Abstract
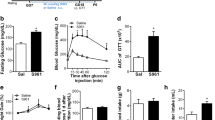
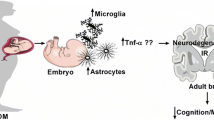
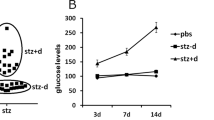
Gestational diabetes (GD) is the glucose intolerance that occurs during pregnancy. Mothers who develop diabetes during gestation are at increased risk of developing type 2 diabetes mellitus (T2DM) later in life, and the risk of adverse fetal and neonatal outcomes are also increased as a function of maternal hyperglycemia. Infants who are exposed to fetal hyperglycemia show an increased risk of becoming obese and developing T2DM later in life. Due to the need of new research on this field, and the difficulty of performing studies in human brain, studies using experimental models are necessary to suggest possible ways to avoid or inhibit offspring brain damage or harmful metabolic alterations. Here, it was made a review about the characteristics of the main animal models of GD, and what are the consequences to the brain and behavior of the offspring. In many experimental models, either by pharmacological induction, diet manipulation, or in the use of transgenic animals, glycemic conditions are severe. S961, a selective insulin receptor antagonist, revealed an increased fasting blood glucose level and glucose intolerance during mid-gestation, which returned to basal levels postpartum in mice. GD contributes to offspring neuroinflammation, influences neuronal distribution in central nervous system (CNS), and apoptosis during embryogenesis, which in turn may contribute to changes in behavior and memory in adult life and aging. The usage of animal models to study GD allows to examine extensively the characteristics of this condition, the molecular mechanisms involved and the consequences to the brain and behavior of the offspring.


Similar content being viewed by others
References
Agha-Jaffar R, Oliver N, Johnston D, Robinson S (2016) Gestational diabetes mellitus: does an effective prevention strategy exist? Nat Rev Endocrinol 12:533–546. https://doi.org/10.1038/nrendo.2016.88
American Diabetes Association (ADA) (2014) Diagnosis and classification of diabetes mellitus. Diabetes Care 37(Suppl 1):S81–S90. https://doi.org/10.2337/dc14-S081
Bergman EN, Havel RJ, Wolfe BM, Bohmer T (1971) Quantitative studies of the metabolism of chylomicron triglycerides and cholesterol by liver and extrahepatic tissues of sheep and dogs. J Clin Invest 50:1831–1839. https://doi.org/10.1172/JCI106674
Buchanan TA, Xiang AH, Page KA (2012) Gestational diabetes mellitus: risks and management during and after pregnancy. Nat Rev Endocrinol 8:639–649. https://doi.org/10.1038/nrendo.2012.96
Carlson A, Drennan F (1911) The control of pancreatic diabetes in pregnancy by the passage of the internal secretion of the pancreas of the fetus to the blood of the mother. Am J Physiol 28:391–395. https://doi.org/10.1017/S0029665113001286
Chandna AR, Kuhlmann N, Bryce CA et al (2015) Chronic maternal hyperglycemia induced during mid-pregnancy in rats increases RAGE expression, augments hippocampal excitability, and alters behavior of the offspring. Neuroscience 303:241–260. https://doi.org/10.1016/j.neuroscience.2015.06.063
Chen H, Charlat O, Tartaglia LA et al (1996) Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell 84:491–495. https://doi.org/10.1016/S0092-8674(00)81294-5
Cuthbert FP, Ivy AC, Isaacs BL, Gray J (1936) The relation of pregnancy and lactation to extirpation diabetes in the dog. Am Physiol Soc 115:480–496
De Sousa RAL, Torres YS, Figueiredo CPCP et al (2017) Consequences of gestational diabetes to the brain and behavior of the offspring. An Acad Bras Cienc 90:2279–2291. https://doi.org/10.1590/0001-3765201720170264
de Sousa RAL (2018a) Gestational diabetes is associated to the development of brain insulin resistance in the offspring. Int J Diabetes Dev Ctries 39:408–416. https://doi.org/10.1007/s13410-018-0618-1
de Sousa RAL (2018b) Brief report of the effects of the aerobic , resistance , and high-intensity interval training in type 2 diabetes mellitus individuals Diabetes mellitus. Int J Diabetes Dev Ctries 38:138–145. https://doi.org/10.1007/s13410-017-0582-1
De Sousa RAL, de Lima EV, da Silva TP et al (2019) Late cognitive consequences of gestational diabetes to the offspring, in a new mouse model. Mol Neurobiol 56:1–11. https://doi.org/10.1007/s12035-019-1624-0
De Sousa RAL, Azevedo LM, Improta-Caria A et al (2020a) Type 2 diabetes individuals improve C-reactive protein levels after high-intensity weight lift training. Sci Sport:1–7. https://doi.org/10.1016/j.scispo.2020.05.008
De Sousa RAL, Caria ACI, De Jesus Silva FM et al (2020b) High-intensity resistance training induces changes in cognitive function, but not in locomotor activity or anxious behavior in rats induced to type 2 diabetes. Physiol Behav 223:1–7. https://doi.org/10.1016/j.physbeh.2020.112998
De Sousa RAL, Hagenbeck KF, Arsa G, Pardono E (2020c) Moderate / high resistance exercise is better to reduce blood glucose and blood pressure in middle-aged diabetic subjects. Rev Bras Educ Física e Esporte 34:165–175
De Sousa RAL, Harmer AR, Freitas DA et al (2020d) An update on potential links between type 2 diabetes mellitus and Alzheimer’s disease. Mol Biol Rep. https://doi.org/10.1007/s11033-020-05693-z
De Sousa RAL, Peixoto MFD, Leite HR et al (2020e) Neurological consequences of exercise during prenatal Zika virus exposure to mice pups Ricardo. Int J Neurosci. https://doi.org/10.1080/00207454.2020.1860970
De Sousa RAL, Rodrigues CM, Mendes BF et al (2020f) Physical exercise protocols in animal models of Alzheimer ’ s disease : a systematic review. Metab Brain Dis:1–11. https://doi.org/10.1007/s11011-020-00633-z
Desisto CL, Kim SY, Sharma AJ (2014) Prevalence estimates of gestational diabetes mellitus in the United States, Pregnancy Risk Assessment Monitoring System ( PRAMS ), 2007–2010. Prev Chronic Dis 11:1–9
Eclarinal JD, Zhu S, Baker MS et al (2016) Maternal exercise during pregnancy promotes physical activity in adult offspring. FASEB J 30:2541–2548. https://doi.org/10.1096/fj.201500018R
El Hajj N, Schneider E, Lehnen H, Haaf T (2014) Epigenetics and life-long consequences of an adverse nutritional and diabetic intrauterine environment. Reproduction 148:R111–R120. https://doi.org/10.1530/REP-14-0334
Fall T, Hedhammar A, Wallberg A et al (2010) Diabetes mellitus in elkhounds is associated with Diestrus and pregnancy. J Vet Int Med 65:1322–1328
Frías JL, Frías JP, Frías PA, Frías MLM (2007) Infrequently studied congenital anomalies as clues to the diagnosis of maternal diabetes mellitus. Am J Med Genet Part A 2909:2904–2909. https://doi.org/10.1002/ajmg.a
Fu J, Tay SSW, Ling EA, Dheen ST (2006) High glucose alters the expression of genes involved in proliferation and cell-fate specification of embryonic neural stem cells. Diabetologia 49:1027–1038. https://doi.org/10.1007/s00125-006-0153-3
Golalipour MJ, Kafshgiri SK, Ghafari S (2012) Gestational diabetes induced neuronal loss in CA1 and CA3 subfields of rat hippocampus in early postnatal life. Folia Morphol (Warsz) 71:71–77
Huot R, Gonzalez M, Ladd C et al (2004) Foster litters prevent hypothalamic-pituitary-adrenal axis sensitization mediated by neonatal maternal separation. Psychoneuroendocrinology 29:279–289. https://doi.org/10.1016/S0306-4530(03)00028-3
Improta-Caria AC, Nonaka CKV, Cavalcante BRR et al (2020) Modulation of microRNAs as a potential molecular mechanism involved in the beneficial actions of physical exercise in Alzheimer disease. Int J Mol Sci 21:1–35. https://doi.org/10.3390/ijms21144977
Kaluski S, Portillo M, Besnard A et al (2017) Neuroprotective functions for the histone report neuroprotective functions for the histone deacetylase SIRT6. CellReports 18:3052–3062. https://doi.org/10.1016/j.celrep.2017.03.008
Knudsen L, Hansen BF, Jensen P et al (2012) Agonism and antagonism at the insulin receptor. PLoS One 7. https://doi.org/10.1371/journal.pone.0051972
Kobayashi K, Kobayashi N, Okitsu T et al (2004) Development of a porcine model of type 1 diabetes by total pancreatectomy and establishment of a glucose tolerance evaluation method. Artif Organs 28:1035–1042. https://doi.org/10.1111/j.1525-1594.2004.00002.x
Liao DM, Ng YK, Tay SSW et al (2004) Altered gene expression with abnormal patterning of the telencephalon in embryos of diabetic Albino Swiss mice. Diabetologia 47:523–531. https://doi.org/10.1007/s00125-004-1351-5
Liu S, Guo Y, Yuan Q et al (2015) Melatonin prevents neural tube defects in the offspring of diabetic pregnancy. J Pineal Res:508–517. https://doi.org/10.1111/jpi.12282
Lourenco MV, Frozza RL, de Freitas GB et al (2019) Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat Med:165–175. https://doi.org/10.1038/s41591-018-0275-4
Markowitz J, Soskin S (1927) Pancreatic diabetes and pregnancy. Am J Phys 11:553–558
McCurdy CE, Bishop JM, Williams SM et al (2009) Maternal high-fat diet triggers lipotoxicity in the fetal livers of nonhuman primates. J Clin Invest 119:323–335. https://doi.org/10.1172/JCI32661
Mills JL, Baker L, Goldman AS (1979) Malformations in infants of diabetic mothers occur before the seventh gestational week implications for treatment. Diabetes 28:292–293
Moussaoui N, Larauche M, Biraud M et al (2016) Limited nesting stress alters maternal behavior and in vivo intestinal permeability in male wistar pup rats. PLoS One:1–14. https://doi.org/10.1371/journal.pone.0155037
Murata M, Takahashi A, Saito I, Kawanishi S (1999) Site-specific DNA methylation and apoptosis: induction by diabetogenic streptozotocin. Biochem Pharmacol 57:881–887. https://doi.org/10.1016/S0006-2952(98)00370-0
Neves FS, Marques PT, Aragão FB et al (2016) Brain-defective insulin signaling is associated to late cognitive impairment in post-septic mice. Mol Neurobiol:1–10. https://doi.org/10.1007/s12035-016-0307-3
Plyte SE, Hughes K, Nikolakaki E et al (1992) Glycogen synthase kinase-3 : functions in oncogenesis and development. Biochim Biophys Acta 1114:147–162
Reis AR, de Azevedo MS, de Souza MA et al (2014) Neonatal handling alters the structure of maternal behavior and affects mother-pup bonding. Behav Brain Res 265:216–228. https://doi.org/10.1016/j.bbr.2014.02.036
Schäffer L, Brand CL, Hansen BF et al (2008) A novel high-affinity peptide antagonist to the insulin receptor. Biochem Biophys Res Commun 376:380–383. https://doi.org/10.1016/j.bbrc.2008.08.151
Sharpe PB, Chan A, Haan EA, Hiller JE (2005) Maternal diabetes and congenital anomalies in South Australia 1986–2000: a population-based cohort study. Birth Defects Res A Clin Mol Teratol 73:605–611. https://doi.org/10.1002/bdra.20172
Silverman BL, Metzger BE, Cho NH, Loeb CA (1995) Impaired glucose tolerance in adolescent offspring of diabetic mothers. Diabetes Care 18:611–617
Sousa RAL, Freitas DA, Leite HR (2019) Cross-talk between obesity and central nervous system: role in cognitive function. Interv Obes Diabetes 3:7–9. https://doi.org/10.31031/IOD.2019.03.000551
Szkudelski T (2001) The mechanism of Alloxan and Streptozotocin action in B cells of the rat pancreas. Physiol Res 50:536–546
Vikram A, Jena G (2010) S961, an insulin receptor antagonist causes hyperinsulinemia, insulin-resistance and depletion of energy stores in rats. Biochem Biophys Res Commun 398:260–265. https://doi.org/10.1016/j.bbrc.2010.06.070
Vuong B, Odero G, Rozbacher S et al (2017) Exposure to gestational diabetes mellitus induces neuroinflammation , derangement of hippocampal neurons , and cognitive changes in rat offspring. J Neuroinflammation:1–13. https://doi.org/10.1186/s12974-017-0859-9
Wrann CD, White JP, Salogiannnis J et al (2013) Exercise induces hippocampal BDNF through a PGC-1α/FNDC5 pathway. Cell Metab 18:649–659. https://doi.org/10.1016/j.cmet.2013.09.008
Yang G, Cancino GI, Zahr SK et al (2016) A glo1-methylglyoxal pathway that is perturbed in maternal diabetes regulates embryonic and adult neural stem cell pools in murine offspring. Cell Rep 17:1022–1036. https://doi.org/10.1016/j.celrep.2016.09.067
Yao L, Wan J, Li H et al (2015) Resveratrol relieves gestational diabetes mellitus in mice through activating AMPK. Reprod Biol Endocrinol 13:118. https://doi.org/10.1186/s12958-015-0114-0
Yi P, Park JS, Melton DA (2013) Betatrophin: a hormone that controls pancreatic β cell proliferation. Cell 153:747–758. https://doi.org/10.1016/j.cell.2013.04.008
Funding
RALS received fellowship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil - Finance Code 001.
Author information
Authors and Affiliations
Contributions
RALS wrote the manuscript; performed the literature research; analyzed and critically discussed the data. The author read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Disclosure of potential conflicts of interest
The author declares no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Data statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
De Sousa, R.A.L. Animal models of gestational diabetes: characteristics and consequences to the brain and behavior of the offspring. Metab Brain Dis 36, 199–204 (2021). https://doi.org/10.1007/s11011-020-00661-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-020-00661-9