Aust J Crop Sci 10:101–110
McGinnis MR, Ajello L (1974) A new species of Exophiala isolated from channel catfish. Mycologia 66:518–520
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular—arbuscular mycorrhizal fungi. New Phytol 115:495–501. https://doi.org/10.1111/j.1469-8137.1990.tb00476.x
Meena VS, Meena SK, Verma JP, Kumar A, Aeron A, Mishra PK, Bisht JK, Pattanayak A, Naveed M, Dotaniya ML (2017) Plant beneficial rhizospheric microorganism (PBRM) strategies to improve nutrients use efficiency: a review. Ecol Eng 107:8–32. https://doi.org/10.1016/j.ecoleng.2017.06.058
Nussaume L, Kanno S, Javot H, Marin E, Nakanishi TM, Thibaud M-C (2011) Phosphate import in plants: focus on the PHT1 transporters. Front Plant Sci 2:83. https://doi.org/10.3389/fpls.2011.00083
Osorio NW, Habte M (2015) Effect of a phosphate-solubilizing fungus and an arbuscular mycorrhizal fungus on leucaena seedlings in tropical soils with contrasting phosphate sorption capacity. Plant Soil 389:375–385. https://doi.org/10.1007/s11104-014-2357-5
Price G (2006) Australian soil fertility manual , 3rd edn. CSIRO publication, Australia
Rausch C, Daram P, Brunner S, Jansa J, Laloi M, Leggewie G, Amrhein N, Bucher M (2001) A phosphate transporter expressed in arbuscule-containing cells in potato. Nature 414:462–465. https://doi.org/10.1038/35106601
Richardson AE, Hadobas PA, Hayes JE (2000) Acid phosphomonoesterase and phytase activities of wheat (Triticum aestivum L.) roots and utilization of organic phosphorus substrates by seedlings grown in sterile culture. Plant Cell Environ 23:397–405. https://doi.org/10.1046/j.1365-3040.2000.00557.x
Rodríguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339. https://doi.org/10.1016/S0734-9750(99)00014-2
Ruiz-Lozano JM, Porcel R, Azcón C, Aroca R (2012) Regulation by arbuscular mycorrhizae of the integrated physiological response to salinity in plants: new challenges in physiological and molecular studies. J Exp Bot 63:4033–4044. https://doi.org/10.1093/jxb/ers126
Santos EA, Ferreira LR, Costa MD, Silva MCS, Reis MR, França AC (2013) Occurrence of symbiotic fungi and rhizospheric phosphate solubilization in weeds. Acta Sci, Agron 35:49–55
Smith SE, Manjarrez M, Stonor R, McNeill A, Smith FA (2015) Indigenous arbuscular mycorrhizal (AM) fungi contribute to wheat phosphate uptake in a semi-arid field environment, shown by tracking with radioactive phosphorus. Appl Soil Ecol 96:68–74. https://doi.org/10.1016/j.apsoil.2015.07.002
Spagnoletti FN, Tobar NE, Fernández Di Pardo A, Chiocchio VM, Lavado RS (2017) Dark septate endophytes present different potential to solubilize calcium, iron and aluminum phosphates. Appl Soil Ecol 111:25–32. https://doi.org/10.1016/j.apsoil.2016.11.010
Takai T, Kondo M, Yano M, Yamamoto T (2010) A quantitative trait locus for chlorophyll content and its association with leaf photosynthesis in rice. Rice 3:172–180. https://doi.org/10.1007/s12284-010-9047-6
Thuynsma R, Kleinert A, Kossmann J, Valentine AJ, Hills PN (2016) The effects of limiting phosphate on photosynthesis and growth of Lotus japonicus. S Afr J Bot 104:244–248. https://doi.org/10.1016/j.sajb.2016.03.001
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515. https://doi.org/10.1038/nbt.1621
Upson R, Read DJ, Newsham KK (2009) Nitrogen form influences the response of Deschampsia antarctica to dark septate root endophytes. Mycorrhiza 20:1–11. https://doi.org/10.1007/s00572-009-0260-3
Valli PPS, Muthukumar T (2018) Dark septate root endophytic fungus Nectria haematococca improves tomato growth under water limiting conditions. Indian J Microbiol 58:489–495. https://doi.org/10.1007/s12088-018-0749-6
Walder F, Brulé D, Koegel S, Wiemken A, Boller T, Courty P-E (2015) Plant phosphorus acquisition in a common mycorrhizal network: regulation of phosphate transporter genes of the Pht1 family in sorghum and flax. New Phytol 205:1632–1645. https://doi.org/10.1111/nph.13292
Wang J-l, Li T, G-y L, Smith JM, Z-w Z (2016) Unraveling the role of dark septate endophyte (DSE) colonizing maize (Zea mays) under cadmium stress: physiological, cytological and genic aspects. Sci Rep 6:22028. https://doi.org/10.1038/srep22028
Wang J, Lan P, Gao H, Zheng L, Li W, Schmidt W (2013) Expression changes of ribosomal proteins in phosphate- and iron-deficient Arabidopsis roots predict stress-specific alterations in ribosome composition. BMC Genomics 14:783. https://doi.org/10.1186/1471-2164-14-783
Wu M, Wei Q, Xu L, Li H, Oelmüller R, Zhang W (2018) Piriformospora indica enhances phosphorus absorption by stimulating acid phosphatase activities and organic acid accumulation in Brassica napus. Plant Soil 432:333–344. https://doi.org/10.1007/s11104-018-3795-2
Xie C, Li C-Y, Gao G, Huang J, Wu J, Kong L, Dong S, Mao X, Ding Y, Wei L (2011) KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res 39:W316–W322. https://doi.org/10.1093/nar/gkr483
Xue S, Barna M (2012) Specialized ribosomes: a new frontier in gene regulation and organismal biology. Nat Rev Mol Cell Bio 13:355–369. https://doi.org/10.1038/nrm3359
Yadav K, Dadarwal K (1997) Phosphate solubilization and mobilization through soil microorganisms. In: Dadarwal KR (ed) Biotechnological approaches in soil microorganisms for sustainable crop production. Scientific Publishers, Jodhpur, pp 293–308
Yadav RS, Tarafdar JC (2003) Phytase and phosphatase producing fungi in arid and semi-arid soils and their efficiency in hydrolyzing different organic P compounds. Soil Biol Biochem 35:745–751. https://doi.org/10.1016/S0038-0717(03)00089-0
Yadav V, Kumar M, Deep DK, Kumar H, Sharma R, Tripathi T, Tuteja N, Saxena AK, Johri AK (2010) A phosphate transporter from the root endophytic fungus Piriformospora indica plays a role in phosphate transport to the host plant. J Biol Chem 285:26532–26544. https://doi.org/10.1074/jbc.M110.111021
Yakti W, Kovács GM, Vági P, Franken P (2019) Impact of dark septate endophytes on tomato growth and nutrient uptake. Plant Ecol Divers 11:637–648. https://doi.org/10.1080/17550874.2019.1610912
Yan H, Zong C, Guo-Hua M, Shan S (2011) Relationship between ribosomal protein L41 and resistance of Aspergillus flavus. Acta Agric Boreali-Sinica 26: 16-19 (in Chinese, with English abstract)
Yang CW, Wang P, Li CY, Shi DC, Wang DL (2008) Comparison of effects of salt and alkali stresses on the growth and photosynthesis of wheat. Photosynthetica 46:107–114. https://doi.org/10.1007/s11099-008-0018-8
Yu T, Nassuth A, Peterson RL (2001) Characterization of the interaction between the dark septate fungus Phialocephala fortinii and Asparagus officinalis roots. Can J Microbiol 47:741–753. https://doi.org/10.1139/w01-065
Zhan F, He Y, Zu Y, Li T, Zhao Z (2011) Characterization of melanin isolated from a dark septate endophyte (DSE), Exophiala pisciphila. World J Microbiol Biotechnol 27:2483–2489
Zhan F, Li B, Jiang M, Qin L, Wang J, He Y, Li Y (2017) Effects of a root-colonized dark septate endophyte on the glutathione metabolism in maize plants under cadmium stress. J Plant Interact 12:421–428. https://doi.org/10.1080/17429145.2017.1385868
Zhang L, Xu M, Liu Y, Zhang F, Hodge A, Feng G (2016) Carbon and phosphorus exchange may enable cooperation between an arbuscular mycorrhizal fungus and a phosphate-solubilizing bacterium. New Phytol 210:1022–1032. https://doi.org/10.1111/nph.13838
Zhang Y, Zhang Y, Liu M, Shi X, Zhao Z (2008) Dark septate endophyte (DSE) fungi isolated from metal polluted soils: their taxonomic position, tolerance, and accumulation of heavy metals In Vitro. J Microbiol 46:624–632. https://doi.org/10.1007/s12275-008-0163-6
Zhu L, Li T, Wang C, Zhang X, Xu L, Xu R, Zhao Z (2018) The effects of dark septate endophyte (DSE) inoculation on tomato seedlings under Zn and cd stress. Environ Sci Pollut R 25:35232–35241. https://doi.org/10.1007/s11356-018-3456-2
Acknowledgments
This research was financially supported by the Yunnan Provincial Science and Technology Department-Yunnan University Joint Fund, the key project (2018FY001-010), “Double First-Class” University Project of Yunnan University (C176282105) and the National Natural Science Foundation of China (31460114 and 41461073).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Felipe E. Albornoz.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. S1
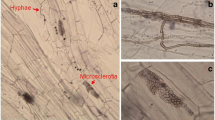
Morphological characteristics of DSE colonizing the roots of maize inoculated with E. pisciphila in the pot culture and compartmentalized microcosm (hypha is showed by dashed arrow while microsclerotium is pointed by solid arrow; bar = 50 μm). (PNG 5915 kb)
Fig. S2
Venn diagram of differentially expressed genes in maize root in response to different treatments with Pi and DSE in pot culture (+, upregulated; −, downregulated; + +, upregulated in both groups; + −, upregulated in the left group and downregulated in the right group; − +, downregulated in the left group and upregulated in the right group; − −, downregulated in both groups.) (PNG 190 kb)
Fig. S3
Significantly enriched pathways in maize roots related to up/downregulated DEGs responsible for Pi deficiency and DSE inoculation in pot culture ([3.1.3.12] trehalose 6-phosphate synthase/phosphatase; [3.2.1.21] beta-glucosidase; [2.7.1.1] hexokinase; [2.7.1.11] 6-phosphofructokinase 1; [4.1.2.13] fructose-bisphosphate aldolase, class I; [5.3.1.1] triosephosphate isomerase (TIM); [2.7.1.40] pyruvate kinase; [4.1.1.1] pyruvate decarboxylase; [1.1.1.27] L-lactate dehydrogenase; [1.1.1.1] S-(hydroxymethyl) glutathione dehydrogenase / alcohol dehydrogenase; [1.4.1.13] glutamate synthase 2 (NADH); [1.4.1.14] glutamate synthase (NADH); [6.3.1.2] glutamine synthetase; [1.13.11.19] cysteamine dioxygenase; [4.1.1.15] glutamate decarboxylase. Colors from dark blue to dark red indicate gene expression from low to high.) (PNG 1078 kb)
Fig. S4
Transcriptional changes in ZmPHT1 genes responsible for Pi deficiency and DSE inoculation in maize roots in pot culture (ZmPHT1;10 was not detected in the present study. Colors from dark blue to dark red indicate gene expression from low to high.) (PNG 87 kb)
Table S1 & S2
Table S1. Inhibition rate (IR, %) of Pi deficiency on parameters of maize growth in pot culture and promotion rate (PR, %) of DSE on the maize tested parameters in pot culture and compartmentalized microcosm. Table S2. DSE biomass and soluble P, phosphatase activity and pH in the fermentation broth (mean ± SE, n = 3) treated with different P source for 10 days. (DOCX 25 kb)
Rights and permissions
About this article
Cite this article
Xu, R., Li, T., Shen, M. et al. Evidence for a Dark Septate Endophyte (Exophiala Pisciphila, H93) Enhancing Phosphorus Absorption by Maize Seedlings. Plant Soil 452, 249–266 (2020). https://doi.org/10.1007/s11104-020-04538-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04538-9