Abstract
Purpose
We investigated if automated TLI selection may be a valuable strategy to identify those euploid embryos with the best chances of success.
Methods
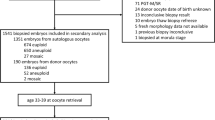
This is a unicentric and retrospective study involving 244 patients undergoing preimplantational genetic screening (PGS) cycles with autologous oocytes or oocyte donation (OD) with single euploid embryo transferred. We examined euploid embryos selected for transfer based on morphology evaluation alone (PGS-only; control group) or by assessment using an automated TLI system (Eeva™; PGS-TLI group).
Results
In both, autologous oocytes and OD patients, significantly better implantation and clinical and ongoing pregnancy rates were obtained in the PGS-TLI group when euploid embryos with high implantation potential as predicted by the automated TLI System (Eeva™) were transferred compared with the PGS-only group. This improvement was also observed when only transfers of good morphological quality embryos were compared. TLI categories showed significant differences on blastocyst formation and euploidy rate.
Conclusions
Automated TLI combined with PGS is a useful prognostic tool to identify euploid embryos with the highest potential for implantation and pregnancy. Further, these results provide evidence that a healthy pregnancy does not only depend upon normal chromosomal status.


Similar content being viewed by others
References
De Sutter P, Gerris J, Dhont M. A health-economic decision-analytic model comparing double with single embryo transfer in IVF/ICSI: a sensitivity analysis. Hum Reprod. 2003:1361.
Bromer JG, Ata B, Seli M, Lockwood CJ, Seli E. Preterm deliveries that result from multiple pregnancies associated with assisted reproductive technologies in the USA: a cost analysis. Curr Opin Obstet Gynecol [Internet]. 2011;23:168–173. Available from: http://ovidsp.ovid.com/ovidweb.cgi?T=JS&PAGE=reference&D=emed10&NEWS=N&AN=2011267305
Swain JE, Carrell D, Cobo A, Meseguer M, Rubio C, Smith GD. Optimizing the culture environment and embryo manipulation to help maintain embryo developmental potential. Fertil Steril. 2016;105:571–87.
Wong KM, Mastenbroek S, Repping S. Cryopreservation of human embryos and its contribution to in vitro fertilization success rates. Fertil Steril [Internet]. Elsevier Inc.; 2014;102:19–26. Available from: https://doi.org/10.1016/j.fertnstert.2014.05.027
Vélez MP, Kadoch IJ, Phillips SJ, Bissonnette F. Rapid policy change to single-embryo transfer while maintaining pregnancy rates per initiated cycle. Reprod BioMed Online. 2013;26:506–11.
Lee AM, Connell MT, Csokmay JM, Styer AK. Elective single embryo transfer—the power of one. Contraception Reprod Med [Internet]; 2016;1:11. Available from: http://contraceptionmedicine.biomedcentral.com/articles/10.1186/s40834-016-0023-4
Steinberg ML, Boulet S, Kissin D, Warner L, Jamieson DJ. Elective single embryo transfer trends and predictors of a good perinatal outcome—United States, 1999 to 2010. Fertil Steril. 2013;99:1937–43.
Balaban B, Brison D, Calderón G, Catt J, Conaghan J, Cowan L, et al. Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Reprod BioMed Online. 2011;22:632–46.
Zhou W, Fu L, Sha W, Chu D, Li Y. Relationship of polar bodies morphology to embryo quality and pregnancy outcome. Zygote [Internet]. 2015;1–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26198980.
Scott L, Finn A, O’Leary T, McLellan S, Hill J. Morphologic parameters of early cleavage-stage embryos that correlate with fetal development and delivery: prospective and applied data for increased pregnancy rates. Hum Reprod. 2007;22:230–40.
Van Den Abbeel E, Balaban B, Ziebe S, Lundin K, Cuesta MJG, Klein BM, et al. Association between blastocyst morphology and outcome of single-blastocyst transfer. Reprod BioMed Online. 2013;27:353–61.
Alfarawati S, Fragouli E, Colls P, Stevens J, Gutiérrez-Mateo C, Schoolcraft WB, et al. The relationship between blastocyst morphology, chromosomal abnormality, and embryo gender. Fertil Steril. 2011;95:520–4.
Obstet AG, Sigalos GA, Triantafyllidou O, Vlahos NF. Novel embryo selection techniques to increase embryo implantation in IVF attempts. Arch Gynecol Obstet. 2016;294(6):1117–24.
Jacobs PA. Retrospective and prospective epidemiological studies of 1,500 karyotyped spontaneous human abortions. Birth Defects Res A Clin Mol Teratol. 2013:487–8.
Plachot M. Chromosome analysis of spontaneous abortions after IVF. A European survey. Hum Reprod [Internet]. 1989;4:425–429. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2501337.
Stephenson MD, Awartani KA, Robinson WP. Cytogenetic analysis of miscarriages from couples with recurrent miscarriage: a case-control study. Hum Reprod. 2002;17:446–51. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11821293%5Cn. http://humrep.oxfordjournals.org/content/17/2/446.full.pdf.
Sugiura-Ogasawara M, Ozaki Y, Katano K, Suzumori N, Kitaori T, Mizutani E. Abnormal embryonic karyotype is the most frequent cause of recurrent miscarriage. Hum Reprod. 2012;27:2297–303.
Barri PN, Coroleu B, Clua E, Tur R, Boada M, Rodriguez I. Investigations into implantation failure in oocyte-donation recipients. Reprod BioMed Online. 2014;28:99–105.
Forman EJ, Hong KH, Ferry KM, Tao X, Taylor D, Levy B, et al. In vitro fertilization with single euploid blastocyst transfer: a randomized controlled trial. Fertil Steril [Internet]. Elsevier Inc.; 2013;100:100–107.e1. Available from: https://doi.org/10.1016/j.fertnstert.2013.02.056
Scott RT, Upham KM, Forman EJ, Hong KH, Scott KL, Taylor D, et al. Blastocyst biopsy with comprehensive chromosome screening and fresh embryo transfer significantly increases in vitro fertilization implantation and delivery rates: a randomized controlled trial. Fertil Steril. 2013;100:697–703.
Yang Z, Liu J, Collins GS, Salem SA, Liu X, Lyle SS, et al. Selection of single blastocysts for fresh transfer via standard morphology assessment alone and with array CGH for good prognosis IVF patients: results from a randomized pilot study. Mol Cytogenet [Internet]. 2012;5:24. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3403960&tool=pmcentrez&rendertype=abstract
Dahdouh EM, Balayla J, García-Velasco JA. Impact of blastocyst biopsy and comprehensive chromosome screening technology on preimplantation genetic screening: a systematic review of randomized controlled trials. Reprod BioMed Online. 2015;30:281–9.
Schoolcraft WB, Surrey E, Minjarez D, Gustofson RL, Scott RT, Katz-Jaffe MG, et al. Comprehensive chromosome screening (CCS) with vitrification results in improved clinical outcome in women >35 years: a randomized control trial. Fertil Steril. Elsevier Ltd; 2012;98:S1. Available from: https://doi.org/10.1016/j.fertnstert.2012.07.002
Lee E, Illingworth P, Wilton L, Chambers GM. The clinical effectiveness of preimplantation genetic diagnosis for aneuploidy in all 24 chromosomes (PGD-A): systematic review. Hum Reprod. 2015:473–83.
Vaiarelli A, Cimadomo D, Capalbo A, Orlando G, Sapienza F, Colamaria S, Palagiano A, Bulletti C, Rienzi L, Ubaldi FM Pre-implantation genetic testing in ART: who will benefit and what is the evidence?. J Assist Reprod Genet [Internet]; 2016;33:1273–1278. Available from: https://doi.org/10.1007/s10815-016-0785-2
Capalbo A, Rienzi L, Cimadomo D, Maggiulli R, Elliott T, Wright G, et al. Correlation between standard blastocyst morphology, euploidy and implantation: an observational study in two centers involving 956 screened blastocysts. Hum Reprod. 2014;29:1173–81.
Montag M, Toth B, Strowitzki T. New approaches to embryo selection. Reprod BioMed Online. 2013:539–46.
Gardner DK, Meseguer M, Rubio C, Treff NR. Diagnosis of human preimplantation embryo viability. Hum Reprod Update. 2015;21:727–47.
Fragouli E, Spath K, Alfarawati S, Kaper F, Craig A, Michel C-E, et al. Altered levels of mitochondrial DNA are associated with female age, aneuploidy, and provide an independent measure of embryonic implantation potential. PLoS Genet [Internet]. 2015;11:e1005241. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4454688&tool=pmcentrez&rendertype=abstract
Diez-Juan A, Rubio C, Marin C, Martinez S, Al-Asmar N, Riboldi M, et al. Mitochondrial DNA content as a viability score in human euploid embryos: less is better. Fertil Steril. 2015;104:534–541.e1.
Tejera A, Castello D, de Los Santos JM, Pellicer A, Remohi J. Meseguer M. Fertil Steril: Combination of metabolism measurement and a time-lapse system provides an embryo selection method based on oxygen uptake and chronology of cytokinesis timing; 2016.
Dominguez F, Meseguer M, Aparicio-Ruiz B, Piqueras P, Quiñonero A, Simón C. New strategy for diagnosing embryo implantation potential by combining proteomics and time-lapse technologies. Fertil Steril. 2015;104:908–14.
Treff NR, Zhan Y, Tao X, Olcha M, Han M, Rajchel J, et al. Levels of trophectoderm mitochondrial DNA do not predict the reproductive potential of sibling embryos. Hum Reprod. 2017;32:954–62.
Wong CC, Loewke KE, Bossert NL, Behr B, De Jonge CJ, Baer TM, et al. Non-invasive imaging of human embryos before embryonic genome activation predicts development to the blastocyst stage. Nat Biotechnol. 2010;28:1115–21. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20890283
Chavez SL, Loewke KE, Han J, Moussavi F, Colls P, Munne S, et al. Dynamic blastomere behaviour reflects human embryo ploidy by the four-cell stage. Nat Commun [Internet]. 2012;3:1251. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3535341&tool=pmcentrez&rendertype=abstract
Conaghan J, Chen AA, Willman SP, Ivani K, Chenette PE, Boostanfar R, et al. Improving embryo selection using a computer-automated time-lapse image analysis test plus day 3 morphology: results from a prospective multicenter trial. Fertil Steril. 2013;100:412–419.e5.
Vermilyea MD, Tan L, Anthony JT, Conaghan J, Ivani K, Gvakharia M, et al. Computer-automated time-lapse analysis results correlate with embryo implantation and clinical pregnancy: a blinded, multi-centre study. Reprod BioMed Online [Internet]. Reproductive Healthcare Ltd.; 2014;29:729–736. Available from: https://doi.org/10.1016/j.rbmo.2014.09.005
Adamson GD, Abusief ME, Palao L, Witmer J, Palao LM, Gvakharia M. Improved implantation rates of day 3 embryo transfers with the use of an automated time-lapse-enabled test to aid in embryo selection. Fertil Steril [Internet]. Elsevier Inc.; 2016;105:369–375. Available from: https://doi.org/10.1016/j.fertnstert.2015.10.030
Aparicio-Ruiz B, Basile N, Pérez Albalá S, Bronet F, Remohí J, Meseguer M. Automatic time-lapse instrument is superior to single-point morphology observation for selecting viable embryos: retrospective study in oocyte donation. Fertil Steril [Internet]. 2016;106:1379–1385.e10. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0015028216626237
Kieslinger DC, De Gheselle S, Lambalk CB, De Sutter P, Kostelijk EH, Twisk JWR, et al. Embryo selection using time-lapse analysis (early embryo viability assessment) in conjunction with standard morphology: a prospective two-center pilot study. Hum Reprod. 2016;31:2450–7.
Diamond MP, Suraj V, Behnke EJ, Yang X, Angle MJ, Lambe-Steinmiller JC, et al. Using the Eeva test™ adjunctively to traditional day 3 morphology is informative for consistent embryo assessment within a panel of embryologists with diverse experience. J Assist Reprod Genet. 2014;32:61–8.
Cruz M, Gadea B, Garrido N, Pedersen KS, Martínez M, Pérez-Cano I, et al. Embryo quality, blastocyst and ongoing pregnancy rates in oocyte donation patients whose embryos were monitored by time-lapse imaging. J Assist Reprod Genet. 2011;28:569–73.
Meseguer M, Herrero J, Tejera A, Hilligsøe KM, Ramsing NB, Remoh J. The use of morphokinetics as a predictor of embryo implantation. Hum Reprod. 2011;26:2658–71. Available from: http://humrep.oxfordjournals.org/lookup/doi/10.1093/humrep/der256
Perez S, Rubio I, Aparicio B, Beltran D, Garcia-Laez V, Meseguer M. Prospective validation of a time-lapse based algorithm for embryo selection. Fertil Steril. 2014:e322.
Chen A A., Tan L, Suraj V, Reijo Pera R, Shen S. Biomarkers identified with time-lapse imaging: discovery, validation, and practical application. Fertil Steril [Internet]. 2013;99:1035–1043. Available from: https://doi.org/10.1016/j.fertnstert.2013.01.143
Rubio I, Galán A, Larreategui Z, Ayerdi F, Bellver J, Herrero J, et al. Clinical validation of embryo culture and selection by morphokinetic analysis: a randomized, controlled trial of the EmbryoScope. Fertil Steril. 2014;102:1287–94.
Goodman LR, Goldberg J, Falcone T, Austin C, Desai N. Does the addition of time-lapse morphokinetics in the selection of embryos for transfer improve pregnancy rates? A randomized controlled trial. Fertil Steril [Internet]. Elsevier Inc.; 2015; Available from: https://doi.org/10.1016/j.fertnstert.2015.10.013
Milewski R, Kuczyńska A, Stankiewicz B, Kuczyński W. How much information about embryo implantation potential is included in morphokinetic data? A prediction model based on artificial neural networks and principal component analysis. Adv Med Sci. 2017;62:202–6.
Pribenszky C, Nilselid AM, Montag M. Time-lapse culture with morphokinetic embryo selection improves pregnancy and live birth chances and reduces early pregnancy loss: a meta-analysis. Reprod BioMed Online. 2017;35:511–20.
Basile N, Nogales MDC, Bronet F, Florensa M, Riqueiros M, Rodrigo L, et al. Increasing the probability of selecting chromosomally normal embryos by time-lapse morphokinetics analysis. Fertil Steril [Internet]. 2014;101:699–704.e1. Available from: https://doi.org/10.1016/j.fertnstert.2013.12.005
Vera-Rodriguez M, Chavez SL, Rubio C, Reijo Pera RA, Simon C. Prediction model for aneuploidy in early human embryo development revealed by single-cell analysis. Nat Commun [Internet]. 2015;6:7601. Available from: http://www.nature.com/doifinder/10.1038/ncomms8601
Campbell A, Fishel S, Bowman N, Duffy S, Sedler M, Thornton S. Retrospective analysis of outcomes after IVF using an aneuploidy risk model derived from time-lapse imaging without PGS. Reprod BioMed Online. 2013;27:140–6.
Rienzi L, Capalbo A, Stoppa M, Romano S, Maggiulli R, Albricci L, et al. No evidence of association between blastocyst aneuploidy and morphokinetic assessment in a selected population of poor-prognosis patients: a longitudinal cohort study. Reprod BioMed Online. 2015;30:57–66.
Kramer YG, Kofinas JD, Melzer K, Noyes N, McCaffrey C, Buldo-Licciardi J, et al. Assessing morphokinetic parameters via time lapse microscopy (TLM) to predict euploidy: are aneuploidy risk classification models universal? J Assist Reprod Genet. 2014;31:1231–42.
Reignier A, Lammers J, Barriere P, Freour T. Can time-lapse parameters predict embryo ploidy? A systematic review. Reprod BioMed Online [Internet]. Elsevier Ltd; 2018; Available from: https://doi.org/10.1016/j.rbmo.2018.01.001.
Yang Z, Zhang J, Salem SA, Liu X, Kuang Y, Salem RD, et al. Selection of competent blastocysts for transfer by combining time-lapse monitoring and array CGH testing for patients undergoing preimplantation genetic screening: a prospective study with sibling oocytes. BMC Med Genet [Internet]. 2014;7:38. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4077552&tool=pmcentrez&rendertype=abstract
Hurtado de Mendoza MV, Ten J. Evaluación morfológica de cada estadio de D+0 a D+3. Cuad Embriol clínica criterios ASEBIR valoración morfológica oocitos, embriones tempranos y blastocistos humanos. 2015;9–20.
Gardner DK, Schoolcraft WB. Culture and transfer of human blastocysts. Curr Opin Obstet Gynecol [Internet]. 1999;11:307–311. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10369209.
Gardner DK, Sakkas D. Assessment of embryo viability: the ability to select a single embryo for transfer—a review. Placenta. 2003;24:S5–S12.
Kuwayama M. Highly efficient vitrification for cryopreservation of human oocytes and embryos: the Cryotop method. Theriogenology. 2007;67:73–80.
Maheshwari A, Papathanasiou A, Osmani B, Teoh P. Morphological assessment of embryo quality during assisted reproduction: a systematic review. Fertil Sci Res [Internet]. 2014;1:67. Available from: http://www.fertilityscienceresearch.org/text.asp?2014/1/2/67/162776
Basile N, Morbeck D, García-Velasco J, Bronet F, Meseguer M. Type of culture media does not affect embryo kinetics: a time-lapse analysis of sibling oocytes. Hum Reprod. 2013;28:634–41.
Kirkegaard K, Hindkjaer JJ, Ingerslev HJ. Effect of oxygen concentration on human embryo development evaluated by time-lapse monitoring. Fertil Steril. 2013;99:738–744.e4.
Muñoz M, Cruz M, Humaidan P, Garrido N, Pérez-Cano I, Meseguer M. The type of GnRH analogue used during controlled ovarian stimulation influences early embryo developmental kinetics: a time-lapse study. Eur J Obstet Gynecol Reprod Biol. 2013;168:167–72.
Cruz M, Garrido N, Gadea B, Muñoz M, Pérez-Cano I, Meseguer M. Oocyte insemination techniques are related to alterations of embryo developmental timing in an oocyte donation model. Reprod BioMed Online. 2013;27:367–75.
Bodri D, Sugimoto T, Serna JY, Kondo M, Kato R, Kawachiya S, et al. Influence of different oocyte insemination techniques on early and late morphokinetic parameters: retrospective analysis of 500 time-lapse monitored blastocysts. Fertil Steril. 2015;104:1175–1181.e2.
Conaghan J, Tan L, Gvakharia M, Ivani K, Shen S, Pera R, et al. Dynamic assessment of early embryo fragmentation by time-lapse analysis may improve cell cycle timing-based embryo selection. Nat Biotech Nat Comm. 2010;28:1115–211251.
Lee YSL, Thouas GA, Gardner DK. Developmental kinetics of cleavage stage mouse embryos are related to their subsequent carbohydrate and amino acid utilization at the blastocyst stage. Hum Reprod. 2015;30:543–52.
Bontekoe S, Mantikou E, van Wely M, Seshadri S, Repping S, Mastenbroek S. Low oxygen concentrations for embryo culture in assisted reproductive technologies. Cochrane database Syst rev [Internet]. 2012;7:CD008950. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22786519.
Herbemont C, Sarandi S, Boujenah J, Cedrin-Durnerin I, Sermondade N, Vivot A, et al. Should we consider day-2 and day-3 embryo morphology before day-5 transfer when blastocysts reach a similar good quality? Reprod BioMed Online. 2017;35:521–8.
Irani M, Reichman D, Robles A, Melnick A, Davis O, Zaninovic N, et al. Morphologic grading of euploid blastocysts influences implantation and ongoing pregnancy rates. Fertil Steril [Internet]. Elsevier Inc.; 2017;107:664–70. Available from: https://doi.org/10.1016/j.fertnstert.2016.11.012
Taylor TH, Patrick JL, Gitlin SA, Wilson JM, Crain JL, Griffin DK. Comparison of aneuploidy, pregnancy and live birth rates between day 5 and day 6 blastocysts. Reprod BioMed Online [Internet]. Reproductive Healthcare Ltd.; 2014;29:305–310. Available from: https://doi.org/10.1016/j.rbmo.2014.06.001
Acknowledgments
The authors thank all the IVF-Spain team, especially the IVF Lab for their work and support. Medical writing assistance in the form of language editing and publication structure was provided by Kaedy Bryson (Zoetic Science, an Ashfield company, part of UDG Healthcare plc, Macclesfield, UK).
Funding
No external funding was either sought or obtained for this study.
Author information
Authors and Affiliations
Contributions
E.R. is responsible for conception, design, acquisition, analysis, and interpretation of data and drafting and critical review of the article. M.E. is responsible for analysis and interpretation of data, drafting, and critical review of the article. A.L. is responsible for recruitment and acquisition of data. J.S. is responsible for analysis and interpretation of the data and critical review of the article. J.A. is responsible for the critical review of the article.
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Rocafort, E., Enciso, M., Leza, A. et al. Euploid embryos selected by an automated time-lapse system have superior SET outcomes than selected solely by conventional morphology assessment. J Assist Reprod Genet 35, 1573–1583 (2018). https://doi.org/10.1007/s10815-018-1265-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-018-1265-7