Abstract
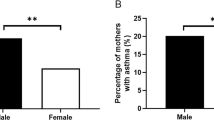
The maternal immune system may play a role in offspring neurodevelopment. We examined whether maternal autoimmune disease, asthma, and allergy were associated with child autism spectrum disorder (ASD) and developmental delay without autism (DD) using 560 ASD cases, 391 typically developing controls, and 168 DD cases from the CHildhood Autism Risk from Genetics and the Environment (CHARGE) study. Results from conditional logistic regression demonstrated few significant associations overall. Maternal autoimmune disease was significantly associated with a modest increase in odds of developmental disorders (combined ASD + DD; OR = 1.46, 95 % CI 1.01, 2.09) but not of ASD alone. Associations with certain allergens and onset periods were also suggested. These findings suggest maternal autoimmune disease may modestly influence childhood developmental disorders (ASD + DD).
Similar content being viewed by others
References
An, B. A. (2002). Performing logistic regression on survey data with the new surveylogistic procedure. SUGI 27, SAS Institute, Inc., Cary, North Carolina, USA. Paper 258–27.
Atladottir, H. O., Pedersen, M. G., Thorsen, P., et al. (2009). Association of family history of autoimmune diseases and autism spectrum disorders. Pediatrics, 124(2), 687–694.
Bandini, L. G., Anderson, S. E., Curtin, C., et al. (2010). Food selectivity in children with autism spectrum disorders and typically developing children. J Pediatr, 157(2), 259–264.
Blalock, J. E., & Smith, E. M. (2007). Conceptual development of the immune system as a sixth sense. Brain, Behavior, and Immunity, 21(1), 23–33.
Boksa, P. (2010). Effects of prenatal infection on brain development and behavior: A review of findings from animal models. Brain, Behavior, and Immunity, 24(6), 881–897.
Boyce, G. C., Smith, T. B., & Casto, G. (1999). Health and educational outcomes of children who experienced severe neonatal medical complications. The Journal of Genetic Psychology, 160(3), 261–269.
Braunschweig, D., & Van de Water, J. (2012). Maternal autoantibodies in autism. Archives of Neurology, 69(6), 693–699.
Braunschweig, D., Ashwood, P., Krakowiak, P., Hertz-Picciotto, I., Hansen, R., Croen, L. A., et al. (2008). Autism: Maternally derived antibodies specific for fetal brain proteins. Neurotoxicology, 29(2), 226–231.
Braunschweig, D., Duncanson, P., Boyce, R., et al. (2012). Behavioral correlates of maternal antibody status among children with autism. Journal of Autism and Developmental Disorders, 42(7), 1435–1445.
Carvalheiras, G., Faria, R., Braga, J., et al. (2012). Fetal outcome in autoimmune diseases. Autoimmunity Reviews, 11(6–7), A520–530.
Comi, A. M., Zimmerman, A. W., Frye, V. H., et al. (1999). Familial clustering of autoimmune disorders and evaluation of medical risk factors in autism. Journal of Child Neurology, 14(6), 384–388.
Croen, L. A., Grether, J. K., Yoshida, C. K., Odouli, R., & Van de Water, J. (2005). Maternal autoimmune diseases, asthma and allergies, and childhood autism spectrum disorders: A case-control study. Archives of Pediatrics and Adolescent Medicine, 159(2), 151–157.
Gardener, H., Spiegelman, D., & Buka, S. L. (2011). Perinatal and neonatal risk factors for autism: A comprehensive meta-analysis. Pediatrics, 128(2), 344–355.
Hertzmark, E., Pazaris, M., & Spiegelman, D. (2009). The SAS mediate macro. Brigham and Women’s Hospital. Boston: Channing Laboratory.
Hertz-Picciotto, I., Croen, L. A., Hansen, R., Jones, C. R., van de Water, J., & Pessah, I. N. (2006). The CHARGE study: An epidemiologic investigation of genetic and environmental factors contributing to autism. Environmental Health Perspectives, 114(7), 1119–1125.
Jones, W. R. (1994). Autoimmune disease and pregnancy. The Australian and New Zealand Journal of Obstetrics Gynaecology, 34(3), 251–258.
Keil, A., Daniels, J. L., Forssen, U., et al. (2010). Parental autoimmune diseases associated with autism spectrum disorders in offspring. Epidemiology, 21(6), 805–808.
Kerstjens, J. M., de Winter, A. F., Bocca-Tjeertes, I. F., et al. (2012). Risk of developmental delay increases exponentially as gestational age of preterm infants decreases: A cohort study at age 4 years. Developmental Medicine and Child Neurology, 54(12), 1096–1101.
Lahita, R. G. (1988). Systemic lupus erythematosus: Learning disability in the male offspring of female patients and relationship to laterality. Psychoneuroendocrinology, 13(5), 385–396.
Lin, D. Y., Fleming, T. R., & De Gruttola, V. (1997). Estimating the proportion of treatment effect explained by a surrogate marker. Statistics in Medicine, 16(13), 1515–1527.
Lord, C., Rutter, M., Goode, S., Heemsbergen, J., Jordan, H., Mawhood, L., et al. (1989). Autism diagnostic observation schedule: A standardized observation of communicative and social behavior. Journal of Autism and Developmental Disorders, 19(2), 185–212.
Lord, C., Rutter, M., & Le Couteur, A. (1994). Autism diagnostic interview-revised: A revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. Journal of Autism and Developmental Disorders, 24(5), 659–685.
McAllister, D. L., Kaplan, B. J., Edworthy, S. M., et al. (1997). The influence of systemic lupus erythematosus on fetal development: Cognitive, behavioral, and health trends. J Int Neuropsychol Soc, 3(4), 370–376.
Money, J., Bobrow, N. A., & Clarke, F. C. (1971). Autism and autoimmune disease: A family study. Journal of Autism and Childhood Schizophrenia, 1(2), 146–160.
Mouridsen, S. E., Rich, B., Isager, T., et al. (2007). Autoimmune diseases in parents of children with infantile autism: A case-control study. Developmental Medicine and Child Neurology, 49(6), 429–432.
Mullen, E. M. (1995). The mullen scales of early learning. Circle Pines, MN: American Guidance Service Inc.
Ross, G., Sammaritano, L., Nass, R., Lockshin, M. (2003). Effects of mothers' autoimmune disease during pregnancy on learning disabilities and hand preference in their children. Arch Pediatr Adolesc Med, 157(4), 397–402.
Rutter, M., Bailey, A., & Lord, C. (2003). The social communication questionnaire. Los Angeles, CA: Western Psychological Services.
Song, Y. W., & Kang, E. H. (2010). Autoantibodies in rheumatoid arthritis: Rheumatoid factors and anticitrullinated protein antibodies. QJM, 103(3), 139–146.
Sparrow, S. S., Balla, D. A., & Cicchetti, D. V. (1984). Vineland adaptive behavior scales. Bloomington, MN: Pearson Assessments.
Sweeten, T. L., Bowyer, S. L., Posey, D. J., et al. (2003). Increased prevalence of familial autoimmunity in probands with pervasive developmental disorders. Pediatrics, 112(5), e420.
Wootla, B., Lacroix-Desmazes, S., Warrington, A. E., et al. (2011). Autoantibodies with enzymatic properties in human autoimmune diseases. Journal of Autoimmunity, 37(2), 144–150.
Zaretsky, M. V., Alexander, J. M., Byrd, W., et al. (2004). Transfer of inflammatory cytokines across the placenta. Obstetrics and Gynecology, 103(3), 546–550.
Zimmerman, A. W., Connors, S. L., Matteson, K. J., et al. (2007). Maternal antibrain antibodies in autism. Brain Behavior Immunity, 21(3), 351–357.
Acknowledgments
This work was supported by the following grants: National Institute of Environmental Health Sciences R01 ES015359, National Institute of Environmental Health Sciences P01 ES11269, Environmental Protection Agency STAR #R829388 & R833292, and 5-T32MH073124. The authors have no financial relationships relevant to this article or conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix: List of Included Maternal Autoimmune Conditions
Appendix: List of Included Maternal Autoimmune Conditions
-
1.
Addison’s disease
-
2.
Adult Still’s disease
-
3.
Alopecia areata
-
4.
Amyotrophic lateral sclerosis (ALS)
-
5.
Ankylosing spondylitis
-
6.
Antiphospholipid antibody syndrome
-
7.
Aplastic anemia
-
8.
Autoimmune hepatitis
-
9.
Autoimmune thyroid disease
-
10.
Autoimmune thyroiditis
-
11.
Behcet’s syndrome
-
12.
Celiac disease
-
13.
CREST syndrome/Scleroderma/Progressive systemic sclerosis
-
14.
Crohn’s disease
-
15.
Dermatitis herpetiformis
-
16.
Diabetes-Type I
-
17.
Discoid lupus
-
18.
Eczema/Psoriasis
-
19.
Giant cell arteritis
-
20.
Grave’s disease
-
21.
Guillain–Barre syndrome
-
22.
Hashimoto’s thyroiditis
-
23.
Hemolytic anemia
-
24.
Inflammatory bowel disease (IBD)
-
25.
Idiopathic thrombocytopenic purpura (ITP)
-
26.
Kawasaki syndrome
-
27.
Meniere’s disease
-
28.
Mixed connective tissue disease
-
29.
Multiple sclerosis
-
30.
Myasthenia gravis
-
31.
Multiple sclerosis
-
32.
Optic neuritis
-
33.
Pemphigus/Pemphigus vulgaris
-
34.
Pernicious anemia
-
35.
Polymyositis/Dermatomyositis
-
36.
Primary biliary cirrhosis
-
37.
Primary Sjogren’s syndrome
-
38.
Psoriasis
-
39.
Raynaud’s disease/phenomenon
-
40.
Reiter’s syndrome/Reiter’s arthritis
-
41.
Rheumatic carditis/Rheumatic heart disease
-
42.
Rheumatoid arthritis (RA)
-
43.
Sarcoidosis
-
44.
Sjogren’s syndrome
-
45.
Stevens-Johnson syndrome
-
46.
Sydenham’s chorea
-
47.
Systemic lupus erythematosus (SLE)
-
48.
Systemic sclerosis/Scleroderma
-
49.
Thrombocytopenia
-
50.
Thyroid disease
-
51.
Tourette syndrome
-
52.
Ulcerative colitis
-
53.
Uveitis/Iritis
-
54.
Vasculitis
Note: The most common autoimmune conditions in the study population were thyroid diseases, psoriasis, and rheumatoid arthritis; other conditions were rare and accounted for 5 % or less of autoimmune diseases reported.
Rights and permissions
About this article
Cite this article
Lyall, K., Ashwood, P., Van de Water, J. et al. Maternal Immune-Mediated Conditions, Autism Spectrum Disorders, and Developmental Delay. J Autism Dev Disord 44, 1546–1555 (2014). https://doi.org/10.1007/s10803-013-2017-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10803-013-2017-2