Abstract
Purpose
The ubiquitous calcium–independent phospholipase A2 enzyme (iPLA2) is inhibited by calmodulin binding and known to be responsible for phospholipid remodeling housekeeping functions including granule exocytosis–associated membrane fusion in normal human neutrophils. We evaluate in human neutrophils the iPLA2 secretagogue effects using normal neutrophils, where reactive oxygen species (ROS) generation has been blocked by diphenyleneiodonium, as well as in neutrophils from chronic granulomatous disease (CGD) patients.
Methods
Neutrophils were pretreated with W7, a calmodulin inhibitor known to activate iPLA2 and exocytosis of granules, and vesicles as well as intra- and extra-microbicidal activity against Staphylococcus aureus and Aspergillus fumigatus were evaluated.
Results
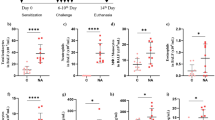
W7 increases exocytosis of primary, secondary, and tertiary granules and vesicles and improves neutrophil microbicidal activity against S. aureus and A. fumigatus.
Conclusions
In neutrophils, calmodulin-mediated iPLA2 inhibition controls granule and vesicle exocytosis in the phagosome and in the extracellular microenvironment. Relieving iPLA2 inhibition results in increased exocytosis of primary, secondary, and tertiary granules and secretory vesicles with correction of defective intracellular and extracellular microbicidal activity. In CGD patients presenting ROS defective production, this increase in the non-oxidative killing pathway partially corrects their microbicidal defects.



Similar content being viewed by others
Abbreviations
- BEL:
-
bromoenol lactone
- Cyt B:
-
cytochalasin B
- CGD:
-
chronic granulomatose disease
- DCFH:
-
dichloro-dihydro-fluorescein
- DPI:
-
diphenyleneiodonium
- iPLA2:
-
calcium-independent phospholipase A2
- MMP-9:
-
matrix metallo-proteinase 9
- MPO:
-
myeloperoxidase
- PAF:
-
platelet-activating factor
- PMN:
-
polymorphonuclear leukocyte
- ROS:
-
reactive oxygen species
- XTT:
-
sodium 3,3′-[1(phenylamino)carbonyl]-3,4-tetrazolium]-3is(4-methoxy-6-nitro) benzene sulfonic acid hydrate
References
Williams R. Killing controversy. J Exp Med. 2006;203(11):2404.
Nauseef WM. How human neutrophils kill and degrade microbes: an integrated view. Immunol Rev. 2007;219:88–102.
Standish AJ, Weiser JN. Human neutrophils kill Streptococcus pneumoniae via serine proteases. J Immunol. 2009;183(4):2602–9.
Ramanadham S, Ali T, Ashley JW, Bone RN, Hancock WD, Lei X. Calcium-independent phospholipases A2 and their roles in biological processes and diseases. J Lipid Res. 2015;56(9):1643–68.
Larsson Forsell PK, Runarsson G, Ibrahim M, Björkholm M, Claesson HE. On the expression of cytosolic calcium-independent phospholipase A2 (88kDa) in immature and mature myeloid cells and its role in leukotriene synthesis in human granulocytes. FEBS Lett. 1998;434(3):295–9.
Zhang D, Shooshtarizadeh P, Laventie BJ, Colin DA, Chich JF, Vidic J, et al. Two chromogranin a-derived peptides induce calcium entry in human neutrophils by calmodulin-regulated calcium independent phospholipase A2. PLoS One. 2009;4(2):e4501.
Ellis JA, Mayer SJ, Jones OT. The effect of the NADPH oxidase inhibitor diphenyleneiodonium on aerobic and anaerobic microbicidal activities of human neutrophils. Biochem J. 1988;251(3):887–91.
Simard JC, Girard D, Tessier PA. Induction of neutrophil degranulation by S100A9 via a MAPK-dependent mechanism. J Leukoc Biol. 2010;87(5):905–14.
Stemerding AM, Köhl J, Pandey MK, Kuipers A, Leusen JH, Boross P, et al. Staphylococcus aureus formyl peptide receptor-like 1 inhibitor (FLIPr) and its homologue FLIPr-like are potent FcγR antagonists that inhibit IgG-mediated effector functions. J Immunol. 2013;191(1):353–62.
Flamand L, Tremblay MJ, Borgeat P. Leukotriene B4 triggers the in vitro and in vivo release of potent antimicrobial agents. J Immunol. 2007;178(12):8036–45.
Lee MJ, Liu H, Barker BM, Snarr BD, Gravelat FN, Al Abdallah Q, et al. The fungal exopolysaccharide galactosaminogalactan mediates virulence by enhancing resistance to neutrophil extracellular traps. PLoS Pathog. 2015;11(10):e1005187.
Jog NR, Rane MJ, Lominadze G, Luerman GC, Ward RA, McLeish KR. The actin cytoskeleton regulates exocytosis of all neutrophil granule subsets. Am J Physiol Cell Physiol. 2007;292(5):C1690–700.
Borregaard N, Sørensen OE, Theilgaard-Mönch K. Neutrophil granules: a library of innate immunity proteins. Trends Immunol. 2007;28(8):340–5.
Jian Lian JP, Crossley L, Zhan Q, Huang R, Coffer P, Toker A, et al. Antagonists of calcium fluxes and calmodulin block activation of the p21-activated protein kinases in neutrophils. J Immunol. 2001;166(4):2643–50.
Verploegen S, van Leeuwen CM, van Deutekom HW, Lammers JW, Koenderman L, Coffer PJ. Role of Ca2+/calmodulin regulated signaling pathways in chemoattractant induced neutrophil effector functions. Comparison with the role of phosphotidylinositol-3 kinase. Eur J Biochem. 2002;269(18):4625–34.
Jenkins CM, Han X, Mancuso DJ, Gross RW. Identification of calcium-independent phospholipase A2 (iPLA2) beta, and not iPLA2gamma, as the mediator of arginine vasopressin-induced arachidonic acid release in A-10 smooth muscle cells. Enantioselective mechanism-based discrimination of mammalian iPLA2s. J Biol Chem. 2002;277:32807–14.
Odell EW, Segal AW. Killing of pathogens associated with chronic granulomatous disease by the non-oxidative microbicidal mechanisms of human neutrophils. J Med Microbiol. 1991;34(3):129–35.
Leliefeld PH, Koenderman L, Pillay J. How neutrophils shape adaptive immune responses. Front Immunol. 2015;6:471.
Bakema JE, Ganzevles SH, Fluitsma DM, Schilham MW, Beelen RH, Valerius T, et al. Targeting FcαRI on polymorphonuclear cells induces tumor cell killing through autophagy. J Immunol. 2011;187(2):726–32.
Driessen HE, Bourgonje VJ, van Veen TA, Vos MA. New antiarrhythmic targets to control intracellular calcium handling. Neth Heart J. 2014;22(5):198–213.
Ashpole NM, Song W, Brustovetsky T, Engleman EA, Brustovetsky N, Cummins TR, et al. Calcium/calmodulin-dependent protein kinase II (CaMKII) inhibition induces neurotoxicity via dysregulation of glutamate/calcium signaling and hyperexcitability. J Biol Chem. 2012;287:8495–506.
Nikolaou A, Kokotou MG, Vasilakaki S, Kokotos G. Small-molecule inhibitors as potential therapeutics and as tools to understand the role of phospholipases A2. Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jun;1864(6):941–56.
Acknowledgements
The authors thank Drs. Christophe Chantrain, Alice Ferster, Benoit Florkin, Pierre Philippet, and Jutte Van Der Werff Ten Bosch for the CGD blood samples and Drs. Brigitte Cantiniaux and Francis Corazza for the discussion.
Funding
This work was supported by the Kriibskrank Kanner Foundatioun of Luxembourg.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
PMN were obtained from 9 CGD patients after written authorization in accordance with Declaration of Helsinki, following protocol approved by the Hôpital des Enfants ethical committee
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 223 kb)
Rights and permissions
About this article
Cite this article
Harfi, I., D’Hondt, S. & Sariban, E. iPLA2 Activation Mediates Granular Exocytosis and Corrects Microbicidal Defects in ROS-Deficient and CGD Human Neutrophils. J Clin Immunol 39, 486–493 (2019). https://doi.org/10.1007/s10875-019-00630-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-019-00630-7