Abstract
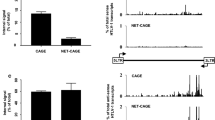
Long terminal repeats (LTRs) of human endogenous retroviruses (HERVs) located near or within genes might affect their expression. We used the KIAA1245/NBPF human gene subfamily in an attempt to assess the regulatory potential of HERV LTRs. The subfamily includes five closely related paralogous genes: three of them contain an LTR in the second intron, and two genes lack it. Earlier we reported that the second and third exons of only LTR-containing genes of this subfamily could be detected in mature mRNAs of various cell lines and human tissues. The corresponding parts of mRNA of LTR-lacking genes analyzed in our study were absent from EST libraries, but other fragments of their mRNAs were available in EST databases. For a more unbiased view on the correlation between gene transcription and the intronic LTRs, in the present work we analyzed non-spliced pre-mRNA thus avoiding splicing effects. Based on RT–PCR analysis, we demonstrated that the KIAA1245/NBPF LTR-lacking gene AL592309/NBPF3 was transcriptionally active, but the LTR-containing genes showed significantly higher transcription levels. The data are in agreement with the suggestion that HERV-K LTRs within the second intron of the KIAA1245/NBPF subfamily genes might affect their transcriptional activity. However, it still remains to be investigated whether the revealed effect is due just to the LTR insertion or other factors are responsible for the difference.



Similar content being viewed by others
Abbreviations
- RE:
-
Retroelement
- LTR:
-
Long terminal repeat
- HERV:
-
Human endogenous retrovirus
- HERV-K:
-
The K family of human endogenous retroviruses
- Mya:
-
Million of years ago
References
Abrarova ND et al (2010) Functional analysis of the HERV-K LTR residing in the KIAA1245/NBPF subfamily genes. Mol Biol 44(4):552–558
Agrawal A et al (1998) Transposition mediated by RAG1 and RAG2 and its implications for the evolution of the immune system. Nature 394(6695):744–751
Bannert N, Kurth R (2004) Retroelements and the human genome: new perspectives on an old relation. Proc Natl Acad Sci USA 101(Suppl 2):14572–14579
Beauregard A et al (2008) The take and give between retrotransposable elements and their hosts. Annu Rev Genet 42:587–617
Bernard M (1993) Molecular cloning software: gene runner 1.11 for windows. Biotechnol Softw 1993(10):4–7
Brandt J et al (2005) Transposable elements as a source of genetic innovation: expression and evolution of a family of retrotransposon-derived neogenes in mammals. Gene 345(1):101–111
Brosius J (1999) RNAs from all categories generate retrosequences that may be exapted as novel genes or regulatory elements. Gene 238(1):115–134
Butler B (1994) Nucleic acid sequence analysis software packages. Curr Opin Biotechnol 5(1):19–23
Buzdin A et al (2006) At least 50% of human-specific HERV-K (HML-2) long terminal repeats serve in vivo as active promoters for host nonrepetitive DNA transcription. J Virol 80(21):10752–10762
Cordaux R, Batzer MA (2009) The impact of retrotransposons on human genome evolution. Nat Rev Genet 10(10):691–703
Deininger PL, Batzer MA (2002) Mammalian retroelements. Genome Res 12(10):1455–1465
Diskin SJ et al (2009) Copy number variation at 1q21.1 associated with neuroblastoma. Nature 459(7249):987–991
Domansky AN et al (2000) Solitary HERV-K LTRs possess bi-directional promoter activity and contain a negative regulatory element in the U5 region. FEBS Lett 472(2–3):191–195
Doxiadis GG et al (2008) Impact of endogenous intronic retroviruses on major histocompatibility complex class II diversity and stability. J Virol 82(13):6667–6677
Elkahloun AG et al (1996) Molecular cytogenetic characterization and physical mapping of 12q13–15 amplification in human cancers. Genes Chromosomes Cancer 17(4):205–214
Elrouby N, Bureau TE (2001) A novel hybrid open reading frame formed by multiple cellular gene transductions by a plant long terminal repeat retroelement. J Biol Chem 276(45):41963–41968
Fablet M et al (2006) Ongoing loss of the tirant transposable element in natural populations of Drosophila simulans. Gene 375:54–62
Femino AM et al (1998) Visualization of single RNA transcripts in situ. Science 280(5363):585–590
Fujita PA et al (2010) UCSC genome browser database: update 2011. Nucleic Acids Res 39:D876–D882
Gifford R, Tristem M (2003) The evolution, distribution and diversity of endogenous retroviruses. Virus Genes 26(3):291–315
Han JS, Boeke JD (2005) LINE-1 retrotransposons: modulators of quantity and quality of mammalian gene expression? Bioessays 27(8):775–784
Hedges DJ, Batzer MA (2005) From the margins of the genome: mobile elements shape primate evolution. Bioessays 27(8):785–794
Hua-Van A et al (2005) Abundance, distribution and dynamics of retrotransposable elements and transposons: similarities and differences. Cytogenet Genome Res 110(1–4):426–440
Ikeda Y et al (2001) Novel compound heterozygous mutations for lipoprotein lipase deficiency. A G-to-T transversion at the first position of exon 5 causing G154 V missense mutation and a 5’ splice site mutation of intron 8. J Lipid Res 42(7):1072–1081
Illarionova AE et al (2007) Only those genes of the KIAA1245 gene subfamily that contain HERV(K) LTRs in their introns are transcriptionally active. Virology 358(1):39–47
Johnson M et al (2008) NCBI BLAST: a better web interface. Nucleic Acids Res 36:W5–W9
Kazazian HH Jr (2004) Mobile elements: drivers of genome evolution. Science 303(5664):1626–1632
Kazazian HH Jr, Moran JV (1998) The impact of L1 retrotransposons on the human genome. Nat Genet 19(1):19–24
King MC, Wilson AC (1975) Evolution at two levels in humans and chimpanzees. Science 188(4184):107–116
Kurth R, Bannert N (2010) Beneficial and detrimental effects of human endogenous retroviruses. Int J Cancer 126(2):306–314
Lander ES et al (2001) Initial sequencing and analysis of the human genome. Nature 409(6822):860–921
Leib-Mosch C, Seifarth W (1995) Evolution and biological significance of human retroelements. Virus Genes 11(2–3):133–145
Lu G, Moriyama EN (2004) VectorNTI, a balanced all-in one sequence analysis suite. Brief Bioinform 5(4):378–388
Mager DL et al (1999) Endogenous retroviruses provide the primary polyadenylation signal for two new human genes (HHLA2 and HHLA3). Genomics 59(3):255–263
Mayer J, Meese E et al (2005) Human endogenous retroviruses in the primate lineage and their influence on host genomes. Cytogenet Genome Res 110(1–4):448–456
McCarthy C (1997) Chromas, Version 1.41. Griffith University, Brisbane
Medstrand P et al (2001) Long terminal repeats are used as alternative promoters for the endothelin B receptor and apolipoprotein C-I genes in humans. J Biol Chem 276(3):1896–1903
Meza-Zepeda LA et al (2002) Positional cloning identifies a novel cyclophilin as a candidate amplified oncogene in 1q21. Oncogene 21(14):2261–2269
Nagase T et al (1998) Prediction of the coding sequences of unidentified human genes. XII. The complete sequences of 100 new cDNA clones from brain which code for large proteins in vitro. DNA Res 5(6):355–364
Petroziello J et al (2004) Suppression subtractive hybridization and expression profiling identifies a unique set of genes overexpressed in non-small-cell lung cancer. Oncogene 23(46):7734–7745
Ruda VM et al (2004) Tissue specificity of enhancer and promoter activities of a HERV-K(HML-2) LTR. Virus Res 104(1):11–16
Sakuraba H et al (1992) Invariant exon skipping in the human alpha-galactosidase A pre-mRNA: Ag + 1 to t substitution in a 5′-splice site causing Fabry disease. Genomics 12(4):643–650
Smit AFA, Hubley R, Green P (1996–2010) RepeatMasker Open-3.0. http://repeatmasker.org
Stefansson H et al (2008) Large recurrent microdeletions associated with schizophrenia. Nature 455(7210):232–236
Stone JL et al (2008) Rare chromosomal deletions and duplications increase risk of schizophrenia. Nature 455(7210):237–241
Sverdlov ED (1998) Perpetually mobile footprints of ancient infections in human genome. FEBS Lett 428(1–2):1–6
Sverdlov ED (2000) Retroviruses and primate evolution. Bioessays 22(2):161–171
Urnovitz HB, Murphy WH (1996) Human endogenous retroviruses: nature, occurrence, and clinical implications in human disease. Clin Microbiol Rev 9(1):72–99
van de Lagemaat LN et al (2003) Transposable elements in mammals promote regulatory variation and diversification of genes with specialized functions. Trends Genet 19(10):530–536
Vandepoele K, van Roy F (2007) Insertion of an HERV(K) LTR in the intron of NBPF3 is not required for its transcriptional activity. Virology 362(1):1–5
Vandepoele K et al (2005) A novel gene family NBPF: intricate structure generated by gene duplications during primate evolution. Mol Biol Evol 22(11):2265–2274
Vandepoele K et al (2008) A constitutional translocation t(1;17)(p36.2;q11.2) in a neuroblastoma patient disrupts the human NBPF1 and ACCN1 genes. PLoS One 3(5):e2207
Vandepoele K et al (2009) The NBPF1 promoter has been recruited from the unrelated EVI5 gene before simian radiation. Mol Biol Evol 26(6):1321–1332
Venter JC et al (2001) The sequence of the human genome. Science 291(5507):1304–1351
Vinogradova TV et al (2002) A new KIAA1245 gene family with or without HERV-K LTRs in their introns. Bioorg Khim 28(4):346–350
Walsh T et al (2008) Rare structural variants disrupt multiple genes in neurodevelopmental pathways in schizophrenia. Science 320(5875):539–543
Xing J et al (2006) Emergence of primate genes by retrotransposon-mediated sequence transduction. Proc Natl Acad Sci USA 103(47):17608–17613
Acknowledgments
The authors are grateful to B. Glotov for critical reading of the manuscript, to V. K. Potapov and N. V. Skaptsova for synthesis of oligonucleotides, as well as to the Interinstitute “Genom” Center for DNA sequencing. We also thank the “Evrogen” company for synthesis of oligonucleotides and DNA sequencing. The study was supported by the Russian Presidential Program “Leading Scientific Schools” (grant 5638.2010.4) and grant of the Presidium RAS Program “Molecular and Cell Biology”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abrarova, N., Simonova, L., Vinogradova, T. et al. Different transcription activity of HERV-K LTR-containing and LTR-lacking genes of the KIAA1245/NBPF gene subfamily. Genetica 139, 733–741 (2011). https://doi.org/10.1007/s10709-011-9577-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-011-9577-x