Abstract
Background
Trinitrobenzenesulfonic acid (TNBS)-induced colitis is one of the most widely used experimental colitis models. However, there is no standard procedure for inducing colitis by TNBS because it is difficult to achieve a uniform distribution of colitis. We have developed a modified method of murine TNBS-induced colitis that involves inhalation anesthesia with sevoflurane combined with both single and repeated TNBS administrations.
Aims
To compare the usefulness of our newly developed method for inducing murine TNBS-induced colitis with that of conventional intraperitoneal anesthesia.
Methods
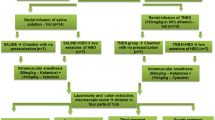
TNBS in ethanol was administered to C57BL/6J mice held in an inverted vertical position either under continuous inhalation anesthesia with sevoflurane, in accordance with our newly developed method, or by intraperitoneal injection with 2.5 % avertin, in accordance with the conventional procedure. Body weight change, cytokine profile, and histological findings were examined during the course of colitis.
Results
The dispersion of anesthesia time, TNBS retention time, and nadir weight during the course of colitis was decreased using the newly developed method compared with the conventional procedure. Optimization of the modified TNBS-induced colitis, as evidenced by the predominant expression of Th1 and Th17 cytokines on day 7, was attained by the injection of 2.25 mg TNBS in 55 % ethanol. Regulation of the TNBS retention time using inhalation anesthesia with sevoflurane allowed strict control of the disease severity of TNBS-induced colitis. Using the modified method we were also able to develop a chronic TNBS-induced colitis model by repeated TNBS administration without excessive mortality of the mice.
Conclusions
Our modified method for murine TNBS-induced colitis using continuous inhalation anesthesia with sevoflurane provides a better experimental colitis model following both single and repeated TNBS administrations.







Similar content being viewed by others
Abbreviations
- CIA:
-
Continuous inhalation anesthesia
- IBD:
-
Inflammatory bowel disease
- IFN-γ:
-
Interferon gamma
- IL:
-
Interleukin
- IPA:
-
Intraperitoneal anesthesia
- Th:
-
T helper
- TNBS:
-
Trinitrobenzenesulfonic acid
- TNF-α:
-
Tumor necrosis factor alpha
References
Pizarro TT, Arseneau KO, Bamias G, Cominelli F. Mouse models for the study of Crohn’s disease. Trends Mol Med. 2003;9:218–222.
Wirtz S, Neurath MF. Mouse models of inflammatory bowel disease. Adv Drug Deliv Rev. 2007;59:1073–1083.
Jurjus AR, Khoury NN, Reimund JM. Animal models of inflammatory bowel disease. J Pharmacol Toxicol Methods. 2004;50:81–92.
Kawada M, Arihiro A, Mizoguchi E. Insights from advances in research of chemically induced experimental models of human inflammatory bowel disease. World J Gastroenterol. 2007;13:5581–5593.
Wirtz S, Neufert C, Weigmann B, Neurath MF. Chemically induced mouse models of intestinal inflammation. Nat Protoc. 2007;2:541–546.
te Velde AA, Verstege MI, Hommes DW. Critical appraisal of the current practice in murine TNBS-induced colitis. Inflamm Bowel Dis. 2006;12:995–999.
Bouma G, Strober W. The immunological and genetic basis of inflammatory bowel disease. Nat Rev Immunol. 2003;3:521–533.
Neurath M, Fuss I, Strober W. TNBS-colitis. Int Rev Immunol. 2000;19:51–62.
Neurath MF, Fuss I, Kelsall BL, Stuber E, Strober W. Antibodies to interleukin 12 abrogate established experimental colitis in mice. J Exp Med. 1995;182:1281–1290.
Foligne B, Nutten S, Steidler L, et al. Recommendations for improved use of the murine TNBS-induced colitis model in evaluating anti-inflammatory properties of lactic acid bacteria: technical and microbiological aspects. Dig Dis Sci. 2006;51:390–400.
Fichtner-Feigl S, Fuss IJ, Young CA, et al. Induction of IL-13 triggers TGF-beta1-dependent tissue fibrosis in chronic 2,4,6-trinitrobenzene sulfonic acid colitis. J Immunol. 2007;178:5859–5870.
Fichtner-Feigl S, Young CA, Kitani A, Geissler EK, Schlitt HJ, Strober W. IL-13 signaling via IL-13R alpha2 induces major downstream fibrogenic factors mediating fibrosis in chronic TNBS colitis. Gastroenterology. 2008;135:2003–20013, 2013 e1–7.
Lawrance IC, Wu F, Leite AZ, et al. A murine model of chronic inflammation-induced intestinal fibrosis down-regulated by antisense NF-kappa B. Gastroenterology. 2003;125:1750–1761.
Cesarovic N, Nicholls F, Rettich A, et al. Isoflurane and sevoflurane provide equally effective anaesthesia in laboratory mice. Lab Anim. 2010;44:329–336.
Michel F, Constantin JM. Sevoflurane inside and outside the operating room. Expert Opin Pharmacother. 2009;10:861–873.
Young CJ, Apfelbaum JL. Inhalational anesthetics: desflurane and sevoflurane. J Clin Anesth. 1995;7:564–577.
Sakai EM, Connolly LA, Klauck JA. Inhalation anesthesiology and volatile liquid anesthetics: focus on isoflurane, desflurane, and sevoflurane. Pharmacotherapy. 2005;25:1773–1788.
Galitovskiy V, Qian J, Chernyavsky AI, et al. Cytokine-induced alterations of alpha7 nicotinic receptor in colonic CD4 T cells mediate dichotomous response to nicotine in murine models of Th1/Th17-versus Th2-mediated colitis. J Immunol. 2011;187:2677–2687.
Brenna O, Furnes MW, Drozdov I, et al. Relevance of TNBS-colitis in rats: a methodological study with endoscopic, historical and transcripttomic characterization and correlation to IBD. PLoS ONE. 2013;8:e54543.
Fiorucci S, Mencarelli A, Palazzetti B, et al. Importance of innate immunity and collagen binding integrin alpha1beta1 in TNBS-induced colitis. Immunity. 2002;17:769–780.
Sugimoto K, Hanai H, Tozawa K, et al. Curcumin prevents and ameliorates trinitrobenzene sulfonic acid-induced colitis in mice. Gastroenterology. 2002;123:1912–1922.
Koon HW, Shih D, Karagiannides I, et al. Substance P modulates colitis-associated fibrosis. Am J Pathol. 2010;177:2300–2309.
Fortin G, Raymond M, Van VQ, et al. A role for CD47 in the development of experimental colitis mediated by SIRPalpha + CD103-dendritic cells. J Exp Med. 2009;206:1995–2011.
He Y, Lin LJ, Zheng CQ, Jin Y, Lin Y. Cytokine expression and the role of Thl7 cells in mice colitis. Hepatogastroenterology. 2012;59:1809–1813.
Alex P, Zachos NC, Nguyen T, et al. Distinct cytokine patterns identified from multiplex profiles of murine DSS and TNBS-induced colitis. Inflamm Bowel Dis. 2009;15:341–352.
Ten Hove T, Corbaz A, Amitai H, et al. Blockade of endogenous IL-18 ameliorates TNBS-induced colitis by decreasing local TNF-alpha production in mice. Gastroenterology. 2001;121:1372–1379.
Zhao J, de Vera J, Narushima S, et al. R-spondin1, a novel intestinotrophic mitogen, ameliorates experimental colitis in mice. Gastroenterology. 2007;132:1331–1343.
Kanai T, Watanabe M, Okazawa A, et al. Macrophage-derived IL-18-mediated intestinal inflammation in the murine model of Crohn’s disease. Gastroenterology. 2001;121:875–888.
Elson CO, Beagley KW, Sharmanov AT, et al. Hapten-induced model of murine inflammatory bowel disease: mucosa immune responses and protection by tolerance. J Immunol. 1996;157:2174–2185.
Mariman R, Kremer B, van Erk M, Lagerweij T, Koning F, Nagelkerken L. Gene expression profiling identifies mechanisms of protection to recurrent trinitrobenzene sulfonic acid colitis mediated by probiotics. Inflamm Bowel Dis. 2012;18:1424–1433.
Ma Y, Guan Q, Bai A, et al. Targeting TGF-beta1 by employing a vaccine ameliorates fibrosis in a mouse model of chronic colitis. Inflamm Bowel Dis. 2010;16:1040–1050.
Wu F, Chakravarti S. Differential expression of inflammatory and fibrogenic genes and their regulation by NF-kappaB inhibition in a mouse model of chronic colitis. J Immunol. 2007;179:6988–7000.
Boirivant M, Amendola A, Butera A, et al. A transient breach in the epithelial barrier leads to regulatory T-cell generation and resistance to experimental colitis. Gastroenterology. 2008;135:1612 e5–1623 e5.
Acknowledgments
This research was supported by Grant-in-Aid for Scientific Research (C) 21590808 and 25460948 from the Ministry of Education, Culture, Sports, Science and Technology, Japan. We are grateful to Soichiro Mimuro for technical assistance regarding anesthesia in mice.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10620_2013_3023_MOESM2_ESM.tif
Supplemental Fig. 1 Evaluation of single TNBS administration in BALB/c mice using continuous inhalation anesthesia. Female BALB/c mice were anesthetized by continuous ventilation with sevoflurane, and TNBS colitis was induced according to the protocol. On day 0, 1.25 mg of TNBS in 50 % ethanol was administered (n = 12). (a) Time course of body weight change during colitis. (b) Time course of TNBS retention volume. ©) Hematoxylin and eosin staining and Masson trichrome staining of representative colon sections on day 0 (control), 3, and 7 after TNBS administration. 10× magnification in upper section; 40× magnification in lower section. Histological findings on day 3 showed distortion of crypts, loss of goblet cells, and focal ulcers accompanied by edema in the mucosal or submucosal regions. By day 7, disrupted crypts and goblet cells regenerated in most parts of the mucosa, but infiltration of mononuclear cells was still prominent in the stromal tissue of basal mucosal areas. Trichrome-stained collagen deposits were observed in the stromal tissue in the mucosa and submucosa on day 3 and 7, respectively. (TIFF 2,885 kb)
Rights and permissions
About this article
Cite this article
Terai, T., Osawa, S., Tani, S. et al. Induction of Murine TNBS Colitis Is Strictly Controlled by a Modified Method Using Continuous Inhalation Anesthesia with Sevoflurane. Dig Dis Sci 59, 1415–1427 (2014). https://doi.org/10.1007/s10620-013-3023-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-013-3023-0