Abstract
Background and aim
While diethylcarbamazine citrate (DEC) displays important anti-inflammatory effects in experimental models of liver injury, the mechanisms of its action remain poorly understood. The aim of the present study was to investigate the fibrolytic potential of DEC.
Methods
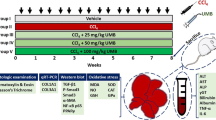
Mice receive two injections of carbon tetrachloride (CCl4) per week for 8 weeks. DEC 50 mg/kg body weight was administered through drinking water during the last 12 days of liver injury.
Results
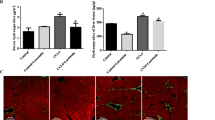
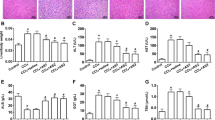
The expression of hepatic stellate cells (HSCs) activation markers, including smooth muscle α-actin (α-SMA), collagen I, transforming growth factor-β 1 (TGF-β1), matrix metalloproteinase-2 (MMP-2) and tissue inhibitor of metalloproteinase-1 (TIMP-1) was assessed. The influence of DEC on the intracellular MAPK pathways of the HSCs (JNK and p38 MAPK) was also estimated. DEC inhibited HSCs activation measured as the production of α-SMA and collagen I. In addition, it down regulated the production of TGF-β1 and TIMP-1, and concomitantly increased MMP-2 activity. Furthermore, DEC significantly inhibited the activation of the JNK and p38 MAPK signaling pathways.
Conclusions
In conclusion, DEC significantly attenuated the severity of CCl4-induced liver injury and the progression of liver fibrosis, exerting a potential fibrolytic effect in the CCl4-induced fibrosis model.






Similar content being viewed by others
References
Akpolat N, Yahsi S, Godekmerdan A et al (2005) The value of α-SMA in the evaluation of hepatic fibrosis severity in hepatitis B infection and cirrhosis development: a histopathological and immunohistochemical study. Histopathology 47:276–280. doi:10.1111/j.1365-2559.2005.02226.x
Bataller R, Brenner D (2005) Science in medicine liver fibrosis. J Clin Invest 115:209–218. doi:10.1172/JCI200524282.The
Bissell DM, Roulot D, George J (2001) Transforming growth factor beta and the liver. Hepatology 34:859–867. doi:10.1053/jhep.2001.28457
Bosserhoff A, Hellerbrand C (2011) Obesity and fatty liver are “grease” for the machinery of hepatic fibrosis. Dig Dis 29:377–383. doi:10.1159/000329800
Carpino G, Morini S, Ginanni Corradini S et al (2005) Alpha-SMA expression in hepatic stellate cells and quantitative analysis of hepatic fibrosis in cirrhosis and in recurrent chronic hepatitis after liver transplantation. Dig Liver Dis 37:349–356. doi:10.1016/j.dld.2004.11.009
Chávez E, Segovia J, Shibayama M et al (2010) Antifibrotic and fibrolytic properties of celecoxib in liver damage induced by carbon tetrachloride in the rat. Liver Int 30:969–978. doi:10.1111/j.1478-3231.2010.02256.x
Cubero FJ, Urtasun R, Nieto N (2009) Alcohol and liver fibrosis. Semin Liver Dis 29:211–221. doi:10.1055/s-0029-1214376
Friedman SL (2008a) Hepatic stellate cells: protean, multifunctional, and enigmatic cells of the liver. Physiol Rev 88:125–172. doi:10.1152/physrev.00013.2007
Friedman SL (2008b) Hepatic fibrosis—overview. Toxicology 254:120–129. doi:10.1016/j.tox.2008.06.013
Friedman SL, Sheppard D, Duffield JS, Violette S (2013) Therapy for fibrotic diseases: nearing the starting line. Sci Transl Med 5:167sr1. doi:10.1126/scitranslmed.3004700
Gonzalez R, Ancheta O, Marquez M, Rodriguez S (1994) Hepatoprotective effects of diethylcarbamazine in acute liver damage induced by carbon tetrachloride in rats. Acta Pharmacol Sin 15:495–497
Gressner OA, Gao CA (2014) Monitoring fibrogenic progression in the liver. Clin Chim Acta 433:111–122. doi:10.1016/j.cca.2014.02.021
Gressner AM, Weiskirchen R, Breitkopf K, Dooley S (2002) Roles of TGF-beta in hepatic fibrosis. Front Biosci 7:d793–d807. doi:10.2741/gressner
Hemmann S, Graf J, Roderfeld M, Roeb E (2007) Expression of MMPs and TIMPs in liver fibrosis—a systematic review with special emphasis on anti-fibrotic strategies. J Hepatol 46:955–975. doi:10.1016/j.jhep.2007.02.003
Hewitt RI (1947) Experimental chemotherapy of filariasis; effect of piperazine derivatives against naturally acquired filarial infections in cotton rats and dogs. J Lab Clin Med 32:1304–1313
Hong IH, Park SJ, Goo MJ et al (2013) JNK1 and JNK2 regulate α-SMA in hepatic stellate cells during CCl4-induced fibrosis in the rat liver. Pathol Int 63:483–491. doi:10.1111/pin.12094
Iredale JP (2007) Models of liver fibrosis: exploring the dynamic nature of inflammation and repair in a solid organ. J Clin Invest 117:539–548. doi:10.1172/JCI30542
Jarčuška P, Janičko M, Veselíny E et al (2010) Circulating markers of liver fibrosis progression. Clin Chim Acta 411:1009–1017. doi:10.1016/j.cca.2010.04.009
Johnson GL, Johnson GL, Lapadat R, Lapadat R (2002) Mitogen-activated protein kinase pathways Mediated by ERK, JNK, and p38 protein kinases. Science (80-) 298:1911–1912
Kluwe J, Pradere JP, Gwak GY et al (2010) Modulation of hepatic fibrosis by c-Jun-N-terminal kinase inhibition. Gastroenterology 138:347–359. doi:10.1053/j.gastro.2009.09.015
Kong X, Horiguchi N, Mori M, Gao B (2012) Cytokines and STATs in liver fibrosis. Front Physiol 3:69. doi:10.3389/fphys.2012.00069
Kweon Y, Kweon Y, Goodman ZD et al (2001) Decreasing brogenesis: an immunohistochemical study of paired liver biopsies following lamivudine therapy for chronic hepatitis B. J Clin Invest 35:749–755
Lee UE, Friedman SL (2011) Mechanisms of hepatic fibrogenesis. Best Pract Res Clin Gastroenterol 25:195–206. doi:10.1016/j.bpg.2011.02.005
Leung T-M, Fung M-L, Liong EC et al (2011) Role of nitric oxide in the regulation of fibrogenic factors in experimental liver fibrosis in mice. Histol Histopathol 26:201–211
Lin X, Zhang S, Huang Q et al (2012) Protective effect of Fufang–Liu–Yue–Qing, a traditional Chinese herbal formula, on CCl4 induced liver fibrosis in rats. J Ethnopharmacol 142:548–556. doi:10.1016/j.jep.2012.05.040
Maizels RM, Denham DA (1992) Diethilcarbamazine (DEC): immunopharmacological interactions of an anti-filarial drug. Parasitology 105:849–860
McCrudden R, Iredale JP (2000) Liver fibrosis, the hepatic stellate cell and tissue inhibitors of metalloproteinases. Histol Histopathol 15:1159–1168
Mohammed FF, Pennington CJ, Kassiri Z et al (2005) Metalloproteinase inhibitor TIMP-1 affects hepatocyte cell cycle via HGF activation in murine liver regeneration. Hepatology 41:857–867. doi:10.1002/hep.20618
Mormone E, George J, Nieto N (2011) Molecular pathogenesis of hepatic fibrosis and current therapeutic approaches. Chem Biol Interact 193:225–231. doi:10.1016/j.cbi.2011.07.001
Parsons CJ, Bradford BU, Pan CQ et al (2004) Antifibrotic effects of a tissue inhibitor of metalloproteinase-1 antibody on established liver fibrosis in rats. Hepatology 40:1106–1115. doi:10.1002/hep.20425
Peixoto CA, Silva BS (2014) Anti-inflammatory effects of diethylcarbamazine: a review. Eur J Pharmacol 734C:35–41. doi:10.1016/j.ejphar.2014.03.046
Probst A, Dang T, Bochud M et al (2011) Role of Hepatitis C virus genotype 3 in liver fibrosis progression—a systematic review and meta-analysis. J Viral Hepat 18:745–759. doi:10.1111/j.1365-2893.2011.01481.x
Raman M, Chen W, Cobb MH (2007) Differential regulation and properties of MAPKs. Oncogene 26:3100–3112. doi:10.1038/sj.onc.1210392
Robinson MJ, Cobb MH (1997) Mitogen-activated protein kinase pathways. Curr Opin Cell Biol 9:180–186. doi:10.1016/S0955-0674(97)80061-0
Rocha SWS, De França MER, Rodrigues GB et al (2014) Diethylcarbamazine reduces chronic inflammation and fibrosis in carbon tetrachloride-(CCl4-) induced liver injury in mice. Mediators Inflamm 2014:1–15. doi:10.1155/2014/696383
Rodrigues GB, Rocha SWS, Dos Santos LAM et al (2015) Diethylcarbamazine: possible therapeutic alternative in the treatment of alcoholic liver disease in C57BL/6 mice. Clin Exp Pharmacol Physiol 42:369–379. doi:10.1111/1440-1681.12369
Santos Rocha SW, Oliveira Dos Santos AC, Dos Santos Silva B et al (2012a) Effects of diethylcarbamazine (DEC) on hepatocytes of C57BL/6 J mice submitted to protein malnutrition. J Food Drug Anal 20:524–531
Santos Rocha SW, Santos Silva B, Dos Santos Gomes FO et al (2012b) Effect of diethylcarbamazine on chronic hepatic inflammation induced by alcohol in C57BL/6 mice. Eur J Pharmacol 689:194–203. doi:10.1016/j.ejphar.2012.05.044
Sapkota GP (2013) The TGFβ-induced phosphorylation and activation of p38 mitogen-activated protein kinase is mediated by MAP3K4 and MAP3K10 but not TAK1. Open Biol 3:130067. doi:10.1098/rsob.130067
Schuppan D, Kim YO (2013) Review series evolving therapies for liver fi brosis. 123:1887–1901. doi:10.1172/JCI66028.The
Schuppan D, Ruehl M, Somasundaram R, Hahn EG (2001) Matrix as a modulator of hepatic fibrogenesis. Semin Liver Dis 21:351–372. doi:10.1055/s-2001-17556
Seki E, de Minicis S, Inokuchi S et al (2009) CCR2 promotes hepatic fibrosis in mice. Hepatology 50:185–197. doi:10.1002/hep.22952
Terpstra A (2001) Differences between humans and mice in efficacy of the body fat lowering effect of conjugated linoleic acid: role of metabolic rate. J Nutr 131:2067–2068
Tock J, Van Putten V, Stenmark KR, Nemenoff RA (2003) Induction of SM-α-actin expression by mechanical strain in adult vascular smooth muscle cells is mediated through activation of JNK and p38 MAP kinase. Biochem Biophys Res Commun 301:1116–1121. doi:10.1016/S0006-291X(03)00087-1
Tsukada S, Parsons CJ, Rippe RA (2006) Mechanisms of liver fibrosis. Clin Chim Acta 364:33–60. doi:10.1016/j.cca.2005.06.014
Xu R, Zhang Z, Wang F-S (2012) Liver fibrosis: mechanisms of immune-mediated liver injury. Cell Mol Immunol 9:296–301. doi:10.1038/cmi.2011.53
Xu J, Liu X, Koyama Y et al (2014) The types of hepatic myofibroblasts contributing to liver fibrosis of different etiologies. Front Pharmacol 5:1–12. doi:10.3389/fphar.2014.00167
Yao Q, Lin Y, Li X et al (2013) Curcumin ameliorates intrahepatic angiogenesis and capillarization of the sinusoids in carbon tetrachloride-induced rat liver fibrosis. Toxicol Lett 222:72–82. doi:10.1016/j.toxlet.2013.06.240
Yata Y, Gotwals P, Koteliansky V, Rockey DC (2002) Dose-dependent inhibition of hepatic fibrosis in mice by a TGF-beta soluble receptor: implications for antifibrotic therapy. Hepatology 35:1022–1030. doi:10.1053/jhep.2002.32673
Yoshiji H, Kuriyama S, Yoshii J et al (2002) Tissue inhibitor of metalloproteinases-1 attenuates spontaneous liver fibrosis resolution in the transgenic mouse. Hepatology 36:850–860. doi:10.1016/S0270-9139(02)00091-5
Zhang YE (2009) Non-Smad pathways in TGF-β signaling. Cell Res 19:128–139. doi:10.1038/cr.2008.328.Non-Smad
Zhao L (2003) Beneficial effects of phosphodiesterase 5 inhibition in pulmonary hypertension are influenced by natriuretic peptide activity. Circulation 107:234–237. doi:10.1161/01.CIR.0000050653.10758.6B
Acknowledgements
This study was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Universidade Federal de Pernambuco (UFPE) and the Centro de Pesquisa Aggeu Magalhães (CPqAM/FIOCRUZ) in Recife, Brazil.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
França, M.E.R., Rocha, S.W.S., Oliveira, W.H. et al. Diethylcarbamazine attenuates the expression of pro-fibrogenic markers and hepatic stellate cells activation in carbon tetrachloride-induced liver fibrosis. Inflammopharmacol 26, 599–609 (2018). https://doi.org/10.1007/s10787-017-0329-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-017-0329-0