Abstract
Purpose
To study the attitude of BRCA1/2 mutation carriers regarding family planning, fertility preservation, and preimplantation genetic testing (PGT).
Methods
A national cross-sectional study was conducted by the distribution of an anonymous questionnaire, from August 2022 to January 2023. The main outcomes measures were discussion, acceptance, and performance rates of fertility preservation and PGT.
Results
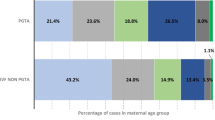
The questionnaire was completed by 530 BRCA1/2 mutation carriers. The mean (SD) age at mutation detection was 36.4 (9.6) years. At the time of mutation detection, 40% did not have children. Following mutation detection, 37% of responders changed their family planning, mostly choosing to have children earlier or to have less children than planned. Twenty-eight percent of BRCA carriers discussed the option of fertility preservation with a physician, 72% agreed that fertility preservation is an acceptable option for BRCA1/2 mutation carriers and finally 11% underwent oocyte/embryo vitrification before RRBSO. 44% of BRCA carriers discussed the option of PGT, 58% agreed that PGT is justified in BRCA1/2 mutation carriers and finally 8% underwent PGT to select non-carrier embryos. In a multivariate analysis, age under 35 years and the a priori need for fertility treatments were both found significant factors increasing the likelihood of performing fertility preservation and PGT.
Conclusion
This study emphasizes that despite a substantial proportion of women admitting that mutation detection affected their family planning and high acceptance rates, performance of fertility preservation and PGT remained exceedingly low. Increasing the knowledge and awareness of these issues is important and should be included in multidisciplinary counselling.

Similar content being viewed by others
Data availability
Data available on request from the authors.
References
Practice Bulletin No 182: Hereditary Breast and Ovarian Cancer Syndrome. Obstet Gynecol. 2017;130(3):e110–e126. https://doi.org/10.1097/AOG.0000000000002296
Lewis KE, Lu KH, Klimczak AM, Mok SC. Recommendations and choices for BRCA mutation carriers at risk for ovarian cancer: A complicated decision. Cancers (Basel) [Internet]. 2018;10 https://doi.org/10.3390/cancers10020057.
Dwyer, Mary. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines ® ) Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic. 2023 [cited 2023 Feb 2]; Available from: https://www.nccn.org/home/member-
Rosen E, Pishvaian M. Targeting the BRCA1/2 tumor suppressors. Curr Drug Targets [Internet]. 2014 [cited 2023 Feb 16];15:17–31. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/24387337/
Lombardi L, Trumello C, Stuppia L, Antonucci I, Brandão T, Babore A. BRCA1/2 pathogenetic variant carriers and reproductive decisions: Gender differences and factors associated with the choice of preimplantation genetic diagnosis (PGD) and prenatal diagnosis (PND). J Assist Reprod Genet [Internet]. 2022; [cited 2023 Feb 16];39:1433–43. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/35661074/
Menon U, Harper J, Sharma A, Fraser L, Burnell M, El Masry K, et al. Views of BRCA gene mutation carriers on preimplantation genetic diagnosis as a reproductive option for hereditary breast and ovarian cancer. Hum Reprod [Internet]. 2007; [cited 2023 Feb 16];22:1573–7. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/17428877/
Gietel-Habets JJG, de Die-Smulders CEM, Derks-Smeets IAP, Tibben A, Tjan-Heijnen VCG, van Golde R, et al. Awareness and attitude regarding reproductive options of persons carrying a BRCA mutation and their partners. Hum Reprod [Internet]. 2017; [cited 2023 Feb 16];32:588–97. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/28073972/
Quinn GP, Pal T, Murphy D, Vadaparampil ST, Kumar A. High-risk consumers’ perceptions of preimplantation genetic diagnosis for hereditary cancers: a systematic review and meta-analysis. Genet Med. 2012;14:191–200.
BRCA: בדיקת סקר גנטית לסרטן השד ולסרטן השחלה | שירותי בריאות כללית [Internet]. [cited 2023 Feb 3]. Available from: https://www.clalit.co.il/he/your_health/family/cancer/Pages/brca_gene_survey_test.aspx
Eysenbach G. Improving the Quality of Web Surveys: The Checklist for Reporting Results of Internet E-Surveys (CHERRIES). J Med Internet Res 2004;6(3):e34 https://www.jmir.org/2004/3/e34 [Internet]. 2004 [cited 2023 Feb 2];6:e132. Available from: https://www.jmir.org/2004/3/e34
Vadaparampil ST, Quinn GP, Knapp C, Malo TL. Friedman S. Factors associated with preimplantation genetic diagnosis acceptance among women concerned about hereditary breast and ovarian cancer. Genet Med [Internet]. 2009; [cited 2023 Feb 20];11:757–65. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/19710615/
Leaver M, Wells D. Non-invasive preimplantation genetic testing (niPGT): the next revolution in reproductive genetics? Hum Reprod Update [Internet]. 2020 [cited 2023 Sep 4];26:16–42. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/31774124/
Laot L, Sonigo C, Nobre J, Benachi A, Dervin T, El Moujahed L, et al. Should Preimplantation Genetic Testing (PGT) Systematically Be Proposed to BRCA Pathogenic Variant Carriers? Cancers (Basel) [Internet]. 2022 [cited 2023 May 15];14. Available from: https://pubmed-ncbi-nlm-nih-gov.carmelmc.idm.oclc.org/36497251/
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Ethical approval was waived by the local Ethics Committee of Carmel Medical Center since the questionnaires were distributed anonymously on social networks, the women answered the questionnaire of their own free will, and there was no way to identify them and access their personal and medical records.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 23.1 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nahshon, C., Lavie, O. & Oron, G. Attitude of BRCA1/2 mutation carriers towards fertility preservation, family planning and preimplantation genetic testing for primary prevention of breast and ovarian cancer in the next generation. J Assist Reprod Genet 40, 2835–2842 (2023). https://doi.org/10.1007/s10815-023-02954-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-023-02954-1