Abstract
Background
Cerebrotendinous xanthomatosis (CTX) is a treatable bile acid disorder caused by mutations of CYP27A1. The pathogenesis of neurological damage has not been completely explained. Oral chenodeoxycholic acid (CDCA) can lead to clinical stabilization, but in a subgroup of patients the disease progresses despite treatment. In the present study, we aimed at clarifying cholesterol metabolism abnormalities and their response to CDCA treatment, in order to identify reliable diagnostic and prognostic markers and understand if differences exist between stable patients and those with neurological progression.
Methods
We enrolled 19 untreated CTX patients and assessed serum profile of bile acids intermediates, oxysterols, cholesterol, lathosterol, and plant sterols. Then we performed a long-term follow up during CDCA therapy, and compared biochemical data with neurological outcome.
Results
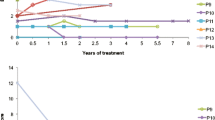
We observed increase of cholestanol, 7α-hydroxy-4-cholesten-3-one (7αC4), lathosterol, and plant sterols, whereas 27-hydroxycholesterol (27-OHC) was extremely low or absent. CDCA treatment at a daily dose of 750 mg normalized all biochemical parameters except for 7αC4 which persisted slightly higher than normal in most patients, and 27-OHC which was not modified by therapy. Biochemical evaluation did not reveal significant differences between stable and worsening patients.
Discussion
Cholestanol and 7αC4 represent important markers for CTX diagnosis and monitoring of therapy. Treatment with CDCA should aim at normalizing serum 7αC4 as well as cholestanol, since 7αC4 better mirrors 7α-hydroxylation rate and is thought to be correlated with cholestanol accumulation in the brain. Assessment of serum 27-OHC is a very good tool for biochemical diagnosis at any stage of disease. Lathosterol and plant sterols should be considered as additional markers for diagnosis and monitoring of therapy. Further studies including long-term assessment of bile acid intermediates in cerebrospinal fluid are needed in patients who show clinical progression despite treatment.


Similar content being viewed by others
References
Alessandrini L, Ciuffreda P, Santaniello E, Terraneo G (2004) Clemmensen reduction of diosgenin and kryptogenin: synthesis of [16,16,22,22,23,23-(2)H(6)]-(25R)-26-hydroxycholesterol. Steroids 69:789–794
Ali Z, Heverin M, Olin M et al (2013) On the regulatory role of side-chain hydroxylated oxysterols in the brain. Lessons from CYP27A1 transgenic and Cyp27a1(-/-) mice. J Lipid Res 54:1033–1043
Barkhof F, Verrips A, Wesseling P et al (2000) Cerebrotendinous xanthomatosis: the spectrum of imaging findings and the correlation with neuropathologic findings. Radiology 217:869–876
Båvner A, Shafaati M, Hansson M et al (2010) On the mechanism of accumulation of cholestanol in the brain of mice with a disruption of sterol 27-hydroxylase. J Lipid Res 51:2722–2730
Berginer VM, Berginer J, Korczyn AD, Tadmor R (1994) Magnetic resonance imaging in cerebrotendinous xanthomatosis: a prospective clinical and neuroradiological study. J Neurol Sci 122:102–108
Berginer VM, Gross B, Morad K et al (2009) Chronic diarrhea and juvenile cataracts: think cerebrotendinous xanthomatosis and treat. Pediatrics 123:143–147
Berginer VM, Salen G, Shefer S (1984) Long-term treatment of cerebrotendinous xanthomatosis with chenodeoxycholic acid. N Engl J Med 311:1649–1652
Bertolotti M, Del Puppo M, Gabbi C et al (2008) Correlation between plasma levels of 7alpha-hydroxy-4-cholesten-3-one and cholesterol 7alpha-hydroxylation rates in vivo in hyperlipidemic patients. Steroids 73:1197–1202
Björkhem I (2013) Cerebrotendinous xanthomatosis. Curr Opin Lipidol 24:283–287
Björkhem I, Diczfalusy U, Lövgren-Sandblom A et al (2014) On the formation of 7-ketocholesterol from 7-dehydrocholesterol in patients with CTX and SLO. J Lipid Res 55:1165–1172
Björkhem I, Hansson M (2010) Cerebrotendinous xanthomatosis: an inborn error in bile acid synthesis with defined mutations but still a challenge. Biochem Biophys Res Commun 396:46–49
Cali JJ, Hsieh CL, Francke U et al (1991) Mutations in the bile acid biosynthetic enzyme sterol 27-hydroxylase underlie cerebrotendinous xanthomatosis. J Biol Chem 266:7779–7783
Camilleri M, Nadeau A, Tremaine WJ et al (2009) Measurement of serum 7alpha-hydroxy-4-cholesten-3-one (or 7alphaC4), a surrogate test for bile acid malabsorption in health, ileal disease and irritable bowel syndrome using liquid chromatography-tandem mass spectrometry. Neurogastroenterol Motil 21:734–e743
Chen W, Chen G, Head DL, Mangelsdorf DJ, Russell DW (2007) Enzymatic reduction of oxysterols impairs LXR signaling in cultured cells and the livers of mice. Cell Metab 5:73–79
DeBarber AE, Connor WE, Pappu AS, Merkens LS, Steiner RD (2010) ESI-MS/MS quantification of 7alpha-hydroxy-4-cholesten-3-one facilitates rapid, convenient diagnostic testing for cerebrotendinous xanthomatosis. Clin Chim Acta 411:43–48
de Sain-van der Velden MG, Verrips A, Prinsen BH, de Barse M, Berger R, Visser G (2008) Elevated cholesterol precursors other than cholestanol can also be a hallmark for CTX. J Inherit Metab Dis 31:S387–S393
De Stefano N, Dotti MT, Mortilla M, Federico A (2001) Magnetic resonance imaging and spectroscopic changes in brains of patients with cerebrotendinous xanthomatosis. Brain 124:121–131
Del Puppo M, Kienle MG, Petroni ML, Crosignani A, Podda M (1998) Serum 27-hydroxycholesterol in patients with primary biliary cirrhosis suggests alteration of cholesterol catabolism to bile acids via the acidic pathway. J Lipid Res 39:2477–2482
Dzeletovic S, Breuer O, Lund E, Diczfalusy U (1995) Determination of cholesterol oxidation products in human plasma by isotope dilution-mass spectrometry. Anal Biochem 225:73–80
Ellis E, Axelson M, Abrahamsson A et al (2003) Feedback regulation of bile acid synthesis in primary human hepatocytes: evidence that CDCA is the strongest inhibitor. Hepatology 38:930–938
Fakheri RJ, Javitt NB (2012) 27-Hydroxycholesterol, does it exist? On the nomenclature and stereochemistry of 26-hydroxylated sterols. Steroids 77:575–577
Galli Kienle M, Anastasia M, Cighetti G, Galli G, Fiecchi A (1980) Studies on the 14 alpha-demethylation mechanism in cholesterol biosynthesis. Eur J Biochem 110:93–105
Ginanneschi F, Mignarri A, Mondelli M et al (2013) Polyneuropathy in cerebrotendinous xanthomatosis and response to treatment with chenodeoxycholic acid. J Neurol 260:268–274
Heverin M, Meaney S, Lütjohann D, Diczfalusy U, Wahren J, Björkhem I (2005) Crossing the barrier: net flux of 27-hydroxycholesterol into the human brain. J Lipid Res 46:1047–1052
Inoue K, Kubota S, Seyama Y (1999) Cholestanol induces apoptosis of cerebellar neuronal cells. Biochem Biophys Res Commun 256:198–203
Kempen HJ, Glatz JF, Gevers Leuven JA, van der Voort HA, Katan MB (1988) Serum lathosterol concentration is an indicator of whole-body cholesterol synthesis in humans. J Lipid Res 29:1149–1155
Kuriyama M, Fujiyama J, Kasama T, Osame M (1991) High levels of plant sterols and cholesterol precursors in cerebrotendinous xanthomatosis. J Lipid Res 32:223–229
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Leoni V, Caccia C (2011) Oxysterols as biomarkers in neurodegenerative diseases. Chem Phys Lipids 164:515–524
Liu J, Lu H, Lu YF et al (2014) Potency of individual bile acids to regulate bile acid synthesis and transport genes in primary human hepatocyte cultures. Toxicol Sci 141:538–546
Martini G, Mignarri A, Ruvio M et al (2013) Long-term bone density evaluation in cerebrotendinous xanthomatosis: evidence of improvement after chenodeoxycholic acid treatment. Calcif Tissue Int 92:282–286
Mignarri A, Dotti MT, Del Puppo M et al (2012) Cerebrotendinous xanthomatosis with progressive cerebellar vacuolation: six-year MRI follow-up. Neuroradiology 54:649–651
Mignarri A, Gallus GN, Dotti MT, Federico A (2014) A suspicion index for early diagnosis and treatment of cerebrotendinous xanthomatosis. J Inherit Metab Dis 37:421–429
Mignarri A, Rossi S, Ballerini M et al (2011) Clinical relevance and neurophysiological correlates of spasticity in cerebrotendinous xanthomatosis. J Neurol 258:783–790
Panzenboeck U, Andersson U, Hansson M, Sattler W, Meaney S, Björkhem I (2007) On the mechanism of cerebral accumulation of cholestanol in patients with cerebrotendinous xanthomatosis. J Lipid Res 48:1167–1174
Pilo de la Fuente B, Jimenez-Escrig A, Lorenzo JR et al (2011a) Cerebrotendinous xanthomatosis in Spain: clinical, prognostic, and genetic survey. Eur J Neurol 18:1203–1211
Pilo de la Fuente B, Sobrido MJ, Girós M et al (2011b) Usefulness of cholestanol levels in the diagnosis and follow-up of patients with cerebrotendinous xanthomatosis. Neurologia 26:397–404
Rubio-Agusti I, Kojovic M, Edwards MJ et al (2012) Atypical parkinsonism and cerebrotendinous xanthomatosis: report of a family with corticobasal syndrome and a literature review. Mov Disord 27:1769–1774
Salen G, Kwiterovich PO Jr, Shefer S et al (1985) Increased plasma cholestanol and 5 alpha-saturated plant sterol derivatives in subjects with sitosterolemia and xanthomatosis. J Lipid Res 26:203–209
Van Heijst AF, Verrips A, Wevers RA, Cruysberg JR, Renier WO, Tolboom JJ (1998) Treatment and follow-up of children with cerebrotendinous xanthomatosis. Eur J Pediatr 157:313–316
van Swieten JC, Koudstaal PJ, Visser MC et al (1988) Interobserver agreement for the assessment of handicap in stroke patients. Stroke 19:604–607
Wolthers BG, Walrecht HT, van der Molen JC, Nagel GT, Van Doormaal JJ, Wijnandts PN (1991) Use of determinations of 7-lathosterol (5 alpha-cholest-7-en-3 beta-ol) and other cholesterol precursors in serum in the study and treatment of disturbances of sterol metabolism, particularly cerebrotendinous xanthomatosis. J Lipid Res 32:603–612
Yahalom G, Tsabari R, Molshatzki N, Ephraty L, Cohen H, Hassin-Baer S (2013) Neurological outcome in cerebrotendinous xanthomatosis treated with chenodeoxycholic acid: early versus late diagnosis. Clin Neuropharmacol 36:78–83
Compliance with Ethic Guidelines
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5). Informed consent was obtained from all patients for being included in the study.
Conflict of interest
None.
Funding
Andrea Mignarri, Alessandro Magni, Marina Del Puppo, Gian Nicola Gallus, Ingemar Björkhem, Antonio Federico, and Maria Teresa Dotti declare no funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Jutta Gaertner
Rights and permissions
About this article
Cite this article
Mignarri, A., Magni, A., Del Puppo, M. et al. Evaluation of cholesterol metabolism in cerebrotendinous xanthomatosis. J Inherit Metab Dis 39, 75–83 (2016). https://doi.org/10.1007/s10545-015-9873-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-015-9873-1