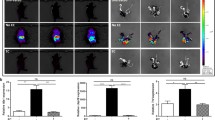
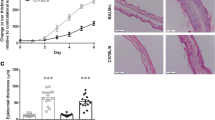
Abstract
Topical imiquimod based creams are indicated as immune stimulants for papillomas and various skin neoplasms. Imiquimod is considered a TLR7 ligand. These creams are also used in research to induce skin inflammation in mice as a model for psoriasis. We observed that this inflammatory response was not strictly imiquimod dependent and we set out to establish which components drive the proinflammatory effects. To this end, we examined the induction response in a BALB/cJRj mouse model, in which 50 mg of cream is applied to 2 cm2 of skin (125 mg/kg imiquimod—5% W/V, and/or 625 mg/kg isostearic acid—25% W/V). Comparing cream formulations containing isostearic acid, imiquimod and the combination, we observed that isostearic acid causes skin inflammation within 2 days, whereas imiquimod requires up to 5 days for initial signs. Isostearic acid activated an inflammasome response, stimulated release of proinflammatory cytokines and upregulated the IL-23/17 axis. Animals treated with isostearic acid had enlarged livers (+ 40% weight), which was not observed with imiquimod alone. Imiquimod was readily metabolized and cleared from plasma and liver, but was maintained at high levels in the skin throughout the body (200 mM at area of application; 200 µM in untreated skin). Imiquimod application was associated with splenomegaly, cytokine induction/release and initial body weight loss over 3 days. Despite high imiquimod skin levels throughout the animal, inflammation was only apparent in the treated areas and was less severe than in isostearic acid groups. As the concentrations in these areas are well above the 10 µM required for TLR7 responses in vitro, there is an implication that skin inflammation following imiquimod is due to effects other than TLR7 agonism (e.g., adenosine receptor agonism). In brain, isostearic caused no major changes in cytokine expression while imiquimod alone sightly stimulated expression of IL-1β and CCL9. However, the combination of both caused brain induction of CCL3, -9, CXCL10, -13, IL-1β and TNFα. The implication of these data is that isostearic acid facilitates the entry of imiquimod or peripherally secreted cytokines into the brain. Our data suggest that psoriaform skin responses in mice are more driven by isostearic acid, than generally reported and that the dose and route used in the model, leads to profound systemic effects, which may complicate the interpretation of drug effects in this model.






Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Bergfeld et al (2018) Safety assessment of fatty acids and soaps as used in cosmetics. In: The 2018 Cosmetic Ingredient Review Expert Panel members are: Chair, Wilma F. [Online]. Available: http://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and
Bourgognon J, Suessmilch M, McColl A, Cavanagh J (2019) Abstract #4297 blood brain barrier breakdown following topical aldara treatment. Brain Behav Immun 81:24–25. https://doi.org/10.1016/j.bbi.2019.08.086
Brück J, Dringen R, Amasuno A, Pau-Charles I, Ghoreschi K (2018) A review of the mechanisms of action of dimethylfumarate in the treatment of psoriasis. Exp Dermatol 27(6):611–624. https://doi.org/10.1111/exd.13548
Butchi NB, Pourciau S, Du M, Morgan TW, Peterson KE (2008) Analysis of the neuroinflammatory response to TLR7 stimulation in the brain: comparison of multiple TLR7 and/or TLR8 agonists. J Immunol 180(11):7604–7612. https://doi.org/10.4049/jimmunol.180.11.7604
Chiricozzi A, Romanelli P, Volpe E, Borsellino G, Romanelli M (2018) Scanning the immunopathogenesis of psoriasis. 19(1)
CIR (2013) Safety assessment of fatty acids and fatty acid salts as used in cosmetics
De Paula D, Martins CA, Bentley MVLB (2008) Development and validation of HPLC method for imiquimod determination in skin penetration studies. Biomed Chromatogr 22(12):1416–1423. https://doi.org/10.1002/bmc.1075
Del Rosso JQ (2005) The use of topical imiquimod for the treatment of actinic keratosis: a status report. Cutis 76(4):241–248
Flutter B, Nestle FO (2013) TLRs to cytokines: mechanistic insights from the imiquimod mouse model of psoriasis. Eur J Immunol 43(12):3138–3146. https://doi.org/10.1002/eji.201343801
Girolomoni G et al (2017) The role of IL-23 and the IL-23/TH17 immune axis in the pathogenesis and treatment of psoriasis. J Eur Acad Dermatol Venereol 31(10):1616–1626. https://doi.org/10.1111/jdv.14433
Global Report on Psoriasis (2023)
Graceway Pharmaceuticals (2009) Center for drug evaluation and pharmacology review: 201153Orig1s000
Hawkes JE, Gudjonsson JE, Ward NL (2017) The snowballing literature on imiquimod-induced skin inflammation in mice: a critical appraisal. J Invest Dermatol 137(3):546–549. https://doi.org/10.1016/j.jid.2016.10.024
Horváth S, Komlódi R, Perkecz A, Pintér E, Gyulai R, Kemény Á (2019) Methodological refinement of Aldara-induced psoriasiform dermatitis model in mice. Sci Rep 9(1):1–8. https://doi.org/10.1038/s41598-019-39903-x
Horváth S, Kemény Á, Pintér E, Gyulai R (2020) A localized aldara (5% imiquimod)–induced psoriasiform dermatitis model in mice using Finn chambers. Curr Protoc Pharmacol 90(1):1–12. https://doi.org/10.1002/cpph.78
Jabeen M et al (2020) Advanced characterization of imiquimod-induced psoriasis-like mouse model. Pharmaceutics 12(9):1–18. https://doi.org/10.3390/pharmaceutics12090789
Luo D, Wu H, Zhao Y, Liu J, Wang F (2016) Different imiquimod creams resulting in differential effects for imiquimod-induced psoriatic mouse models. 1733–1738. https://doi.org/10.1177/1535370216647183
McColl A, Thomson CA, Nerurkar L, Graham GJ, Cavanagh J (2016) TLR7-mediated skin inflammation remotely triggers chemokine expression and leukocyte accumulation in the brain. J Neuroinflammation 13(1):1–16. https://doi.org/10.1186/s12974-016-0562-2
Nakajima K, Sano S (2018) Mouse models of psoriasis and their relevance. J Dermatol 45(3):252–263. https://doi.org/10.1111/1346-8138.14112
Nerurkar L, Mccoll A, Graham G, Cavanagh J (2017) The systemic response to topical aldara treatment is mediated through direct TLR7 stimulation as imiquimod enters the circulation. Sci Rep. https://doi.org/10.1038/s41598-017-16707-5
Pockros PJ et al (2007) Oral resiquimod in chronic HCV infection: Safety and efficacy in 2 placebo-controlled, double-blind phase IIa studies. J Hepatol 47(2):174–182. https://doi.org/10.1016/j.jhep.2007.02.025
Schön MP, Schön M (2007) Imiquimod: mode of action. Br J Dermatol 157(SUPPL. 2):8–13. https://doi.org/10.1111/j.1365-2133.2007.08265.x
Schön MP, Schön M, Klotz KN (2006) The small antitumoral immune response modifier imiquimod interacts with adenosine receptor signaling in a TLR7- and TLR8-independent fashion. J Invest Dermatol 126(6):1338–1347. https://doi.org/10.1038/sj.jid.5700286
Straß S, Schwamborn A, Keppler M, Guse J, Burnet M (2021) Synthesis, characterization and in vivo distribution of intracellular delivered macrolide SCFA derivatives. 1–16. https://doi.org/10.1002/cmdc.202100139
Sun L et al (2018) Comparison of normal versus imiquimod-induced psoriatic skin in mice for penetration of drugs and nanoparticles. Int J Nanomed 13:5625–5635. https://doi.org/10.2147/IJN.S170832
Telò I, Pescina S, Padula C, Santi P, Nicoli S (2016) Mechanisms of imiquimod skin penetration. Int J Pharm 511(1):516–523. https://doi.org/10.1016/j.ijpharm.2016.07.043
van den Höfel N (2018) Basiscreme DAC. https://flexikon.doccheck.com/de/Basiscreme_DAC#
van der Fits L et al (2009) Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol 182(9):5836–5845. https://doi.org/10.4049/jimmunol.0802999
Walter A et al (2013) Aldara activates TLR7-independent immune defence. Nat Commun 4(1):1–13. https://doi.org/10.1038/ncomms2566
Wolff F, Loipetzberger A, Gruber W, Esterbauer H, Aberger F, Frischauf AM (2013) Imiquimod directly inhibits Hedgehog signalling by stimulating adenosine receptor/protein kinase A-mediated GLI phosphorylation. Oncogene 32(50):5574–5581. https://doi.org/10.1038/onc.2013.343
Yang K, Oak ASW, Elewski BE (2021) Use of IL-23 inhibitors for the treatment of plaque psoriasis and psoriatic arthritis: a comprehensive review. Am J Clin Dermatol 22(2):173–192. https://doi.org/10.1007/s40257-020-00578-0
Zhang D et al (2022) Comparison of efficacy of anti-interleukin-17 in the treatment of psoriasis between caucasians and asians: a systematic review and meta-analysis. Front Med 8(January):1–9. https://doi.org/10.3389/fmed.2021.814938
Acknowledgements
We would like to thank colleagues from Synovo GmbH and the University of Tübingen who assisted in this research. Special thanks to the members of the in vivo facility and the team of the analytics/bioanalytics department at both institutions.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
All authors have read and agreed to the published version of the manuscript. Conceptualization, SS, TLS and MB; methodology, SS, TLS, MM and MB; validation, SS, TLS and MB; formal analysis, SS, MM and TLS; investigation, JG, SS, SC, MM, MK, TLS, NS, TF and AS; resources, MB and SL; writing—original draft preparation, SS; writing—review and editing, TLS, MB and JHG; visualization, SS; supervision, MB, JHG and SL; project administration, MB; funding acquisition, MB.
Corresponding author
Ethics declarations
Conflict of interest
SS, JG, MM, TF, NC, MK, LR, AS, NS, SC, JHG, TLS are employees of Synovo GmbH. MB is general manager of Synovo GmbH. Synovo provides models of inflammation to the pharmaceutical industry as a service and the investigation of disease model mechanisms is part of this commercial offering.
Institutional review board statement
All study protocols were approved by the local Animal Care and Ethics Committee (Federal government ethics committee, Tübingen, Germany under the license 35/9185.81-7/SYN 07/18).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Straß, S., Geiger, J., Martorelli, M. et al. Isostearic acid is an active component of imiquimod formulations used to induce psoriaform disease models. Inflammopharmacol 31, 799–812 (2023). https://doi.org/10.1007/s10787-023-01175-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01175-3