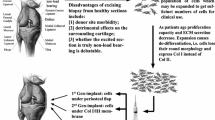
Abstract
Over the years, surgical strategies have been developed in hope of full regeneration of the injured cartilage. In our study, we aimed to develop an optimized chondrocyte culture isolation technique as an active ingredient of a standardized autologous chondrocte implantation product, which is able to maintain the phenotype along with the molecular features of the cartilage. We compared different enzymes, which suggested optimal performance with collagenase type II at 5 mg/ml concentration. Thereafter, we observed that COL2 and GAG expression is substantially reduced with passaging. There was a need to omit passaging to reach the optimal isolation method. We then tested various growth factors and media in order to maintain the natural character of chondrocytes. Our study also suggested the highest COL2 and GAG expressions with the highest recovery in the presence of Advanced DMEM. Autologous chondrocyte implantation manufacturing approval was recently received from the national competent authority, making it possible to utilize the process engineering protocol developed with this study at our Tissue and Cell Manufacturing Center as a part of the autologous chondrocyte implantation manufacturing standard operation procedure (SOP).









Similar content being viewed by others
References
Adkisson HD, Milliman C, Zhang X, Mauch K, Maziarz RT, Streeter PR (2010) Immune evasion by neocartilage-derived chondrocytes: implications for biologic repair of joint articular cartilage. Stem Cell Res. 4(1):57–68. https://doi.org/10.1016/j.scr.2009.09.004
Akgun I, Unlu MC, Erdal OA, Ogut T, Erturk M, Ovali E (2015) Matrix-induced autologous mesenchymal stem cell implantation versus matrix-induced autologous chondrocyte implantation in the treatment of chondral defects of the knee: a 2-year randomized study. Arch Orthop Trauma Surg 135(2):251–263. https://doi.org/10.1007/s00402-014-2136-z
Atilla E, Kilic P, Gurman G (2018) Cellular therapies: day by day, all the way. Transfus Apher Sci. 57:187–196. https://doi.org/10.1016/j.transci.2018.04.019
Demoor M, Ollitrault D, Gomez-Leduc T, Bouyoucef M, Hervieu M, Lafont J (2014) Cartilage tissue engineering: molecular control of chondrocyte differentiation for proper cartilage matrix reconstruction. Biochim Biophys Acta 1840:2414–2440. https://doi.org/10.1016/j.bbagen.2014.02.030
EMA (2017) ChondroCelect: EPAR—All authorised presentations. https://www.ema.europa.eu/en/documents/all-authorised-presentations/chondrocelect-epar-all-authorised-presentations_en.pdf. Accessed 8 Nov 2019
Gosset M, Berenbaum F, Thirion S, Jacques C (2008) Primary culture and phenotyping of murine chondrocytes. Nat Protoc. https://doi.org/10.1038/nprot.2008.95
Huang BJ, Hu JC, Athanasiou KA (2016) Cell-based tissue engineering strategies used in the clinical repair of articular cartilage. Biomaterials 98:1–22. https://doi.org/10.1016/j.biomaterials.2016.04.018
Ishibashi M, Hikita A, Fujihara Y, Takato T, Hoshi K (2017) Human auricular chondrocytes with high proliferation rate show high production of cartilage matrix. Regen Ther. 6:21–28. https://doi.org/10.1016/j.reth.2016.11.001
Lau TT, Peck Y, Huang W, Wang DA (2015) Optimization of chondrocyte isolation and phenotype characterization for cartilage tissue engineering. Tissue Engineering Part C: Methods. 21(2):105–111. https://doi.org/10.1089/ten.TEC.2014.0159
Marcacci M, Kon E, Zaffagnini S (2007) Arthroscopic second generation autologous chondrocyte implantation. Knee Surg Sports Traumatol Arthrosc 15:610–619. https://doi.org/10.1177/0363546508323256
Naranda J, Gradišnik L, Gorenjak M, Vogrin M, Maver U (2017) Isolation and characterization of human articular chondrocytes from surgical waste after total knee arthroplasty (TKA). PeerJ. 21(5):e3079. https://doi.org/10.7717/peerj.3079
Ochi M, Uchio Y, Tobita M, Kuriwaka M (2001) Current concepts in tissue engineering technique for repair of cartilage defect. Artif Organs 25:172–179. https://doi.org/10.1046/j.1525-1594.2001.025003172.x
Oseni AO, Butler PE, Seifalian AM (2013) Optimization of chondrocyte isolation and characterization for large-scale cartilage tissue engineering. J Surg Res 181(1):41–48. https://doi.org/10.1016/j.jss.2012.05.087
Shui W, Yin L, Luo J, Li R, Zhang W, Zhang J (2013) Characterization of chondrocyte scaffold carriers for cell-based gene therapy in articular cartilage repair. J Biomed Mater Res A. 101(12):3542–3550. https://doi.org/10.1002/jbm.a.34661
Vedicherla S, Buckley CT (2017) Rapid chondrocyte isolation for tissue engineering applications: the effect of enzyme concentration and temporal exposure on the matrix forming capacity of nasal derived chondrocytes. Biomed Res Int. https://doi.org/10.1155/2017/2395138
Wakitani S, Kawaguchi A, Tokuhara Y, Takaoka K (2008) Present status and future direction for articular cartilage repair. J Bone Miner Metab 26:115–122. https://doi.org/10.1007/s00774-007-0802-8
Yonenaga K, Nishizawa S, Nakagawa T, Fujihara Y, Asawa Y, Hikita A (2017) Optimal conditions of collagenase treatment for isolation of articular chondrocytes from aged human tissues. Regen Ther 6:9–14. https://doi.org/10.1016/j.reth.2016.08.001
Acknowledgements
We thank Assoc. Prof. Dr. Kerem Başarır, Assoc. Prof. Dr. Ramazan Akmeşe and Sp. Dr. Batu Malatyalı at Ankara University School of Medicine Department of Orthopedics and Traumatology for their technical and clinical support in cartilage tissue harvest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Pelin Kilic, Cansu Gurcan, Gunhan Gurman and Acelya Yilmazer. The first draft of the manuscript was written by Pelin Kilic and Cansu Gurcan, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical statement
The research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki, with our University’s Ethics Committee Approval No. 15-1007-18.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kilic, P., Gurcan, C., Gurman, G. et al. Understanding factors affecting human chondrocyte culturing: an experimental study. Cell Tissue Bank 21, 585–596 (2020). https://doi.org/10.1007/s10561-020-09847-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-020-09847-y