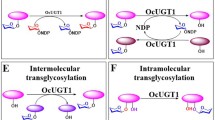
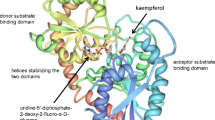
Abstract
Flavonoid glycosides are required for a number of crucial roles in planta and have the potential for development in a variety of agricultural, medicinal, and biotechnological applications. A number of recent advancements have been made in characterizing glycosyltransferases, the enzymes that are responsible for the synthesis of these important molecules. In this review, glycosyltransferases are considered with regard to biochemical properties, expression patterns, levels of enzyme activity during development, and structure/function relationships. This is presented with historical context to highlight critical findings, particularly with regard to the innovative work that has come from research on citrus species. The plant glycosyltransferase crystal structures that have been solved over the past decade, either alone or in complex with sugar donor and/or acceptor molecules, are discussed. The application of results from these structures to inform current structure/function work as well as implications and goals for future crystallography and tertiary modeling studies are considered. A thorough understanding of the properties of glycosyltransferases will be a critical step in any future biotechnological application of these enzymes in areas such as crop improvement and custom design of enzymes to produce desired compounds for nutritional and/or medicinal usage.




Similar content being viewed by others
Notes
To simplify identifying residues and areas of interest throughout this discussion, locations will be given in relation to the lower case letters indicated in Fig. 4 (alignment).
Abbreviations
- ACT:
-
Acetate
- B3P:
-
Bis–tris propane
- Cp3GT:
-
Citrus paradisi flavonol 3-O-glucosyltransferse
- DLM:
-
Delphinidin
- EDO:
-
Ethylene glycol
- Gal:
-
Galactose
- Glc:
-
Glucose
- GlcNAC:
-
N-Acetylglucosamine
- GOL:
-
Glycerol
- GT:
-
Glycosyltransferase
- KMP:
-
Kaempferol
- Man:
-
Mannose
- MYC:
-
Myricetin
- P5M:
-
Petunidin
- PSPG:
-
Plant secondary product glycosyltransferase
- QUE:
-
Quercetin
- Rhm:
-
Rhamnose
- RT:
-
Rhamnosyltransferase
- TC7:
-
Trichlorophenol
- TRS:
-
Tris buffer
- U2F:
-
Uridine-5′-diphosphate-2-deoxy-2-fluoro
- UDP:
-
Uridine-5′-diphosphate
- UDPG:
-
Uridine-5′-diphosphate glucose
- Xyl:
-
Xylose
References
Alam MA, Subhan N, Rahman MM, Uddin SJ, Reza HM, Sarker SD (2014) Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanism of action. Adv Nutr 5:404–417
Bar-Peled M, Lewinsohn E, Fluhr R, Gressel J (1991) UDP-rhamnose:flavanone-7-O-glucoside-2″-O-rhamnosyltransferase: purification and characterization of an enzyme catalyzing the production of bitter compounds in citrus. J Biol Chem 266:20953–20959
Bar-Peled M, Fluhr R, Gressel J (1993) Juvenile-specific localization and accumulation of a rhamnosyltransferase and its bitter flavonoid in foliage, flowers, and young citrus fruits. Plant Physiol 103:1377–1384
Barthe GA, Jourdan PS, McIntosh CA, Mansell RL (1987) Naringin and limonin production in callus cultures and regenerated plants from Citrus sp. J Plant Physiol 127:55–65
Barthe GA, Jourdan PS, McIntosh CA, Mansell RL (1988) Radioimmunoassay for the quantitative determination of hesperidin in Citrus sinensis. Phytochemistry 27:249–254
Benavente-Garcia O, Castillo J (2008) Update on uses and properties of citrus flavonoids: new findings in anticancer, cardiovascular, and anti-inflammatory activity. J Agric Food Chem 56(15):6185–6205
Benavente-García O, Castillo J, Marin FR, Ortuño A, Del Río JA (1997) Uses and properties of citrus flavonoids. J Agric Food Chem 45(12):4505–4515
Berhow M, Smolensky D (1995) Developmental and substrate specificity of hesperitin-7-O-clucosyltransferase activity in Citrus limon tissues using high performance liquid chromatography analysis. Plant Sci 112:139–147
Berhow MA, Vandercook CE (1989) Biosynthesis of naringin and prunin in detached grapefruit. Phytochemistry 28:627–1630
Berhow M, Tisserat B, Kanes K, Vandercook C (1998) Survey of phenolic compounds produced in citrus. USDA ARS Technical Bull #158
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) The protein data bank. Nucl Acids Res 28:235-242
Brazier-Hicks M, Offen WA, Gershater MC, Revett TJ, Lim EK, Bowles DJ, Davies G, Edwards R (2007) Characterization and engineering of the bifunctional N- and O-glucosyltransferase involved in xenobiotic metabolism in plants. PNAS 104(51):20238–20243
Breton C, Fournel-Gigleux S, Palcic MM (2012) Recent structures, evolution and mechanisms of glycosyltransferases. Curr Opin Struct Biol 22(5):540–549
Cheng J, Wei G, Zhou H, Gu C, Vimolmangkang S, Liao L, Han Y (2014) Unraveling the mechanism underlying the glycosylation and methylation of anthocyanins in peach. Plant Physiol 166:1044–1058
Cody V, Middleton E Jr, Harborne JB (1986) Plant flavonoids in biology and medicine: biochemical, pharmacological, and structure–activity relationships. In: Cody V, Middleton E Jr, Harborne JB (eds) Progress in clinical and biological research, vol 213. Alan R. Liss, Inc, New York
Craig WJ (1997) Phytochemicals: guardians of our health. J Am Diet Assoc 97(10 Suppl 2):S199–S204
Daniel JJ, Owens DK, McIntosh CA (2011) Secondary product glucosyltransferase and putative glucosyltransferase expression during Citrus paradisi (c.v.Duncan) growth and development. Phytochemistry 72:1732–1738
Demonty I, Lin Y, Zebregs YE, Vermeer MA, van der Knaap HC, Jakel M, Trautwein EA (2010) The citrus flavonoids hesperidin and naringin do not affect serum cholesterol in moderately hypercholesterolemic men and women. J Nutr 140(9):1615–1620
Devaiah SP, Owens DK, Sibhatu MB, Roy Sarkar T, Strong CL, Mallampalli VKPS, Asiago J, Cooke J, Kaiser S, Lin Z, Wamucho A, Hayford D, Williams BE, Loftis P, Berhow M, Pike LM, McIntosh CA (2016) Isolation, recombinant expression, and biochemical characterization of putative secondary product glucosyltransferases from Citrus paradisi. J Agric Food Chem. doi:10.1021/acs.jafc.5b05430
Fisher JF (1968) A procedure for obtaining radioactive naringin from grapefruit leaves fed l-phenylalanine (14C). Phytochemistry 7:769–771
Ford CM, Boss PK, Høj PB (1998) Cloning and characterization of Vitis vinifera UDP-glucose: flavonoid 3-O-glucosyltransferase, a homologue of the enzyme encoded by the maize bronze-1 locus that may primarily serve to glucosylate anthocyanidins in vivo. J Biol Chem 273(15):9224–9233
Frydman A, Weisshaus O, Bar-Peled M, Huhman D, Summer L, Marin F, Lewinsohn E, Fluhr R, Gressel J, Eyal Y (2004) Citrus bitter flavors: isolation and functional characterization of the gene Cm1,2RhaT encoding a 1,2-rhamnosyltransferase, a key enzyme in the biosynthesis of the bitter flavonoids in citrus. Plant J 40:88–100
Frydman A, Liberman R, Huhman DV, Carmeli-Weissberg M, Sapir-Mir M, Ophir R, Sumner L, Eyal Y (2013) The molecular and enzymatic basis of bitter/non-bitter flavor of citrus fruit: evolution of branch-forming rhamnosyltransferases under domestication. Plant J 73:166–178
Gachon CM, Langlois-Meurinne M, Saindrenan P (2005) Plant secondary metabolism glycosyltransferases: the emerging functional analysis. Trends Plant Sci 10(11):542–549
Gattuso G, Barreca D, Gargiulli C, Leuzzi U, Caristi C (2007) Flavonoid composition of citrus juices. Molecules 12:1641–1673
Gentili B, Horowitz RM (1968) Flavonoids of citrus. IX. Some new C-glycosylflavones and a nuclear magnetic resonance method for differentiating 6- and 8-C-glycosyl isomers. J Org Chem 33:1571–1577
Gloster TM (2014) Advances in understanding glycosyltransferases from a structural perspective. Curr Opin Struct Biol 28:131–141
Hendrickson R, Kesterson W (1964) Hesperidin in Florida oranges. Univ Fla Exp Stn Bull #684
Hiromoto T, Honjo E, Tamada T, Noda N, Kazuma K, Suzuki M, Kuroki R (2013) Crystal structure of UDP-glucose: anthocyanidin 3-O-glucosyltransferase from Clitoria ternatea. J Synchrotron Radiat 20(6):894–898
Hiromoto T, Honjo E, Noda N, Tamada T, Kazuma K, Suzuki M, Blaber M, Kuroki R (2015) Structural basis for acceptor-substrate recognition of UDP-glucose: anthocyanidin 3-O-glucosyltransferase from Clitoria ternatea. Protein Sci 24(3):395–407
Hughes J, Hughes MA (1994) Multiple secondary plant product UDP-glucose glucosyltransferase genes expressed in cassava (Manihot esculenta Crantz) cotyledons. DNA Seq 5(1):41–49
Iwashina T (2003) Flavonoid function and activity to plants and other organisms. Biol Sci Space 17(1):24–44
Jackson R, Knisley D, McIntosh C, Pfeiffer P (2011) Predicting flavonoid UGT regioselectivity. Adv Bioinform. doi:10.1155/2011/506583 (Article ID 506583)
Jourdan PS, Mansell RL (1982) Isolation and partial characterization of three glucosyltransferases involved in the biosynthesis of flavonol triglucosides in Pisum sativum L. Arch Biochem Biophys 213:434–443
Jourdan PD, McIntosh CA, Mansell RL (1985) Naringin levels in citrus tissues: II. Quantitative distribution of naringin in Citrus paradisi Macfad. Plant Physiol 77:903–908
Jung UJ, Kim HJ, Lee JS, Kim HO, Park EJ, Kim HK, Jeong TS, Choi MS (2003) Naringin supplementation lowers plasma lipids and enhances erythrocyte antioxidant enzyme activities in hypercholesterolemic subjects. Clin Nutr 22(6):561–568
Kleinehollenhorst GH, Behrens H, Pegels G, Srunk N, Wiermann R (1982) Formation of flavonol 3-O-diglycosides and flavonol 3-O-triglucosides by enzyme extracts from anthers of Tulipa cv. Apeldoorn: characterization and activity of three different O-glucosyltransferases during anther development. Z Naturforsch 37:587–599
Knisley D, Seier E, Lamb D, Owens D, McIntosh C (2009) A graph-theoretic model based on primary and predicted secondary structure reveals functional specificity in a set of plant secondary product UDP-glucosyltransferases. In: Loging W, et al. (eds) Proceedings of the 2009 international conference on bioinformatics, computational biology, genomics and chemoinformatics, (BCBGC-09), pp 65–72, ISRST
Kubo A, Arai Y, Nagashima S, Yoshikawa T (2004) Alteration of sugar donor specificities of plant glycosyltransferases by a single point mutation. Arch Biochem Biophys 429(2):198–203
Lairson LL, Henrissat B, Davies GJ, Withers SG (2008) Glycosyltransferases: structures, functions, and mechanisms. Annu Rev Biochem 77(1):521–555
Lewinsohn E, Berman E, Mazur Y, Gressel J (1986) Glucosylation of exogenous flavanones by grapefruit (Citrus paradisi) cell cultures. Phytochemistry 25:2531–2535
Lewinsohn E, Berman E, Mazur Y, Gressel J (1989a) 7-Glucosylation and (1–6) rhamnosylation of exogenous flavanones by undifferentiated citrus cell cultures. Plant Sci 61:23–28
Lewinsohn E, Britsch EL, Mazur Y, Gressel J (1989b) Flavanone glycoside biosynthesis in citrus: chalcone synthase, UDP-glucose:flavanone-7-O-glucosyltransferase and -rhamnosyltransferase activities in cell free extracts. Plant Physiol 91:1323–1328
Li L, Modolo LV, Escamilla-Trevino LL, Achnine L, Dixon RA, Wang X (2007) Crystal structure of Medicago truncatula UGT85H2—insights into the structural basis of a multifunctional (iso) flavonoid glycosyltransferase. J Mol Biol 370(5):951–963
Liang DM, Liu JH, Wu H, Wang BB, Zhu HJ, Qiao JJ (2015) Glycosyltransferases: mechanisms and applications in natural product development. Chem Soc Rev 44(22):8350–8374
Lombard V, Golaconda Ramulu H, Drula E, Coutinho PM, Henrissat B (2014) The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Res 42:D490–D495
Mackenzie PI, Owens IS, Burchell B, Bock KW, Bairoch A, Belanger A, Gigleux SF, Green M, Hum DW, Iyanagi T, Lancet D, Louisot P, Magdalou J, Roy Chowdhury J, Ritter Joseph K, Tephly TR, Schachter H, Tephly T, Tipton KF, Nebert DW (1997) The UDP glycosyltransferase gene superfamily: recommended nomenclature update based on evolutionary divergence. Pharmacogenetics 7(4):255–269
Mallampalli VKPS (2009) Expression and biochemical function of putative flavonoid GT clones from grapefruit and identification of new clones using the harvEST database. ETSU electronic theses and dissertations. http://dc.etsu.edu/etd/1788
Mansell RL, McIntosh CA (1991) Citrus spp.: in vitro culture and the production of naringin and limonin. In: Bajaj YPS (ed) Biotechnology of medicinal and aromatic plants, vol 3. Springer, Berlin, pp 193–210
Mansell RL, Vest SE, McIntosh CA (1983) An analysis of the limonin and naringin content of grapefruit juice samples collected from Florida state test houses. J Agric Food Chem 31:156–162
McIntosh CA (2000) Quantification of limonin and limonoate A-ring monolactone during growth and development of citrus fruit and vegetative tissues by radioimmunoassay. In: Berhow MA, Hasegawa S, Manners GD (eds) Citrus limonoids: functional chemicals in agriculture and food, Chapter 6. ACS Symposium Series #758, ACS, Washington, pp 73–95
McIntosh CA, Mansell RL (1990) Biosynthesis of naringin in Citrus paradisi: UDP-glucosylltransferase activity in grapefruit seedlings. Phytochemistry 29:1533–1538
McIntosh CA, Latchinian L, Mansell RL (1990) Flavanone-specific O-7-glucosyltransferase activity in Citrus paradisi seedlings: purification and characterization. Arch Biochem Biophys 282:50–57
Modolo LV, Li L, Pan H, Blount JW, Dixon RA, Wang X (2009) Crystal structures of glycosyltransferase UGT78G1 reveal the molecular basis for glycosylation and deglycosylation of (iso) flavonoids. J Mol Biol 392(5):1292–1302
Offen W, Martinez-Fleites C, Yang M, Kiat-Lim E, Davis BG, Tarling CA, Ford CM, Bowles DJ, Davies GJ (2006) Structure of a flavonoid glucosyltransferase reveals the basis for plant natural product modification. EMBO 25(6):1396–1405
Ohashi T, Hasegawa Y, Misaki R, Fujiyama K (2015) Substrate preference of citrus naringenin rhamnosyltransferases and their application to flavonoid glycoside production in fission yeast. Appl Microbiol Biotechnol. doi:10.1007/s00253-015-6982-6
Owens DK, McIntosh CA (2009) Identification, recombinant expression, and biochemical characterization of a flavonol 3-O-glucosyltransferase clone from Citrus paradisi. Phytochemistry 70:1382–1391
Owens DK, McIntosh CA (2011) Biosynthesis and function of citrus glycosylated flavonoids. In: Gang DR (ed) Chapter 6 in Rec Adv Phytochem vol 41, “The biological activity of phytochemicals. Springer, New York, pp 67–96
Paine MF, Widmer WW, Hart HL, Pusek SN, Beavers KL, Criss AB, Brown SS, Thomas BF, Watkins PB (2006) A furanocoumarin-free grapefruit juice establishes furanocoumarins as the mediators of the grapefruit juice–felodipine interaction. Am J Clin Nutr 83(5):1097–1105
Peterson JJ, Dwyer JT, Beecher GR, Bhagwat SA, Gebhardt SE, Haytowitz DB, Holden JM (2006) Flavanones in oranges, tangerines (mandarins), tangors, and tangelos: a compilation and review of the data from the analytical literature. J Food Compos Anal 19:S66–S73
Rizza S, Muniyappa R, Iantorno M, Kim J, Chen H, Pullikotil P, Senese N, Tesauro M, Lauro D, Cardillo C, Quon MJ (2011) Citrus polyphenol hesperidin stimulates production of nitric oxide in endothelial cells while improving endothelial function and reducing inflammatory markers in patients with metabolic syndrome. J Clin Endocrinol Metab 96:E782-E792. doi:10.1210/jc.2010-2879
Rouseff RL (1980) Flavonoids and citrus quality. In: Nagy S, Attaway JA (eds) Chapter 5 in citrus nutrition and quality. ACS symposium series #143, ACS. Washington, pp 83–108
Shao H, He X, Achnine L, Blount JW, Dixon RA, Wang X (2005) Crystal structures of a multifunctional triterpene/flavonoid glycosyltransferase from Medicago truncatula. Plant Cell 17(11):3141–3154
Sigrist CJA, de Castro E, Cerutti L, Cuche BA, Hulo N, Bridge A, Bougueleret L, Xenarios I (2012) New and continuing developments at PROSITE. Nucleic Acids Res. doi:10.1093/nar/gks1067
Stoclet JC, Schini-Kerth V (2011) Dietary flavonoids and human health. Ann Pharm Fr 69(2):78–90
Toh JY, Tan VM, Lim PC, Lim ST, Chong MF (2013) Flavonoids from fruit and vegetables: a focus on cardiovascular risk factors. Curr Atheroscler Rep 15(12):1–7
Wang X (2009) Structure, mechanism and engineering of plant natural product glycosyltransferases. FEBS Lett 583(20):3303–3309
Watson RR, Preedy VR, Zibadi S (eds) (2014) Polyphenols in human health and disease. Academic Press, USA
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126(2):485–493
Yamane K, Kato Y (eds) (2012) Handbook of flavonoids: dietary sources, properties and health benefits. Nova, New York
Acknowledgments
The authors would like to acknowledge financial support from the United States Department of Agriculture, the National Science Foundation, and East Tennessee State University for the work cited from the McIntosh lab. We also appreciate the undergraduate students, graduate students, and postdocs who have engaged in the research, most of whom are also coauthors in references cited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McIntosh, C.A., Owens, D.K. Advances in flavonoid glycosyltransferase research: integrating recent findings with long-term citrus studies. Phytochem Rev 15, 1075–1091 (2016). https://doi.org/10.1007/s11101-016-9460-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-016-9460-6