Abstract
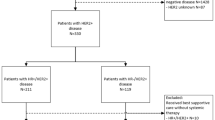
Biomarkers of breast cancer such as hormone receptors (HR) and human epidermal growth factor 2 (HER2) can be altered after neoadjuvant chemotherapy (NAC). However, whether the conversion of these receptors affects the prognosis of patients remains to be determined. We sought to evaluate the prognostic value of HR and HER2 receptors before and after NAC and to analyze their clinical implications. Relevant studies were used to calculate the pooled hazard ratios, 95% confidence interval (95% CI). This meta-analysis included eight studies with 2847 patients. Compared to patients with HR+ → +, patients with HR+ → − have shorter disease free survival (DFS) (hazard ratio = 2.64, 95% CI 1.86–3.75) and overall survival (OS) (hazard ratio = 2.99, 95% CI 1.97–4.54). Furthermore, patients with HR− → + tend to achieve better DFS (hazard ratio = 0.83, 95% CI 0.60–1.17) compared to patients with HR− → −. Patients with HR− → + gain better OS (hazard ratio = 0.67, 95% CI 0.46–0.99) compared to patients exhibiting HR− → −. When comparing patients with HER2+ → − to patients with HER2+ → +, patients with HER2+ → − tended to achieve better DFS (hazard ratio = 1.65, 95% CI 1.08–2.53) though results for OS (hazard ratio = 1.16, 95% CI 0.54–2.49) were not statistically significant. Our data strongly support the need for redetection of HR and HER2 receptor status of surgical sample following neoadjuvant therapy. Changes in HR status induced by NAC can be used as a prognostic factor in breast cancer patients for predicting both OS and DFS. HER2 change may also be valuable for predicting prognosis. Further research should explore therapeutic strategies for those presenting receptor status conversion.




Similar content being viewed by others
References
Fisher B, Bryant J, Wolmark N et al (1998) Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J Clin Oncol Off J Am Soc Clin Oncol 16(8):2672–2685
Mauri D, Pavlidis N, Ioannidis JPA (2005) Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. Breast Dis A Year Book Q 97(3):188–194
Von MG, Untch M, Blohmer JU et al (2012) Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol Off J Am Soc Clin Oncol 30(30):1796–1804
Kong X, Moran MS, Zhang N et al (2011) Meta-analysis confirms achieving pathological complete response after neoadjuvant chemotherapy predicts favourable prognosis for breast cancer patients. Eur J Cancer 47(14):2084–2090
Houssami N, Macaskill P, Minckwitz GV et al (2012) Meta-analysis of the association of breast cancer subtype and pathologic complete response to neoadjuvant chemotherapy. Eur J Cancer 48(18):3342–3354
Morris DM, Edwards J, Gelder F (1991) Hormonal receptors in locally advanced breast cancer: change with response to neoadjuvant chemotherapy? J Surg Oncol 46(3):156–158
Hirata T, Shimizu C, Yonemori K et al (2009) Change in the hormone receptor status following administration of neoadjuvant chemotherapy and its impact on the long-term outcome in patients with primary breast cancer. Br J Cancer 101(9):1529–1536
Neubauer H, Gall C, Vogel U et al (2008) Changes in tumour biological markers during primary systemic chemotherapy (PST). Anticancer Res 28(3B):1797
Ven SVD, Smit VTHBM, Dekker TJA et al (2011) Discordances in ER, PR and HER2 receptors after neoadjuvant chemotherapy in breast cancer. Cancer Treat Rev 37(6):422–430
Zhang N, Moran MS, Huo Q et al (2011) The hormonal receptor status in breast cancer can be altered by neoadjuvant chemotherapy: a meta-analysis. Cancer Invest 29(9):594
Niikura N, Liu J, Hayashi N et al (2011) Loss of human epidermal growth factor receptor 2 (HER2) expression in metastatic sites of HER2-overexpressing primary breast tumors. J Clin Oncol 30(6):593–599
Jin X, Jiang YZ, Chen S et al (2015) Prognostic value of receptor conversion after neoadjuvant chemotherapy in breast cancer patients: a prospective observational study. Oncotarget 6(11):9600–9611
Tacca O, Penault-Llorca F, Abrial C et al (2007) Changes in and prognostic value of hormone receptor status in a series of operable breast cancer patients treated with neoadjuvant chemotherapy. Oncologist 12(6):636–643
Wang RX, Chen S, Jin X et al (2016) Weekly paclitaxel plus carboplatin with or without trastuzumab as neoadjuvant chemotherapy for HER2-positive breast cancer: loss of HER2 amplification and its impact on response and prognosis. Breast Cancer Res Treat 161:1–9
Guarneri V, Dieci MV, Barbieri E et al (2013) Loss of HER2 positivity and prognosis after neoadjuvant therapy in HER2-positive breast cancer patients. Ann Oncol 24(12):2990–2994
Stroup DF, Berlin JA, Morton SC et al (2008) Meta-analysis of observational studies in epidemiology: a proposal for reporting. J Am Med Assoc 283(15):2008–2012
Tierney JF, Stewart LA, Ghersi D et al (2007) Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 8(1):16
Tan QX, Qin QH, Yang WP et al (2014) Prognostic value of hormone receptor status conversion following neoadjuvant chemotherapy in a series of operable breast cancer patients. Int J Clin Exp Pathol 7(7):4
Lim SK, Lee MH, Park IH et al (2015) Impact of molecular subtype conversion of breast cancers after neoadjuvant chemotherapy on clinical outcome. Cancer Res Treat 48(1):133–141
Niikura N, Tomotaki A, Miyata H et al (2015) Changes in tumor expression of HER2 and hormone receptors status after neoadjuvant chemotherapy in 21 755 patients from the, Japanese breast cancer registry. Ann Oncol Off J Eur Soc Med Oncol 27(3):480
Mittendorf EA, Wu Y, Scaltriti M et al (2009) Loss of HER2 amplification following trastuzumab-based neoadjuvant systemic therapy and survival outcomes. Clin Cancer Res 15(23):7381–7388
Yoshida A, Hayashi N, Suzuki K et al (2017) Change in HER2 status after neoadjuvant chemotherapy and the prognostic impact in patients with primary breast cancer. Surg Oncol 116(8):1021–1028
Wolff AC, Hammond MEH, Hicks DG et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31(31):3997–4013
Wu JY, Chen WG, Chen XS et al (2014) Long-term outcomes following adjuvant endocrine therapy in breast cancer patients with a positive-to-negative change of hormone receptor status following neoadjuvant chemotherapy. Mol Clin Oncol 2(6):997–1002
Lower EE, Khan S, Kennedy D et al (2017) Discordance of the estrogen receptor and HER-2/neu in breast cancer from primary lesion to first and second metastatic site. Breast Cancer Targets Therapy 9:515–520
Lindström LS, Karlsson E, Wilking UM et al (2012) Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol Off J Am Soc Clin Oncol 30(21):2601–2608
Zardavas D, Irrthum A, Swanton C et al (2015) Clinical management of breast cancer heterogeneity. Nat Rev Clin Oncol 12(7):381–394
Liedtke C, Broglio K, Moulder S et al (2009) Prognostic impact of discordance between triple-receptor measurements in primary and recurrent breast cancer. Ann Oncol 20:1953–1958
Sidoni A, Scheibel M, Bellezza G (2005) Biopathologic profile of breast cancer core biopsy: is it always a valid method? Cancer Lett 218(1):117–121
Juo YY, Johnston FM, Zhang DY et al (2014) Prognostic value of CpG island methylator phenotype among colorectal cancer patients: a systematic review and meta-analysis. Ann Oncol Off J Eur Soc Med Oncol 25(12):2314–2327
Acknowledgements
This study was supported by grants from the National Key R&D Program of China No. 2016YFC0904900, National Natural Science Foundation (No. 81673509 and No. 81573504) of PR China, Beijing Municipal Natural Science Foundation (No. 7171012) and National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (No. 2017ZX09304028 and No. 2017ZX09101001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable- this article does not contain any studies with human participants performed by any of authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, C., Fan, H., Xiang, Q. et al. Prognostic value of receptor status conversion following neoadjuvant chemotherapy in breast cancer patients: a systematic review and meta-analysis. Breast Cancer Res Treat 178, 497–504 (2019). https://doi.org/10.1007/s10549-019-05421-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05421-7