Abstract
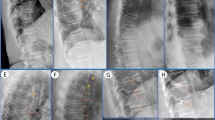
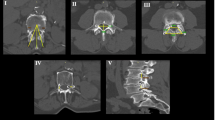
Loss of function mutations in STAT3 (STAT3-LOF; autosomal dominant hyper-IgE (Job’s) syndrome) are associated with a variety of musculoskeletal manifestations, including scoliosis, osteoporosis, and minimal trauma fractures. This retrospective magnetic resonance (MR) imaging study sought to characterize an unusual pattern of cervical spine degeneration among a cohort of STAT3-LOF patients. Cervical spine MR images of the STAT3-LOF cohort (n = 38) were assessed for a variety of degenerative changes and compared to age-matched groups of controls (n = 42) without known immune or musculoskeletal abnormalities. A unique pattern of premature cervical spine degeneration was identified among the STAT3-LOF cohort which included straightening and eventual reversal of the normal cervical lordosis, mainly due to multi-level spondylolisthesis, as well as early development of spinal canal narrowing, cord compression, and myelomalacia. Cervical spine degeneration in the STAT3-LOF cohort was significantly worse than controls in both the 30–45 and 45 + age groups. Moderate to severe degenerative changes were present after age 30, and markedly worsened over time in several cases. Bone mineral density (BMD) had a moderate negative correlation with cervical degeneration severity and a strong negative correlation with age among STAT3-LOF participants. Cervical degeneration in STAT3-LOF appears to be progressive and could result in cord compromise if left unaddressed. Focused history and physical examination for signs of neurologic compromise as well as periodic MR imaging are thus recommended for the evaluation of premature cervical spine degeneration in STAT3-LOF patients after age 30 so that timely surgical interventions may be considered to prevent spinal cord damage and permanent neurological deficits.





Similar content being viewed by others
References
Farmand S, Sundin M. Hyper-IgE syndromes: recent advances in pathogenesis, diagnostics and clinical care. Curr Opin Hematol. 2015;22(1):12–22. https://doi.org/10.1097/moh.0000000000000104.
Gernez Y, Freeman AF, Holland SM, Garabedian E, Patel NC, Puck JM, et al. Autosomal dominant hyper-IgE syndrome in the USIDNET Registry. J Allergy Clin Immunol Pract. 2018;6(3):996–1001. https://doi.org/10.1016/j.jaip.2017.06.041.
Hashemi H, Mohebbi M, Mehravaran S, Mazloumi M, Jahanbani-Ardakani H, Abtahi SH. Hyperimmunoglobulin E syndrome: genetics, immunopathogenesis, clinical findings, and treatment modalities. J Res Med Sci. 2017;22:53. https://doi.org/10.4103/jrms.JRMS_1050_16.
Sowerwine KJ, Shaw PA, Gu W, Ling JC, Collins MT, Darnell DN, et al. Bone density and fractures in autosomal dominant hyper IgE syndrome. J Clin Immunol. 2014;34(2):260–4. https://doi.org/10.1007/s10875-013-9982-2.
Zhang Q, Boisson B, Beziat V, Puel A, Casanova JL. Human hyper-IgE syndrome: singular or plural? Mamm Genome. 2018;29(7–8):603–17. https://doi.org/10.1007/s00335-018-9767-2.
Tang Y, Xu X, Zhu F, Chen C, Wang F, Lu M, et al. Incidence and risk factors of cervical kyphosis in patients with adolescent idiopathic scoliosis. World Neurosurg. 2019;127:e788–e92. https://doi.org/10.1016/j.wneu.2019.03.264.
Akbar M, Almansour H, Lafage R, Diebo BG, Wiedenhofer B, Schwab F, et al. Sagittal alignment of the cervical spine in the setting of adolescent idiopathic scoliosis. J Neurosurg Spine. 2018;29(5):506–14. https://doi.org/10.3171/2018.3.Spine171263.
Lippa L, Lippa L, Cacciola F. Loss of cervical lordosis: what is the prognosis? J Craniovertebr Junction Spine. 2017;8(1):9–14. https://doi.org/10.4103/0974-8237.199877.
Zheng C, Zhu Y, Lu F, Ma X, Tian D, Jiang J. Trans-synaptic degeneration of motoneurons distal to chronic cervical spinal cord compression in cervical spondylotic myelopathy. Int J Neurosci. 2017;127(11):988–95. https://doi.org/10.1080/00207454.2017.1287701.
Sun Y, Yu K, Wang H, Shen Y, Kong L, Zhang J. Diagnosis and treatment of hidden lesions in “mild” cervical spondylotic myelopathy patients with apparent symptoms. Medicine (Baltimore). 2017;96(30):e7623. https://doi.org/10.1097/MD.0000000000007623.
Dimai HP. Use of dual-energy X-ray absorptiometry (DXA) for diagnosis and fracture risk assessment; WHO-criteria, T- and Z-score, and reference databases. Bone. 2017;104:39–43. https://doi.org/10.1016/j.bone.2016.12.016.
Grimbacher B, Schaffer AA, Holland SM, Davis J, Gallin JI, Malech HL, et al. Genetic linkage of hyper-IgE syndrome to chromosome 4. Am J Hum Genet. 1999;65(3):735–44. https://doi.org/10.1086/302547.
Woellner C, Gertz EM, Schaffer AA, Lagos M, Perro M, Glocker EO, et al. Mutations in STAT3 and diagnostic guidelines for hyper-IgE syndrome. J Allergy Clin Immunol. 2010;125(2):424–32 e8. https://doi.org/10.1016/j.jaci.2009.10.059.
DeWitt CA, Bishop AB, Buescher LS, Stone SP. Hyperimmunoglobulin E syndrome: two cases and a review of the literature. J Am Acad Dermatol. 2006;54(5):855–65. https://doi.org/10.1016/j.jaad.2005.10.022.
Grimbacher B, Holland SM, Puck JM. Hyper-IgE syndromes. Immunol Rev. 2005;203:244–50. https://doi.org/10.1111/j.0105-2896.2005.00228.x.
Scheuerman O, Hoffer V, Cohen AH, Woellner C, Grimbacher B, Garty BZ. Reduced bone density in patients with autosomal dominant hyper-IgE syndrome. J Clin Immunol. 2013;33(5):903–8. https://doi.org/10.1007/s10875-013-9895-0.
Been E, Shefi S, Soudack M. Cervical lordosis: the effect of age and gender. Spine J. 2017;17(6):880–8. https://doi.org/10.1016/j.spinee.2017.02.007.
Deburge A, Mazda K, Guigui P. Unstable degenerative spondylolisthesis of the cervical spine. J Bone Joint Surg Br. 1995;77(1):122–5.
Woiciechowsky C, Thomale UW, Kroppenstedt SN. Degenerative spondylolisthesis of the cervical spine--symptoms and surgical strategies depending on disease progress. Eur Spine J. 2004;13(8):680–4. https://doi.org/10.1007/s00586-004-0673-9.
Jiang SD, Jiang LS, Dai LY. Degenerative cervical spondylolisthesis: a systematic review. Int Orthop. 2011;35(6):869–75. https://doi.org/10.1007/s00264-010-1203-5.
Beziat V, Tavernier SJ, Chen YH, Ma CS, Materna M, Laurence A, et al. Dominant-negative mutations in human IL6ST underlie hyper-IgE syndrome. J Exp Med. 2020;217(6). https://doi.org/10.1084/jem.20191804.
Shahin T, Aschenbrenner D, Cagdas D, Bal SK, Conde CD, Garncarz W, et al. Selective loss of function variants in IL6ST cause hyper-IgE syndrome with distinct impairments of T-cell phenotype and function. Haematologica. 2019;104(3):609–21. https://doi.org/10.3324/haematol.2018.194233.
Nieminen P, Morgan NV, Fenwick AL, Parmanen S, Veistinen L, Mikkola ML, et al. Inactivation of IL11 signaling causes craniosynostosis, delayed tooth eruption, and supernumerary teeth. Am J Hum Genet. 2011;89(1):67–81. https://doi.org/10.1016/j.ajhg.2011.05.024.
Dmitrieva NI, Walts AD, Nguyen DP, Grubb A, Zhang X, Wang X, et al. Impaired angiogenesis and extracellular matrix metabolism in autosomal-dominant hyper-IgE syndrome. J Clin Invest. 2020. https://doi.org/10.1172/JCI135490.
Goel S, Sahu S, Minz RW, Singh S, Suri D, Oh YM, et al. STAT3-mediated transcriptional regulation of osteopontin in STAT3 loss-of-function related hyper IgE syndrome. Front Immunol. 2018;9:1080. https://doi.org/10.3389/fimmu.2018.01080.
Li Y, Xiao W, Sun M, Deng Z, Zeng C, Li H, et al. The expression of osteopontin and Wnt5a in articular cartilage of patients with knee osteoarthritis and its correlation with disease severity. Biomed Res Int. 2016;2016:9561058–7. https://doi.org/10.1155/2016/9561058.
Attur MG, Dave MN, Stuchin S, Kowalski AJ, Steiner G, Abramson SB, et al. Osteopontin: an intrinsic inhibitor of inflammation in cartilage. Arthritis Rheum. 2001;44(3):578–84. https://doi.org/10.1002/1529-0131(200103)44:3<578::AID-ANR106>3.0.CO;2-7.
Funding
This project has been funded in part by the intramural research program at the National Institutes of Health (NIH), in part by the Center for Infectious Disease Imaging (CIDI), Clinical Center, and in part by the National Cancer Institute (under Contract No. 75N91019D00024, Task Order No. 75N91019F00130). The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services or does mention of trade names, commercial products, or organizations imply endorsement by the US Government.
Author information
Authors and Affiliations
Contributions
DAH and AFF conceived of and designed the study. AKU and ALM evaluated the study subjects and/or collected and analyzed the data. ALM performed the statistical analysis and wrote the first draft of the manuscript. DAH and AFF reviewed and finalized the manuscript. All authors participated in revising the paper critically for intellectual content and gave final approval of the submitted manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mitchell, A.L., Urban, A.K., Freeman, A.F. et al. An Unusual Pattern of Premature Cervical Spine Degeneration in STAT3-LOF. J Clin Immunol 41, 576–584 (2021). https://doi.org/10.1007/s10875-020-00926-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-020-00926-z