Abstract
The fragmentation of tropical forests remains a major driver of avian biodiversity loss, particularly for insectivores, yet the mechanisms underlying area sensitivity remain poorly understood. Studies in lowland systems suggest that loss of food resources, changes to light microenvironments, increased nest predation, and dispersal limitation are possible mechanisms, but these are untested for montane tropical bird communities. In this study, we related avian functional traits to area sensitivity (quantified using beta estimates from a multi-species occupancy model) to test the above four hypotheses for a cloud forest bird community (both resident species and just resident insectivores) in the Colombian Western Andes. We found that species with more specialized diets and those that use the canopy and subcanopy (loss of food hypothesis), larger relative eye sizes (light microhabitat hypothesis), and larger clutch sizes (nest predation hypothesis) were significantly more area sensitive. By contrast, there was no support for the dispersal limitation hypothesis; instead, we found that insectivores with more pointed wing shapes, and more aerial lifestyles, were significantly more fragmentation sensitive. These results suggest that reduced vegetation structure, loss of late-successional plant species, and loss of epiphytic plants may reduce food availability in fragments. Similarly, the ability to tolerate higher light intensity near fragment edges, or when traversing matrix habitat, may be important for persistence in fragments and suggests that habitat configuration may be of special importance in fragmented Andean landscapes. Overall, a lack of information on foraging, movement, and breeding ecology complicates avian conservation in the Andes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among the most important drivers of biodiversity loss is the conversion of forest to other land uses, a trend that has continued unabated over the past decade (Heino et al. 2015; Watson et al. 2016), particularly at the tropical latitudes that house most of the world’s biodiversity (Lewis et al. 2015; Barlow et al. 2018). High rates of forest loss, especially in intact forested landscapes, are a leading cause of vertebrate endangerment (Tracewski et al. 2016; Betts et al. 2017), an effect amplified by forest fragmentation, or division of forest remnants into isolated patches of varying configuration and size that are separated by non-forest ‘matrix’ (Haddad et al. 2015). Forest fragmentation has particularly negative effects on tropical biodiversity, which declines with decreasing fragment area and increasing isolation (Fletcher et al. 2018; Betts et al. 2019). One ongoing conservation challenge in tropical fragmented landscapes, however, is understanding the mechanisms underlying sensitivity to patch area, even for well-studied taxa such as birds (Stratford and Robinson 2005; Sodhi et al. 2008; Robinson and Sherry 2012; Powell et al. 2015; Visco et al. 2015; Sherry 2021). Bird species respond differently to fragmentation, with some functional groups showing greater sensitivity than others (Bregman et al. 2014; Keinath et al. 2017). Insectivores, a group that makes up ~60% of both all tropical forest (Şekercioğlu 2012) and all Neotropical (Sherry et al. 2020) bird species, are particularly sensitive to fragmentation (Şekercioğlu et al. 2002; Bregman et al. 2014; Pavlacky et al. 2015; Powell et al. 2015; Sherry 2021). Therefore, mechanisms explaining area sensitivity are particularly needed for this diet guild.
One potential mechanism is the loss of specialized foraging microhabitats or resources in fragments. Many tropical forest insectivores show extreme foraging specialization (Sherry et al. 2020) and are often restricted to specific vertical forest strata (Marra and Remsen 1997; Walther 2002a) and foraging substrates, such as hanging dead vegetation (Mansor et al. 2019) or lianas (Schnitzer et al. 2020). Similarly, many montane frugivores (Dehling et al. 2014; Bender et al. 2018; Quitian et al. 2018), and nectarivores (Tinoco et al. 2017; Weinstein and Graham 2017; Sonne et al. 2019) are specialized on specific plant species that may be lost from forest fragments. Globally, bird species with more stereotyped foraging behavior are more at-risk than species with more plastic behavior (Ducatez et al. 2020), and dietary specialization has been linked to avian fragmentation sensitivity in some tropical forest habitats (Hadley et al. 2018; dos Anjos et al. 2019). Therefore, the simplification of vegetation structure in fragments and consequent loss of species-specific foraging microhabitats (Michel et al. 2015; Stratford and Stouffer 2015) and late-successional fruiting and flowering plants (Putz et al. 2011) in fragments may be important drivers of extirpations. In particular, dietary and microhabitat specialists should be most area sensitive if loss of food or foraging microhabitats is a dominant mechanism of area sensitivity.
Alternative hypotheses, however, abound. Fragment edges, for example, may alter the normally stable abiotic conditions of the forest understory, including light intensity, temperature, and humidity (Stratford and Robinson 2005; Robinson and Sherry 2012; Powell et al. 2015). Of these, light intensity may be most important because tropical forest birds are restricted to specific light microenvironments linked to their foraging stratum (Ausprey et al. 2021; Walther 2002b), and light intensity is more affected by edge effects than temperature or humidity (Patten and Smith-Patten 2012). Forest-interior birds have large eyes relative to their bodies (Ausprey 2021) and avoid high-light-intensity microhabitats (Pollock et al. 2015); this trait has been associated with increased edge avoidance (Martinez-Ortega et al. 2014) and disturbance sensitivity (Ausprey et al. 2021; but see Rutt et al. 2019). A third hypothesis is that many tropical bird species are dispersal limited (Visco et al. 2015), due in part to morphological adaptations to their specialized foraging ecology (Salisbury et al. 2012; Sherry et al. 2020). Many such species have a poor flight capacity (Moore et al. 2008), an aversion to gap crossing (Lees and Peres 2009; Ibarra-Macias et al. 2011), a short perceptual range (Awade et al. 2017), and therefore a limited ability to disperse among fragments (e.g., Woltmann et al. 2012). Wing shape, quantified as the hand wing index (HWI) has been shown to strongly correlate with dispersal distance (Arango et al. 2022), and is a strong predictor of fragmentation sensitivity globally (Weeks et al. 2023). The recent publication of a global HWI dataset (Sheard et al. 2020) allows us to test the dispersal limitation hypothesis indirectly through this trait.
The fourth hypothesized mechanism underlying area-sensitivity is an increase in nest predation due to a greater abundance of mesopredators in fragments (‘mesopredator release’: Crooks and Soulé 1999). The evidence for higher nest predation rates in tropical fragments, however, remains mixed (Robinson and Sherry 2012; Visco et al. 2015). While increased nest predation rates in fragments occur in some systems (Young et al. 2008; Newmark and Stanley 2011), nest predation rates can also decline in tropical fragments (Spanhove et al. 2014; Visco and Sherry 2015). This discrepancy may result from differences in nest predator identity and fragmentation sensitivity (Vetter et al. 2013; Visco et al. 2015); therefore, system- and landscape-specific tests of the nest predation hypothesis are required. If nest predation is an important factor, it should not affect all species equally. For one, nest type affects the nest predation rates of tropical birds, with open-cup nests being depredated at higher rates than covered and cavity nests (Brawn et al. 2011; Newmark and Stanley 2011). Cavity-nesting birds, in particular, appear to be less affected by forest disturbance as long as suitable nesting cavities are present (Cockle et al. 2015). Second, larger clutch sizes should buffer against the negative effects of predation and increase the demographic impact of a successful nesting attempt; greater fragmentation sensitivity has been significantly linked to smaller clutch sizes (Pavlacky et al. 2015). Finally, species with slow life histories should be more vulnerable to higher predation because of their smaller breeding populations and the greater demographic cost of a nest predation event (Owens and Bennett 2000). Alternatively, if primary nest predators themselves are fragmentation sensitive (e.g., Visco and Sherry 2015), then we might predict the opposite.
In this study, we tested these four hypothesized mechanisms of fragmentation sensitivity in a montane avifauna of the Colombian Western Andes. In a previous study (Jones et al. 2021), we used a multi-species occupancy model to derive beta estimates of the effects of patch size on species occupancy across a gradient of fragment sizes and a continuous forest reference site in the same landscape. In this study we relate these beta estimates to functional traits predicted to be associated with each of the four hypotheses (see Table 1 for hypotheses and predictions). Specifically, we asked: (1) if loss of foraging microhabitats, sensitivity to changing light intensity, dispersal limitation, or increased nest predation explain fragmentation sensitivity, and (2) if the mechanisms of sensitivity differ across the full bird community and the insectivore guild. We hypothesized that if loss of foraging microhabitats is a major mechanism, then species with more specialized diets and using specific foraging microhabitats, strata, and substrates should show a greater fragmentation sensitivity. Alternatively, greater sensitivity should be associated with a smaller HWI if dispersal limitation is a major mechanism, and with larger relative eye size if light sensitivity is a driver. Finally, we predicted that species with larger clutch sizes, ‘fast’ life histories, and cavity nests would be less fragmentation sensitive if altered nest predation is the dominant mechanism.
Methods
Study system
Field work was conducted in subtropical Andean forest and forest fragments in the municipality of El Cairo (4° 45′ 39″ N, 76° 13′ 21″ W; Valle del Cauca department) in the Western Andes of Colombia. Little is known about fragmentation sensitivity in tropical montane avifaunas, which represent hotspots of avian diversity (Sherry 2021). Andean cloud forests, in particular, contain a high richness of endemic and endangered birds (Orme et al. 2005; Kier et al. 2009) and are suffering extensive levels of deforestation for conversion to agriculture and cattle ranching (Tejedor-Garavito et al. 2012; Tracewski et al. 2016; Karger et al. 2021). Our study sites consisted of mid- to late-successional forest fragments embedded in an agricultural landscape of shade coffee and cattle pasture. We stratified forest fragments within the same elevational band (~1900–2300 m.a.s.l.) and same matrix type (cattle pasture) into large (≥ 100 ha), medium (~30–50 ha), and small (≤ 20 ha) size categories, and surveyed at least two replicates of each category (N = 8 fragments, range = 10–173 ha). We also surveyed a continuous-forest reference site in the same landscape connected to thousands of hectares of forest, the Reserva Natural Comunitaria Cerro El Inglés. A full description of the study system and study sites is available in Jones and Robinson (2020). Because local vegetation structure and land-use histories varied across and within fragments, largely due to selective logging, we established 500-m transects within forest interior as the sample unit (N = 14 transects). To maximize ecological variability within sites, we placed one transect in disturbed (logged) forest and another in relatively undisturbed forest in large fragments, where possible.
Collection of fragmentation sensitivity data
We obtained quantitative estimates of area sensitivity for all bird species encountered in our focal landscape from a multispecies occupancy model, which allowed us to estimate species occupancy while accounting for imperfect detection (Devarajan et al. 2020). The full model is specified in Jones et al. (2021); in brief, we fit a hierarchical model (Kéry and Royle 2016, Ch. 2), implemented in a Bayesian framework without data augmentation, that combined a state process model (i.e., the transect-level occupancy of each species) described by a Bernoulli distribution with an observation model of repeated detections from different survey techniques (i.e., the technique-specific detection probability) described by a binomial distribution. Input data consisted of presences and absences of each species along a transect on a sampling day from one of three complimentary survey techniques: audio-visual transect walks, understory mist netting, and playback surveys for owls. We sampled each of the 14 transects with each technique for 2.5 sequential survey days [surveys were not conducted concurrently; see Jones et al. (2021) for details of survey methods]. We then used an intercept term for survey type in the detectability sub-model to integrate the presence-absence data across techniques. Therefore, each modeled species had a unique detectability for each survey method, and we let species occupancy and detection parameters covary within the model. We fitted five fixed-effect covariates on occupancy: percentage forest cover and edge density (m/ha) within 1 km of the transect, an index of vertical vegetation structure along the transect, and two PCA axes describing the densities of understory vegetation and large-diameter trees, respectively. We also included a random effect of site (N = 9) to account for the non-independence of survey transects in the same forest fragment. Methods of predictor variable collection are described in Jones et al. (2021).
We used the median of the joint posterior distributions of beta estimates of percentage forest within 1 km of the transect on transect-level occupancy to quantify the area sensitivity of each species. We selected beta estimates for percentage forest within 1 km of the transect because this covariate was explicitly accounted for in the design (see above) and had the greatest number of significant effects on species occupancy (Jones et al. 2021). Positive values of this variable indicate higher species occupancy with greater percentage cover. Following Carrara et al. (2015), we used percentage forest as a proxy for patch size because our continuous forest reference site had no value for patch size. The matrix around our study sites was largely composed of treeless cattle pasture, and the percentage forest variable was highly correlated with patch size (Pearson’s correlation = 0.96).
Collection of avian functional traits
We collected 13 avian functional traits, representing four mechanisms of fragmentation sensitivity, for our analyses (Table 1). Data on foraging microhabitat, foraging behavior, and diet were obtained from Birds of the World species accounts (Billerman et al. 2021), supplemented by the authors’ field observations. Avian taxonomy therefore follows the eBird/Clements checklist. For all species (N = 178), we recorded use of five diet categories (insects, fruits, seeds, nectar, and vertebrates), use of six forest strata (ground, understory, midstory, subcanopy, canopy, aerial), and use of three microhabitats: ravines and small streams, bamboo patches (Chusquea spp.), and treefall gaps. For an additional subset of insectivores (N = 80) we also recorded the use of 11 foraging maneuvers (glean, sally, sally-glean, probe, peck, hang, hover-glean, hammer, flake, flush-pursue, gape) and eight foraging microhabitats (air, trunk, branch/twig, live leaf, dead leaf, vine, epiphyte, ground). We defined foraging maneuvers according to Remsen and Robinson (1990). For each diet category, we assigned a diet use score ranging from 0 (no use) to 3 (majority of the diet). We scored all other categorical variables as used (1) or not used (0). All categories were non-exclusive, and all species were scored by HHJ. We then created continuous measures of height stratum, foraging maneuver, and foraging microhabitat by ordinating these variables using principal coordinates analysis (PCoA; Gower 2015) with the Gower dissimilarity index. Following the methods of Morelli et al. (2019), we measured diet specialization using the Gini coefficient, a measure of statistical dispersion, on the diet use scores. Greater values of the Gini coefficient indicate greater dietary specialism.
We used lateral eye size as a proxy for light sensitivity, as this trait is correlated with a species’ light microhabitat (Ausprey et al. 2021). Eye sizes were obtained from two sources. For one subset of species (N = 72; 40%), we used mean eye size values from mist net captures in Peru, measured using calipers to the nearest 0.1 mm (data from Ausprey et al. 2021). For the other subset of species (N = 106; 60%), we measured lateral eye size from photos following the methods of Ausprey et al. (2021). We selected six images of each species in which the individual was at eye level and the bill and eye within the same focal plane. To ensure replicability, we used images from Cornell’s Macaulay Library with a unique identifier; all photos were measured by HHJ. We then scaled the photos in ImageJ using measurements of bill height at the nares from a database of Colombian bird morphology (Montoya et al. 2018) complemented with measurements from mist net captures (G. Londoño, unpublished data). To obtain a final eye size we multiplied the mean photo measurement by 1.03, as Ausprey et al. (2021) found that photo measurements were 3% smaller than field measurements. We then calculated relative eye size as the residual of the regression between log-transformed body mass (the mean mass of the male from Birds of the World) and log-transformed eye size (Figure S1). The hand wing index (HWI), a measure of wing shape (rounded versus pointed), correlates well with dispersal distance (Dawideit et al. 2009), and we used this trait as a proxy for dispersal ability. Larger values indicate a more pointed wing shape, which is associated with a higher flight performance, while lower values indicate a more rounded wing. Measurements of HWI were taken from the Sheard et al. (2020) global data set, sourced from museum collections and field data.
Finally, we collected data on reproductive traits from two sources. Nest type and maximum reported clutch size were taken from Birds of the World accounts, and we included information on nest type and clutch size from recent published literature where these were not described in the species account. When we could not find published accounts of nest type or clutch size, we used the values of the nearest congener. We divided nest type into four exclusive categories (cavity, dome, cup, platform) and reported the most common type for the species. To measure life history strategy, we obtained estimates of generation length, the average age of a species’ breeding population, from Bird et al. (2020).
Data analysis
We used phylogenetic least square (PLS) models to test for significant correlations between functional traits and sensitivity to fragment area (quantified as percentage forest within 1 km, see above). We tested for effects on both (1) the full resident bird community (N = 166 species; Fig. 1) and (2) only resident insectivores (N = 73 species). We excluded Nearctic-Neotropical migrant species (N = 14) from the analysis because these species do not breed in Andean forests, so functional traits based on nesting ecology are not appropriate for them. We also excluded the swift Chaetura cinereiventris from the insectivore analysis because this species had a large outlier value for HWI that was biasing analyses. The full community is here defined as all species detected at least once during field sampling with three different methods [mist netting, audio-visual transects, owl playbacks; see Jones et al. (2021)] for which we could estimate area sensitivity. We created phylogenies for both resident species and insectivores by subsetting the global Jetz et al. (2012) phylogeny; this phylogeny does not contain data for the recently described Scytalopus alvarezlopezi, so we substituted its sister species S. stilesi in the phylogeny. In each case, we downloaded 1000 trees from the Bird Tree website (www.birdtree.org) using the Hackett et al. (2008) backbone phylogeny, and then calculated a 50% majority-rule consensus tree using mean edge lengths with the consensus.edge function of the phytools package (Revell 2022). We ran phylogenetic least square models using the phylolm function of the same package (Tung Ho et al. 2020) and using the Brownian motion evolutionary model. We assumed that the area sensitivity of tropical resident birds should be the result of their natural history, itself shaped by numerous selective pressures. Because a species’ natural history should change gradually over time, and area sensitivity is not a trait that should evolve towards an optimum value, we believe that Ornstein–Uhlenbeck models are not appropriate for our data (Symonds and Blomberg 2014). Similarly, we selected the Brownian motion model over Pagel’s λ because that model estimated λ to be near zero, but phylogenetic effects on fragmentation sensitivity are frequently described for Andean bird families (e.g., greater sensitivity of Cotingidae, Icteridae, Trogonidae; Renjifo 1999; Santillan et al. 2019).
Fragment area sensitivity plotted onto a phylogeny of Andean birds. Plotted species (N = 166) represent all resident species detected during field surveys; Neotropical-Nearctic migrant species were excluded from analyses. Light yellow colors indicate greater fragmentation sensitivity, while red colors indicate species more commonly encountered in fragments
We fit models with ten and thirteen predictor variables for resident species and insectivores, respectively; all continuous predictor variables were z-scale transformed prior to analysis to allow for direct comparison of effect sizes. The continuous predictor variables all showed a Pearson correlation of less than 0.5, indicating a lack of multicollinearity. The foraging maneuver and foraging substrate variables were the only covariates with a correlation > 0.4 and removing these variables did not affect covariate significance in the averaged model. We then used an information theoretic framework to address model selection uncertainty, comparing models using the Akaike Information Criterion adjusted for small sample sizes (AICc; Burnham and Anderson 2002). For both subsets of species, we ranked all subsets of the global model, considering models equivalent to the best model if their ΔAICc value was ≤ 2. We then performed full model averaging of the candidate model set (i.e., all models within 2 ΔAICc of the best model), as there was no single best model. We used full model averaging because this method is recommended when evaluating the relative importance of predictor variables (Nakagawa and Freckleton 2011). AICc values and Akaike weights for all model subsets were calculated using the dredge function of the MuMIn package (Bartoń 2022), whereas model averaging was conducted using the model.avg function. Because p-values from model averaging do not account for the false discovery rate, we instead considered covariates to be significant when the 95% confidence interval did not overlap zero. We evaluated model goodness-of-fit using the adjusted r2 values calculated by the phylolm function, averaged across the candidate model set.
Results
Principal coordinates ordination of foraging height, behavior, and substrate
Based on the examination of a scree plot, we retained the first coordinate axis for the ordination of vertical foraging stratum use; this axis had an Eigenvalue of 13.50 and explained 52.40% of the variance (Table S1). Use of the ground (− 0.26) and understory (− 0.21) strata had negative loadings, while use of the subcanopy (0.21) and canopy (0.27) had positive loadings (Table S2, Figure S2). As such, we interpreted this axis as a measure of vertical stratum use. We similarly retained only the first axis from our ordination of foraging maneuver use (Eigenvalue = 2.03, percentage variance explained = 37.68%; Table S3). On this axis, aerial foraging maneuvers had high positive loadings (sally = 0.12, hover glean = 0.16, flush-pursue = 0.17; Table S4, Figure S3) whereas near-perch maneuvers had negative loadings (probe = − 0.17, hang = -0.16, flake = − 0.17). As such, we interpreted this axis as a measure of the extent to which a species uses aerial versus near-perch maneuvers. Finally, we retained the first two axes from our PCoA of foraging substrate. The Eigenvalues were 3.10 and 2.67 for the first and second axes, respectively, and these axes explained 29.52% and 25.41% of the variance (Table S5). The first axis had high positive loadings for use of the trunk (0.30) and epiphyte (0.31) substrates, and high negative loadings for use of live leaves (− 0.11) and air (− 0.11) as a substrate (Table S6, Figure S4). We interpreted this axis as a measure of near-trunk versus far-from-trunk foraging substrate use. The second axis had high positive loadings for use of the ground (0.16), dead leaves (0.20), and vine tangles (0.11) as substrates, and high negative loadings for use of air (− 0.14), branches and twigs (− 0.13), and trunk (− 0.21; Table S6). We therefore interpreted this axis as a measure of exposed (negative values) versus occluded (positive values) foraging microhabitats.
PLS analysis of area sensitivity
We found support for three of the four hypotheses for resident species: loss of foraging microhabitats and food, changing light intensity, and increased nest predation (Table 2, Fig. 2). Standardized (z-scale transformed) effect sizes were similar (≈1) across predictors associated with the three hypotheses, suggesting the multiple mechanisms were all similarly important. For resident birds, dietary specialists were more area sensitive (β = 1.09, 95% CI 0.46, 1.73), as were species that more frequently use the subcanopy and canopy (β = 0.69, 95% CI 0.01, 1.37; Fig. 3b); these results were both in the direction expected based on the loss of food resources hypothesis. Also as predicted, species with larger clutch sizes (β = − 1.15, 95% CI − 1.93, − 0.37) were less area sensitive, whereas species with larger relative eye size were more area sensitive (β = 0.89, 95% CI 0.09, 1.68; Fig. 3c). For the insectivore subset, we also found that species which forage at higher strata (β = 0.90, 95% CI 0.02, 1.77) were significantly more area sensitive, while those with larger maximum clutch sizes were less area sensitive (β = − 1.51, 95% CI − 2.30, − 0.72). Counterintuitively, species with a larger hand-wing index (more pointed wing shape) were also significantly more area sensitive for this subset of species (β = 1.67, 95% CI 0.11, 3.24). Finally, we found a near-significant effect of use of Chusquea bamboo stands on insectivore area sensitivity (β = 0.58, 95% CI − 0.005, 1.17). Model goodness-of-fit was relatively low, with a mean adjusted r2 of 0.16 and 0.19 for the full community and insectivore subset, respectively.
Effect sizes and 95% confidence intervals of functional traits on fragment area sensitivity. Effect sizes are from phylogenetic linear models using a Brownian motion model of evolution, run for the full community of resident species (A) and a subset of just insectivores (B). Colors correspond to the mechanism of fragmentation sensitivity: loss of food resources (blue), changes to predation patterns (red), changes to light intensity (black), and dispersal limitation (orange)
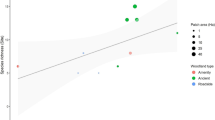
Andean forest vegetation structure and climate contribute to novel patterns of fragmentation sensitivity. A View of late-successional subtropical Andean forest subcanopy and canopy under heavy fog and cloud cover. Note the low-light conditions, prominent subcanopy stratum, and abundant epiphytic plants. B Area sensitivity was higher for species that use the canopy and subcanopy than the understory, perhaps because epiphytes and subcanopy tree species are lost from fragments. Labels correspond to the weighted averages of use of each stratum category from a PCoA. C Species with larger relative eye size were more fragmentation sensitive, perhaps because of adaptation to low-light conditions
Discussion
In this study, we used functional traits to test hypothesized mechanisms of fragmentation sensitivity for a cloud-forest bird community, finding indirect support for the changes to food and foraging microhabitat availability, increased predation pressure, and changes to light intensity hypotheses as drivers of bird extirpation in Andean fragments (Robinson and Sherry 2012; Powell et al. 2015; Visco et al. 2015; Ausprey et al. 2021). Conversely, there was no support for the dispersal-limitation hypothesis, and we instead found that insectivores with a larger HWI were more fragmentation sensitive. Overall, multiple simultaneous mechanisms with relatively even effect sizes appeared to explain bird extirpations in Andean fragments, and dietary specialists, subcanopy and canopy-dwellers, species with small clutch sizes, species with large relative eyes, and long-winged insectivores were particularly area sensitive. Many field studies have documented changes to the richness and composition of Andean bird communities after fragmentation (Kattan et al. 1994; Renjifo 1999; Castaño-Villa and Patino-Zabala 2008; Palacio et al. 2020; Jones et al. 2021), but here we test specific mechanisms of fragmentation sensitivity while controlling for phylogeny. Similarly, many studies have examined how avian functional diversity is affected by forest fragmentation (Santillán et al. 2019; Gómez et al. 2021; Ausprey et al. 2022), or which functional traits best predict sensitivity to fragmentation (Lees and Peres 2008; Vetter et al. 2011; Bregman et al. 2014; Keinath et al. 2017), but these functional traits are seldom, if ever, related to mechanisms of fragmentation sensitivity. We believe our methods have promise for testing and disentangling competing hypotheses of fragmentation sensitivity with field data from landscape-level studies.
However, our models did not have a high goodness-of-fit, suggesting that our hypotheses cannot explain all fragmentation sensitivity. This may be partly attributable to stochastic, rather than deterministic, species extirpations, yet lowland tropical bird communities consistently show non-random patterns of species loss (Hill et al. 2011; Luther et al. 2020). Instead, other mechanisms of extirpation may be taking place that we cannot easily quantify using functional traits. First, the connectivity of forest fragments to both upslope and downslope forest, and the persistence of habitat at these elevations, may be important for elevational migrants (e.g., Hilty 1997), though such behavior remains poorly described for Andean birds (Barçante et al. 2017; Jahn et al. 2020). The distance (and isolation) of a fragment from the center of a species’ elevational distribution may also be important (Terborgh 1985; Kattan et al. 1994; Mills et al. 2023), since populations at the periphery of their elevational range may face less suitable habitat and climate and occur at lower densities. Second, Andean landscapes are topographically variable, and relief type influences microclimate and plant communities. Most forest fragments in our study landscape are located on hilltops, yet Ribon et al. (2021) found that many Atlantic Forest species were specialized by relief type, and many preferred ravine microhabitats. Genera associated with ravines and streams in the Andes (e.g., Doryfera, Habia, Ochthoeca) may also be more fragmentation sensitive (Palacio et al. 2020; this study), and topographical complexity could be an important factor for cloud forest species conservation (Martinez-Morales 2005). Third, the elevational ranges of tropical montane birds are increasingly recognized to be constrained by competition with closely related species, often congeners (Jankowski et al. 2010; Freeman et al. 2019, 2022). Because fragments in the Andes are frequently invaded by downslope (Renjifo 1999) and edge-adapted (Jones et al. 2021) species, they may territorially exclude congeners from fragment habitat. We encourage future work to empirically evaluate these hypotheses for Andean avifauna.
Foraging and ecological specialization predict fragmentation sensitivity
Increasing dietary specialization, as quantified by the Gini index, was significantly associated with increased fragmentation sensitivity, suggesting a loss of overall food sources, host plants, or foraging microhabitats for specialists in fragments. This agrees with the findings that ecologically specialized forest species are more fragmentation sensitive (Khimoun et al. 2016; Keinath et al. 2017). Dietary specialists are likely to occur at lower densities on the landscape, a trait associated with fragmentation sensitivity by Renjifo (1999). We found that 53 (84%) of the 63 most specialized species by diet (i.e., where the Gini index = 1) in our community were insectivores, a guild considered particularly sensitive to fragmentation (Bregman et al. 2014; Sherry 2021). Because insectivores are specialized by foraging substrate and stratum (Sherry et al. 2020), the overall simplification of forest structure due to fragmentation (Rocha-Santos et al. 2016) is thought to lead to the loss of species-specific foraging microhabitats (Michel et al. 2015; Stratford and Stouffer 2015). This hypothesis is further corroborated by the fact that avian body condition was significantly correlated (both positively and negatively) with changes to vegetation structure in the same landscape (Jones et al. 2022). Nectarivores and frugivores that are specialized on late-successional plant species might also be affected by the retrogressive succession process in fragments, where the plant species composition more closely approximates early successional communities (Aubad et al. 2008; Putz et al. 2011; Rocha-Santos et al. 2016). For example, more morphologically specialized Andean hummingbirds (likely indicating greater specialization on plant species) were found to be more fragmentation sensitive (Hadley et al. 2018) and both frugivores and nectarivores showed high turnover across the El Cairo fragmentation gradient (turnover represented > 90% of beta dissimilarity in assemblage composition of these guilds; Jones et al. 2021).
Birds that use the subcanopy and canopy were also more sensitive to patch area than those using lower strata. This result agrees with Palacio et al. (2020), who found that Andean canopy species were more vulnerable, and Renjifo (1999), who reported the relative resilience of understory birds in Andean fragments. The finding, however, contrasts with the conventional wisdom that it is understory species, particularly insectivores, that are more fragmentation sensitive (Şekercioğlu et al. 2002; Powell et al. 2015). While many understory species were area sensitive (e.g., Grallaricula flavirostris, Sclerurus obscurior, Formicarius rufipectus, Cyphorhinus thoracicus, Ochthoeca chat-tyrants), we found a trend, also noted by Renjifo (1999), of many understory genera sharing fragmentation-averse and fragment-loving species pairs (e.g., Scytalopus vicinior and S. alvarezlopezi, Grallaria flavotincta and G. guatimalensis). The frequent landslide disturbance in Andean forests (Crausbay and Martin 2016), leading to dynamic and heterogenous landscapes with forest patches of different ages (Freund et al. 2021), may create niches for more early-successional understory species, leading to a less-sensitive assemblage. Andean canopy and subcanopy birds may also be more uniquely fragmentation sensitive, however. Many subcanopy and midstory trees are late successional species which are lost during retrogressive succession (see above), and such species tend to have larger fruits (Opler et al. 1980). Large fruits are the primary food source for many large-bodied frugivores (e.g., genera Trogon, Pharomachrus, Snowornis, Semnornis, Pipreola), a guild that is extremely fragmentation sensitive in the Andes (Palacio et al. 2020; Gómez et al. 2021). The subcanopy also contains the highest abundances of epiphytic plants (Hernandez-Perez et al. 2018; Alzate et al. 2019), which represent important foraging habitat for many specialized insectivores (e.g., Margarornis stellatus, Pseudocolaptes boissoneautii, Xiphorhynchus triangularis) and provide nectar and fruit for canopy specialists. However, epiphytic plant diversity decreases in cloud forest fragments, particularly near edges (Koster et al. 2009; Hundera et al. 2013; Bianchi and Kersten 2014;), likely resulting in the loss of key food resources for canopy species in fragments.
Changes to light microhabitat: edges, canopy gaps, and dispersal limitation?
A large relative eye size was significantly correlated with fragmentation sensitivity, suggesting that increases to light intensity in forest fragments may be an important mechanism of extirpation. This result extends the findings of Ausprey et al. (2021) from a gradient of human land use intensity to a gradient of forest fragment sizes. While changes to temperature and humidity in fragments have often been discussed as a mechanism of fragmentation sensitivity (Stratford and Robinson 2005; Robinson and Sherry 2012), light intensity may change more in fragments that these other factors (Patten and Smith-Patten 2012). Large relative eye size has been correlated with use of darker forest interior microhabitats (Ausprey 2021; Ausprey et al. 2021), yet tropical forest fragments show higher understory light intensities due to edges and canopy gaps (Reis et al. 2021) associated with reduced canopy cover from retrogressive succession and selective logging disturbance (Aubad et al. 2008; Rocha-Santos et al. 2016). Lowland forest-interior birds have been shown to select for darker microhabitats (Pollock et al. 2015; Jirinec et al. 2022), and this pattern correlates with relative eye size (Martinez-Ortega et al. 2014). Forest-specialist species in tropical montane forests also show reduced richness (Martinez-Morales 2005) and occupancy (Jankowski et al. 2021; Jones et al. 2021) with increasing edge density, and changes to light intensity may explain this pattern. Mist net captures show that the composition of Andean understory bird communities was affected up to 200 m away from fragment edges (Restrepo and Gómez 1998), and sensitivity to high-light environments may be even more pronounced in montane forests because of the lower light availability associated with higher fog and cloud cover (e.g., a reduction in incident light by ~80%; Bittencourt et al. 2019). In addition, large relative eye size may influence the ability of a species to disperse across fragmented landscapes. Such species are likely more susceptible to glare in bright, non-forest matrix (Fernandez-Juricic et al. 2012), and it may be the ability to tolerate higher light intensity, rather than wing shape, that explains dispersal success in the El Cairo landscape. The high potential for edge and dispersal effects based on light intensity strongly suggests that habitat configuration (i.e., fragmentation per se; Fletcher et al. 2018), in addition to habitat loss, is an important determinant of fragment extirpations in Andean cloud forests.
Clutch size effect: changes to predation in Andean fragments?
Species with larger maximum clutch sizes, both amongst insectivores and the full community, were significantly less likely to be fragmentation sensitive, yet the nest predation hypothesis was not fully supported as there was no significant effect of generation length or nest type on sensitivity. We found a relatively small range of maximum reported clutch sizes (1–5), but the species with single-egg clutches were high-montane species with restricted elevational ranges (e.g., Chlorochrysa nitidissima, Dendrocincla tyrannina, Snowornis cryptolophus), a pattern that may be common in tropical mountains (Boyce et al. 2015). The evidence for higher nest predation rates in Andean fragments is equivocal (Arango-Velez and Kattan 1997; Trujillo and Ahumada 2005) and is based on artificial nest studies, which may not accurately describe patterns of predation (Robinson and Sherry 2012). System-specific factors, likely including nest predator identity, influence whether fragmentation has negative or positive effects on tropical nest survival (Vetter et al. 2013) and, while the Andes have lower nest predation rates than the lowlands (Londoño et al. 2023), they also harbor a distinct nest predator community. This community contains fewer snakes and primates and is dominated by large-bodied birds (e.g., Turdus thrushes, Aulacorhynchus toucanets) and medium- and small-bodied mammals (Arango-Velez and Kattan 1997; Londoño et al. 2023). A camera-trap study in the El Cairo landscape found that large-bodied mammals were extirpated from fragments, while the occupancy of many medium-sized mammals that may predate nests increased (e.g., Eira barbara, Procyon cancrivorous, Dasyprocta punctata; Bedoya-Durán et al. 2023). Thus, the ‘mesopredator’ release hypothesis (Crooks and Soulé 1999) may hold true in subtropical Andean forests, where larger clutch sizes could help buffer against a higher predation rate. The invasion of Andean fragments by lowland bird species (see above) might also increase the density of nest-predating birds in small fragments, such as Turdus ignobilis or Cyanocorax yncas. Alternatively, changes to vegetation structure associated with fragmentation, such as lower canopy cover, could affect nest predation rates even in the absence of changes to predator densities by making nests more conspicuous (Nana et al. 2015). A better understanding of how nest predator communities are affected by fragmentation (e.g., Tallei et al. 2022), and empirical data on nest survival and conspicuousness in fragments are sorely needed for the Tropical Andes.
Positive HWI effect: the importance of aerial lifestyles?
There was no statistical support for the dispersal limitation hypothesis, as measured by the HWI, and we instead found that, among insectivores, there was a positive association between fragmentation sensitivity and HWI. This is a surprising result, given that tropical insectivores are thought to be dispersal limited (Powell et al. 2015), and that dispersal limitation (measured using HWI) was a strong predictor of fragmentation sensitivity globally (Weeks et al. 2023). Yet this finding agrees with Ausprey et al. (2022), who also found that wing pointedness increased with fragment size in the Andes. HWI has also been positively correlated with the ‘aerial lifestyle’ of a species, however, encompassing both aerial foraging maneuvers and the extent to which species make flights in the course of their daily movement patterns (Weeks et al. 2022). Insectivores with a higher HWI included both aerially foraging insectivores (e.g., Hapaloptila castanea, Pachyramphus versicolor), particularly tyrant flycatchers (genera Phyllomyias, Phylloscartes, Octhoeca), as well as woodpeckers (Picidae) and woodcreepers (subfamily Dendrocolaptinae), which make long flights from one foraging tree to another. Woodcreepers have previously been described as fragmentation sensitive at middle elevations in the Andes (Castaño-Villa and Patino-Zabala 2008; Santillán et al. 2019). Species with aerial lifestyles may be more sensitive for a few reasons. For one, aerial foragers have larger relative eye sizes (Ausprey 2021) and may be more sensitive to changes to light intensity (see above). Many Andean flycatchers also use upward-sally maneuvers to live foliage, and as such are particularly sensitive to simplified vegetation structure in fragments. Species that make long flights between foraging patches may use specialized resources, especially large-diameter trees, which require larger home ranges and more frequent movement. Not only are large-diameter, late-successional trees lost from fragments (Aubad et al. 2008), but habitat fragmentation may limit movement to other foraging patches. Indeed, large-bodied frugivores that track fruiting events on the landscape (Chatterjee and Basu 2018; Bonfim et al. 2021) and granivores that track bamboo seeding events (e.g., Spodiornis rusticus, Sanchez 2005) may be similarly vulnerable. Thus, a better understanding is needed of home range sizes and daily and seasonal movement patterns of Andean birds.
Conclusions and conservation implications
The unique structural features and disturbance regimes of Andean forests produce patterns of fragmentation sensitivity that are both similar to, and strikingly different from, the Neotropical lowlands. As in other systems, foraging and vertical stratum specialization was a good predictor of fragmentation sensitivity, suggesting that loss of food and foraging strata is a major mechanism. However, subcanopy and canopy species, rather than understory birds, were more area sensitive, perhaps because some understory species are adapted to post-landslide successional habitat. We suggest that illegal logging disturbances that simplify vegetation structure and the negative effects of edges on epiphytes and late-successional plants may be particularly important in reducing food sources in fragments. There was no evidence of dispersal limitation in our focal community; instead, insectivores that make more frequent flights in their foraging or daily movements were more vulnerable. The ability of a species to tolerate increased light intensity during cross-fragment dispersal, or within edge and canopy gap habitat in fragments, may thus be a more important driver of sensitivity in forests characterized by frequent fog and cloud cover. Minimizing edge effects and maintaining patches of large-diameter, late-successional trees on the landscape are therefore critical for successful management of area-sensitive species in Tropical Andean cloud forest. Our results highlight the paucity of natural history knowledge (foraging ecology, home range sizes, seasonal and altitudinal movement patterns, identity of nest predators) necessary to effectively implement avian conservation strategies in Andean landscapes.
Data availability
The datasets generated during the current study will be archived at The Institutional Repository at the University of Florida upon acceptance of the manuscript.
References
Alzate NF, Garcia-Franco JG, Flores-Palacios A, Kromer T, Laborde J (2019) Influence of land use types on the composition and diversity of orchids and their phorophytes in cloud forest fragments. Flora 260:151463. https://doi.org/10.1016/j.flora.2019.151463
Arango A, Pinto-Ledezma J, Rojas-Soto O, Lindsay AM, Mendenhall CD, Villalobos F (2022) Hand-Wing Index as a surrogate for dispersal ability: the case of the Emberizoidea (Aves: Passeriformes) radiation. Biol J Linn Soc 137:137–144. https://doi.org/10.1093/biolinnean/blac071
Arango-Velez N, Kattan GH (1997) Effects of forest fragmentation on experimental nest predation in Andean cloud forest. Biol Conserv 81:137–143. https://doi.org/10.1016/s0006-3207(96)00138-3
Aubad J, Aragon P, Olalla-Tarraga MA, Rodriguez MA (2008) Illegal logging, landscape structure and the variation of tree species richness across North Andean forest remnants. For Ecol Manag 255:1892–1899. https://doi.org/10.1016/j.foreco.2007.12.011
Ausprey IJ (2021) Adaptations to light contribute to the ecological niches and evolution of the terrestrial avifauna. Proc Royal Soc B 288:20210853. https://doi.org/10.1098/rspb.2021.0853
Ausprey IJ, Newell FL, Robinson SK (2021) Adaptations to light predict the foraging niche and disassembly of avian communities in tropical countrysides. Ecology 102:e03213. https://doi.org/10.1002/ecy.3213
Ausprey IJ, Newell FL, Robinson SK (2022) Functional response traits and altered ecological niches drive the disassembly of cloud forest bird communities in tropical montane countrysides. J Anim Ecol 91:2314–2328. https://doi.org/10.1111/1365-2656.13816
Awade M, Candia-Gallardo C, Cornelius C, Metzger JP (2017) High emigration propensity and low mortality on transfer drives female-biased dispersal of Pyriglena leucoptera in fragmented landscapes. Plos One 12:e0170493. https://doi.org/10.1371/journal.pone.0170493
Barçante L, Vale MM, Alves MAS (2017) Altitudinal migration by birds: a review of the literature and a comprehensive list of species. J Field Ornithol 88:321–335. https://doi.org/10.1111/jofo.12234
Barlow J, Franca F, Gardner TA, Hicks CC, Lennox GD, Berenguer E, Castello L, Economo EP, Ferreira J, Guenard B, Leal CG, Isaac V, Lees AC, Parr CL, Wilson SK, Young PJ, Graham NAJ (2018) The future of hyperdiverse tropical ecosystems. Nature 559:517–526. https://doi.org/10.1038/s41586-018-0301-1
Bartoń, K (2022) Package 'MuMIn': Multi-Model Inference. Version 1.46.0.
Bedoya-Durán MJ, Jones HH, Malone K, Branch L (2023) Continuous forest at higher elevation plays a key role in maintaining bird and mammal diversity across an Andean coffee-growing landscape. Anim Conserv. https://doi.org/10.1111/acv.12857
Bender IMA, Kissling WD, Blendinger PG, Bohning-Gaese K, Hensen I, Kuhn I, Muñoz MC, Neuschulz EL, Nowak L, Quitian M, Saavedra F, Santillan V, Topfer T, Wiegand T, Dehling DM, Schleuning M (2018) Morphological trait matching shapes plant-frugivore networks across the Andes. Ecography 41:1910–1919. https://doi.org/10.1111/ecog.03396
Betts MG, Wolf C, Ripple WJ, Phalan B, Millers KA, Duarte A, Butchart SHM, Levi T (2017) Global forest loss disproportionately erodes biodiversity in intact landscapes. Nature 547:441–444. https://doi.org/10.1038/nature23285
Betts MG, Wolf C, Pfeifer M, Banks-Leite C, Arroyo-Rodriguez V, Ribeiro DB, Barlow J, Eigenbrod F, Faria D, Fletcher RJ, Hadley AS, Hawes JE, Holt RD et al (2019) Extinction filters mediate the global effects of habitat fragmentation on animals. Science 366:1236–1239. https://doi.org/10.1126/science.aax9387
Bianchi JS, Kersten RD (2014) Edge effect on vascular epiphytes in a subtropical Atlantic Forest. Acta Bot Brasilica 28:120–126. https://doi.org/10.1590/s0102-33062014000100012
Billerman SM, Keeney BK, Rodewald PG, Schulenberg TS (2021) Birds of the world. Cornell Lab of Ornithology, Ithaca
Bird JP, Martin R, Akcakaya HR, Gilroy J, Burfield IJ, Garnett ST, Symes A, Taylor J, Sekercioglu CH, Butchart SHM (2020) Generation lengths of the world’s birds and their implications for extinction risk. Conserv Biol 34:1252–1261. https://doi.org/10.1111/cobi.13486
Bittencourt PRL, Barros FD, Eller CB, Muller CS, Oliveira RS (2019) The fog regime in a tropical montane cloud forest in Brazil and its effects on water, light and microclimate. Agric for Meteorol 265:359–369. https://doi.org/10.1016/j.agrformet.2018.11.030
Bonfim FCG, Dodonov P, Cazetta E (2021) Landscape composition is the major driver of the taxonomic and functional diversity of tropical frugivorous birds. Landsc Ecol 36:2535–2547. https://doi.org/10.1007/s10980-021-01266-y
Boyce AJ, Freeman BG, Mitchell AE, Martin TE (2015) Clutch size declines with elevation in tropical birds. Auk 132:424–432. https://doi.org/10.1642/AUK-14-150.1
Brawn JD, Angehr G, Davros N, Robinson WD, Styrsky JN, Tarwater CE (2011) Sources of variation in the nesting success of understory tropical birds. J Avian Biol 42:61–68. https://doi.org/10.1111/j.1600-048X.2010.04897.x
Bregman TP, Sekercioglu CH, Tobias JA (2014) Global patterns and predictors of bird species responses to forest fragmentation: implications for ecosystem function and conservation. Biol Conserv 169:372–383. https://doi.org/10.1016/j.biocon.2013.11.024
Burnham K, Anderson D (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Carrara E, Arroyo-Rodriguez V, Vega-Rivera JH, Schondube JE, de Freitas SM, Fahrig L (2015) Impact of landscape composition and configuration on forest specialist and generalist bird species in the fragmented Lacandona rainforest, Mexico. Biol Conserv 184:117–126. https://doi.org/10.1016/j.biocon.2015.01.014
Castaño-Villa GJ, Patino-Zabala JC (2008) Extinciones locales de aves en fragmentos de bosque en la region de Santa Elena, Andes centrales, Colombia. Hornero 23:23–34
Chatterjee S, Basu P (2018) Food preferences determine habitat selection at multiple scales: implication for bird conservation in tropical forests. Anim Conserv 21:332–342. https://doi.org/10.1111/acv.12397
Cockle KL, Bodrati A, Lammertink M, Martin K (2015) Cavity characteristics, but not habitat, influence nest survival of cavity-nesting birds along a gradient of human impact in the subtropical Atlantic Forest. Biol Conserv 184:193–200. https://doi.org/10.1016/j.biocon.2015.01.026
Crausbay SD, Martin PH (2016) Natural disturbance, vegetation patterns and ecological dynamics in tropical montane forests. J Trop Ecol 32:384–403. https://doi.org/10.1017/s0266467416000328
Crooks KR, Soulé ME (1999) Mesopredator release and avifaunal extinctions in a fragmented system. Nature 400:563–566. https://doi.org/10.1038/23028
Dawideit BA, Phillimore AB, Laube I, Leisler B, Bohning-Gaese K (2009) Ecomorphological predictors of natal dispersal distances in birds. J Anim Ecol 78:388–395. https://doi.org/10.1111/j.1365-2656.2008.01504.x
Dehling DM, Fritz SA, Topfer T, Packert M, Estler P, Bohning-Gaese K, Schleuning M (2014) Functional and phylogenetic diversity and assemblage structure of frugivorous birds along an elevational gradient in the tropical Andes. Ecography 37:1047–1055. https://doi.org/10.1111/ecog.00623
Devarajan K, Morelli TL, Tenan S (2020) Multi-species occupancy models: review, roadmap, and recommendations. Ecography 43:1612–1624. https://doi.org/10.1111/ecog.04957
dos Anjos L, Menezes Bochio G, Reis Medeiros H, de Arruda AB, Arakaki Lindsey B, Corsini Calsavara L, Cezar Ribeiro M, Domingues Torezan JM (2019) Insights on the functional composition of specialist and generalist birds throughout continuous and fragmented forests. Ecol Evol 9:6318–6328. https://doi.org/10.1002/ece3.5204
Ducatez S, Sol D, Sayol F, Lefebvre L (2020) Behavioural plasticity is associated with reduced extinction risk in birds. Nat Ecol Evol 4:788–793. https://doi.org/10.1038/s41559-020-1168-8
Fernandez-Juricic E, Deisher M, Stark AC, Randolet J (2012) Predator detection is limited in microhabitats with high light intensity: an experiment with Brown-Headed Cowbirds. Ethology 118:341–350. https://doi.org/10.1111/j.1439-0310.2012.02020.x
Fletcher RJ, Didham RK, Banks-Leite C, Barlow J, Ewers RM, Rosindell J, Holt RD, Gonzalez A, Pardini R, Damschen EI, Melo FPL, Ries L, Prevedello JA, Tscharntke T, Laurance WF, Lovejoy T, Haddad NM (2018) Is habitat fragmentation good for biodiversity? Biol Conserv 226:9–15. https://doi.org/10.1016/j.biocon.2018.07.022
Freeman BG, Tobias JA, Schluter D (2019) Behavior influences range limits and patterns of coexistence across an elevational gradient in tropical birds. Ecography 42:1832–1840. https://doi.org/10.1111/ecog.04606
Freeman BG, Strimas-Mackey M, Miller ET (2022) Interspecific competition limits bird species’ ranges in tropical mountains. Science 377:416–420. https://doi.org/10.1126/science.abl7242
Freund CA, Clark KE, Curran JF, Asner GP, Silman MR (2021) Landslide age, elevation and residual vegetation determine tropical montane forest canopy recovery and biomass accumulation after landslide disturbances in the Peruvian Andes. J Ecol 109:3555–3571. https://doi.org/10.1111/1365-2745.13737
Gómez C, Tenorio EA, Cadena CD (2021) Change in avian functional fingerprints of a Neotropical montane forest over 100 years as an indicator of ecosystem integrity. Conserv Biol 35:1552–1563. https://doi.org/10.1111/cobi.13714
Gower J (2015) Principal coordinates analysis. Wiley StatsRef: Statistics Reference Online 1. https://doi.org/10.1002/9781118445112.stat05670.pub2
Hackett SJ, Kimball RT, Reddy S, Bowie RCK, Braun EL, Braun MJ, Chojnowski JL, Cox WA, Han KL, Harshman J, Huddleston CJ, Marks BD, Miglia KJ, Moore WS, Sheldon FH, Steadman DW, Witt CC, Yuri T (2008) A phylogenomic study of birds reveals their evolutionary history. Science 320:1763–1768. https://doi.org/10.1126/science.1157704
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, King AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song DX, Townshend JR (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv. https://doi.org/10.1126/sciadv.1500052
Hadley AS, Frey SJK, Robinson WD, Betts MG (2018) Forest fragmentation and loss reduce richness, availability, and specialization in tropical hummingbird communities. Biotropica 50:74–83. https://doi.org/10.1111/btp.12487
Heino M, Kummu M, Makkonen M, Mulligan M, Verburg PH, Jalava M, Rasanen TA (2015) Forest loss in protected areas and intact forest landscapes: a global analysis. Plos One 10:e0138918. https://doi.org/10.1371/journal.pone.0138918
Hernandez-Perez E, Solano E, Rios-Gomez R (2018) Host affinity and vertical distribution of epiphytic orchids in a montane cloud forest in southern Mexico. Bot Sci 96:200–217. https://doi.org/10.17129/botsci.1869
Hill JK, Gray MA, Khen CV, Benedick S, Tawatao N, Hamer KC (2011) Ecological impacts of tropical forest fragmentation: how consistent are patterns in species richness and nestedness? Philos Trans R Soc B 366:3265–3276. https://doi.org/10.1098/rstb.2011.0050
Hilty S (1997) Seasonal distribution of birds at a cloud-forest locality, the Anchicayá valley, in Western Colombia. Ornithol Monogr 48:321–343. https://doi.org/10.2307/40157541
Hundera K, Aerts R, De Beenhouwer M, Van Overtveld K, Helsen K, Muys B, Honnay O (2013) Both forest fragmentation and coffee cultivation negatively affect epiphytic orchid diversity in Ethiopian moist evergreen Afromontane forests. Biol Conserv 159:285–291. https://doi.org/10.1016/j.biocon.2012.10.029
Ibarra-Macias A, Robinson WD, Gaines MS (2011) Experimental evaluation of bird movements in a fragmented Neotropical landscape. Biol Conserv 144:703–712. https://doi.org/10.1016/j.biocon.2010.08.006
Jahn AE, Cueto VR, Fontana CS, Guaraldo AC, Levey DJ, Marra PP, Ryder TB (2020) Bird migration within the Neotropics. Ornithol 137:ukaa033. https://doi.org/10.1093/auk/ukaa033
Jankowski JE, Robinson SK, Levey DJ (2010) Squeezed at the top: Interspecific aggression may constrain elevational ranges in tropical birds. Ecology 91:1877–1884. https://doi.org/10.1890/09-2063.1
Jankowski JE, Kyle KO, Gasner MR, Ciecka AL, Rabenold KN (2021) Response of avian communities to edges of tropical montane forests: Implications for the future of endemic habitat specialists. Glob Ecol Conserv 30:e01776. https://doi.org/10.1016/j.gecco.2021.e01776
Jetz W, Thomas GH, Joy JB, Hartmann K, Mooers AO (2012) The global diversity of birds in space and time. Nature 491:444–448. https://doi.org/10.1038/nature11631
Jirinec V, Rodrigues PF, Amaral BR, Stouffer PC (2022) Light and thermal niches of ground-foraging Amazonian insectivorous birds. Ecology 103:e3645. https://doi.org/10.1002/ecy.3645
Jones HH, Robinson SK (2020) Patch size and vegetation structure drive changes to mixed-species flock diversity and composition across a gradient of fragment sizes in the Western Andes of Colombia. Ornithol App 122:duaa006. https://doi.org/10.1093/condor/duaa006
Jones HH, Barreto E, Murillo O, Robinson SK (2021) Turnover-driven loss of forest-dependent species changes avian species richness, functional diversity, and community composition in Andean forest fragments. Glob Ecol Conserv 32:e01922. https://doi.org/10.1016/j.gecco.2021.e01922
Jones HH, Colorado G, Robinson SK (2022) Widespread bird species show idiosyncratic responses in residual body mass to selective logging and edge effects in the Colombian Western Andes. Ornithol App 124:duac026. https://doi.org/10.1093/ornithapp/duac026
Karger DN, Kessler M, Lehnert M, Jetz W (2021) Limited protection and ongoing loss of tropical cloud forest biodiversity and ecosystems wordwide. Nat Ecol Evol 5:854–862. https://doi.org/10.1038/s41559-021-01450-y
Kattan GH, Alvarezlopez H, Giraldo M (1994) Forest fragmentation and bird extinctions: San Antonio 80 years later. Conserv Biol 8:138–146. https://doi.org/10.1046/j.1523-1739.1994.08010138.x
Keinath DA, Doak DF, Hodges KE, Prugh LR, Fagan W, Sekercioglu CH, Buchart SHM, Kauffman M (2017) A global analysis of traits predicting species sensitivity to habitat fragmentation. Glob Ecol Biogeogr 26:115–127. https://doi.org/10.1111/geb.12509
Kéry M, Royle JA (2016) Applied hierarchical modeling in ecology: analysis of distribution, abundance, and species richness in R and BUGS. Academic Press, London
Khimoun A, Eraud C, Ollivier A, Arnoux E, Rocheteau V, Bely M, Lefol E, Delpuech M, Carpentier ML, Leblond G, Levesque A, Charbonnel A, Faivre B, Garnier S (2016) Habitat specialization predicts genetic response to fragmentation in tropical birds. Mol Ecol 25:3831–3844. https://doi.org/10.1111/mec.13733
Kier G, Kreft H, Lee TM, Jetz W, Ibisch PL, Nowicki C, Mutke J, Barthlott W (2009) A global assessment of endemism and species richness across island and mainland regions. Proc Natl Acad Sci USA 106:9322–9327. https://doi.org/10.1073/pnas.0810306106
Koster N, Friedrich K, Nieder J, Barthlott W (2009) Conservation of epiphyte diversity in an Andean landscape transformed by human land use. Conserv Biol 23:911–919. https://doi.org/10.1111/j.1523-1739.2008.01164.x
Lees AC, Peres CA (2008) Avian life-history determinants of local extinction risk in a hyper-fragmented neotropical forest landscape. Anim Conserv 11:128–137. https://doi.org/10.1111/j.1469-1795.2008.00162.x
Lees AC, Peres CA (2009) Gap-crossing movements predict species occupancy in Amazonian forest fragments. Oikos 118:280–290. https://doi.org/10.1111/j.1600-0706.2008.16842.x
Lewis SL, Edwards DP, Galbraith D (2015) Increasing human dominance of tropical forests. Science 349:827–832. https://doi.org/10.1126/science.aaa9932
Londoño GA, Gomez JP, Sánchez-Martínez MA, Levey DJ, Robinson SK (2023) Changing patterns of nest predation and predator communities along a tropical elevation gradient. Ecol Letters 26:609–620. https://doi.org/10.1111/ele.14189
Luther DA, Cooper WJ, Wolfe JD, Bierregaard RO, Gonzalez A, Lovejoy TE (2020) Tropical forest fragmentation and isolation: Is community decay a random process? Glob Ecol Conserv 23:e01168. https://doi.org/10.1016/j.gecco.2020.e01168
Mansor MS, Rozali FZ, Abdullah NA, Nor SM, Ramli R (2019) How important is aerial leaf litter for insectivorous birds foraging in a Malaysian tropical forest? Glob Ecol Conserv 20:e00722. https://doi.org/10.1016/j.gecco.2019.e00722
Marra PP, Remsen JV Jr (1997) Insights into the maintenance of high species diversity in the Neotropics: habitat selection and foraging behavior in understory birds of tropical and temperate forests. Ornithol Monogr 48:445–483. https://doi.org/10.2307/40157547
Martinez-Morales MA (2005) Landscape patterns influencing bird assemblages in a fragmented Neotropical cloud forest. Biol Conserv 121:117–126. https://doi.org/10.1016/j.biocon.2004.04.015
Martinez-Ortega C, Santos ESA, Gil D (2014) Species-specific differences in relative eye size are related to patterns of edge avoidance in an Amazonian rainforest bird community. Ecol Evol 4:3736–3745. https://doi.org/10.1002/ece3.1194
Michel NL, Carson WP, Sherry TW (2015) Do collared peccaries negatively impact understory insectivorous rain forest birds indirectly via lianas and vines? Biotropica 47:745–757. https://doi.org/10.1111/btp.12261
Mills SC, Socolar JB, Edwards FA, Parra E, Martínez-Revelo DE, Ochoa Quintero JM, Haugaasen T, Freckleton RP, Barlow J, Edwards DP (2023) High sensitivity of tropical forest birds to deforestation at lower altitudes. Ecology 104:e3867. https://doi.org/10.1002/ecy.3867
Montoya P, Gonzalez MA, Tenorio EA, Lopez-Ordonez JP, Gomez AP, Cueva D, Rincon AAA, Yanes CA, Martinez HMA, Armesto O, Betancur JS, Castro AC, Leyton JJC, Calpa-Anaguano EV, Cardenas-Posada G et al (2018) A morphological database for 606 Colombian bird species. Ecology 99:1693–1693. https://doi.org/10.1002/ecy.2368
Moore RP, Robinson WD, Lovette IJ, Robinson TR (2008) Experimental evidence for extreme dispersal limitation in tropical forest birds. Ecol Lett 11:960–968. https://doi.org/10.1111/j.1461-0248.2008.01196.x
Morelli F, Benedetti Y, Moller AP, Fuller RA (2019) Measuring avian specialization. Ecol Evol 9:8378–8386. https://doi.org/10.1002/ece3.5419
Nakagawa S, Freckleton RP (2011) Model averaging, missing data, and multiple imputation: a case study for behavioral ecology. Behav Ecol Sociobiol 65:103–116. https://doi.org/10.1007/s00265-010-1044-7
Nana ED, Sedlacek O, Dolezal J, Dancak M, Altman J, Svoboda M, Majesky L, Horak D (2015) Relationship between survival rate of avian artificial nests and forest vegetation structure along a tropical altitudinal gradient on Mount Cameroon. Biotropica 47:758–764. https://doi.org/10.1111/btp.12262
Newmark WD, Stanley TR (2011) Habitat fragmentation reduces nest survival in an Afrotropical bird community in a biodiversity hotspot. Proc Natl Acad Sci USA 108:11488–11493. https://doi.org/10.1073/pnas.1104955108
Opler PA, Baker HG, Frankie GW (1980) Plant reproductive characteristics during secondary succession in Neotropical lowland forest ecosystems. Biotropica 12:40–46. https://doi.org/10.2307/2388155
Orme CDL, Davies RG, Burgess M, Eigenbrod F, Pickup N, Olson VA, Webster AJ, Ding TS, Rasmussen PC, Ridgely RS, Stattersfield AJ, Bennett PM, Blackburn TM, Gaston KJ, Owens IPF (2005) Global hotspots of species richness are not congruent with endemism or threat. Nature 436:1016–1019. https://doi.org/10.1038/nature03850
Owens IPF, Bennett PM (2000) Ecological basis of extinction risk in birds: habitat loss versus human persecution and introduced predators. Proc Natl Acad Sci USA 97:12144–12148. https://doi.org/10.1073/pnas.200223397
Palacio RD, Kattan GH, Pimm SL (2020) Bird extirpations and community dynamics in an Andean cloud forest over 100 years of land-use change. Conserv Biol 34:677–687. https://doi.org/10.1111/cobi.13423
Patten MA, Smith-Patten BD (2012) Testing the microclimate hypothesis: light environment and population trends of Neotropical birds. Biol Conserv 155:85–93. https://doi.org/10.1016/j.biocon.2012.06.004
Pavlacky DC, Possingham HP, Goldizen AW (2015) Integrating life history traits and forest structure to evaluate the vulnerability of rainforest birds along gradients of deforestation and fragmentation in eastern Australia. Biol Conserv 188:89–99. https://doi.org/10.1016/j.biocon.2014.10.020
Pollock HS, Cheviron ZA, Agin TJ, Brawn JD (2015) Absence of microclimate selectivity in insectivorous birds of the Neotropical forest understory. Biol Conserv 188:116–125. https://doi.org/10.1016/j.biocon.2014.11.013
Powell LL, Cordeiro NJ, Stratford JA (2015) Ecology and conservation of avian insectivores of the rainforest understory: a pantropical perspective. Biol Conserv 188:1–10. https://doi.org/10.1016/j.biocon.2015.03.025
Putz S, Groeneveld J, Alves LF, Metzger JP, Huth A (2011) Fragmentation drives tropical forest fragments to early successional states: a modelling study for Brazilian Atlantic forests. Ecol Modell 222:1986–1997. https://doi.org/10.1016/j.ecolmodel.2011.03.038
Quitián M, Santillan V, Espinosa CI, Homeier J, Bohning-Gaese K, Schleuning M, Neuschulz EL (2018) Elevation-dependent effects of forest fragmentation on plant-bird interaction networks in the tropical Andes. Ecography 41:1497–1506. https://doi.org/10.1111/ecog.03247
Reis IP, Rocha-Santos L, Leal A, Faria D, Mielke MS (2021) Landscape forest loss changes sunfleck dynamics in forest fragments of southern Bahia, Brazil. J Trop Ecol 37:64–71. https://doi.org/10.1017/S0266467421000110
Remsen JV Jr, Robinson SK (1990) A classification scheme for foraging behvior of birds in terrestrial habitats. In: Morrison ML (ed) Studies in avian biology, No 13 avian foraging: theory, methodology, and applications. Cooper Ornithological Society, Los Angeles
Renjifo LM (1999) Composition changes in a subandean avifauna after long-term forest fragmentation. Conserv Biol 13:1124–1139. https://doi.org/10.1046/j.1523-1739.1999.98311.x
Restrepo C, Gómez N (1998) Responses of understory birds to anthropogenic edges in a Neotropical montane forest. Ecol App 8:170–183. https://doi.org/10.2307/2641319
Revell LJ (2022) Package 'phytools': phylogenetic tools for comparative biology (and Other Things). Version 1.0–3.
Ribon R, dos Santos LR, De Marco P, Marini MA (2021) Topography as a determinant of bird distribution in secondary Atlantic Forest fragments. J Trop Ecol 37:228–234. https://doi.org/10.1017/S0266467421000328
Robinson WD, Sherry TW (2012) Mechanisms of avian population decline and species loss in tropical forest fragments. J Ornithol 153:S141–S152. https://doi.org/10.1007/s10336-011-0806-y
Rocha-Santos L, Pessoa MS, Cassano CR, Talora DC, Orihuela RLL, Mariano-Neto E, Morante JC, Faria D, Cazetta E (2016) The shrinkage of a forest: landscape-scale deforestation leading to overall changes in local forest structure. Biol Conserv 196:1–9. https://doi.org/10.1016/j.biocon.2016.01.028
Rutt CL, Midway SR, Jirinec V, Wolfe JD, Stouffer PC (2019) Examining the microclimate hypothesis in Amazonian birds: indirect tests of the “visual constraints” mechanism. Oikos 128:798–810. https://doi.org/10.1111/oik.05781
Salisbury CL, Seddon N, Cooney CR, Tobias JA (2012) The latitudinal gradient in dispersal constraints: ecological specialisation drives diversification in tropical birds. Ecol Lett 15:847–855. https://doi.org/10.1111/j.1461-0248.2012.01806.x
Sanchez C (2005) First description of the nest and eggs of the Slaty Finch (Haplospiza rustica) and observations on song and breeding behavior. Ornitol Neotrop 16:493–501
Santillán V, Quitian M, Tinoco BA, Zarate E, Schleuning M, Bohning-Gaese K, Neuschulz EL (2019) Different responses of taxonomic and functional bird diversity to forest fragmentation across an elevational gradient. Oecologia 189:863–873. https://doi.org/10.1007/s00442-018-4309-x
Schnitzer SA, Michel NL, Powers JS, Robinson WD (2020) Lianas maintain insectivorous bird abundance and diversity in a Neotropical forest. Ecology 101:e03176. https://doi.org/10.1002/ecy.3176
Şekercioğlu CH (2012) Bird functional diversity and ecosystem services in tropical forests, agroforests and agricultural areas. J Ornithol 153:S153–S161. https://doi.org/10.1007/s10336-012-0869-4
Şekercioğlu CH, Ehrlich PR, Daily GC, Aygen D, Goehring D, Sandi RF (2002) Disappearance of insectivorous birds from tropical forest fragments. Proc Natl Acad Sci USA 99:263–267. https://doi.org/10.1073/pnas.012616199
Sheard C, Neate-Clegg MHC, Alioravainen N, Jones SEI, Vincent C, MacGregor HEA, Bregman TP, Claramunt S, Tobias JA (2020) Ecological drivers of global gradients in avian dispersal inferred from wing morphology. Nat Commun 11:2463. https://doi.org/10.1038/s41467-020-16313-6
Sherry TW (2021) Sensitivity of tropical insectivorous birds to the Anthropocene: a review of multiple mechanisms and conservation implications. Front Ecol Evol. https://doi.org/10.3389/fevo.2021.662873
Sherry TW, Kent CM, Sanchez NV, Şekercioğlu CH (2020) Insectivorous birds in the Neotropics: ecological radiations, specialization, and coexistence in species-rich communities. Ornithol 137:ukaa049. https://doi.org/10.1093/auk/ukaa049
Sodhi NS, Posa MRC, Lee TM, Warkentin IG (2008) Effects of disturbance or loss of tropical rainforest on birds. Auk 125:511–519. https://doi.org/10.1525/auk.2008.1708
Sonne J, Zanata TB, Gonzalez AMM, Torres NLC, Fjeldsa J, Colwell RK, Tinoco BA, Rahbek C, Dalsgaard B (2019) The distributions of morphologically specialized hummingbirds coincide with floral trait matching across an Andean elevational gradient. Biotropica 51:205–218. https://doi.org/10.1111/btp.12637
Spanhove T, Callens T, Hallmann CA, Pellikka P, Lens L (2014) Nest predation in Afrotropical forest fragments shaped by inverse edge effects, timing of nest initiation and vegetation structure. J Ornithol 155:411–420. https://doi.org/10.1007/s10336-013-1021-9
Stratford JA, Robinson WD (2005) Gulliver travels to the fragmented tropics: geographic variation in mechanisms of avian extinction. Front Ecol Environ 3:91–98. https://doi.org/10.1890/1540-9295(2005)003[0085:GTTTFT]2.0.CO;2
Stratford JA, Stouffer PC (2015) Forest fragmentation alters microhabitat availability for Neotropical terrestrial insectivorous birds. Biol Conserv 188:109–115. https://doi.org/10.1016/j.biocon.2015.01.017
Symonds MRE, Blomberg SP (2014) A primer on phylogenetic generalised least squares. In: Garamszegi L (ed) Modern phylogenetic comparative methods and their application in evolutionary biology. Springer, Berlin
Tallei E, Rivera L, Schaaf A, Scheffer M, Politi N (2022) Post-logging effects on nest predation and avian predator assemblages in a subtropical forest. For Ecol Manag 505:119858. https://doi.org/10.1016/j.foreco.2021.119858
Tejedor-Garavito N, Álvarez E, Arango Caro S, Araujo Murakami A, Blundo C, Boza Espinoza TE, La Torre Cuadros MA, Gaviria J, Gutíerrez N, Jørgensen PM, León B, López Camacho R, Malizia L, Millán B, Moraes M, Pacheco S, Rey Benayas JM, Reynel C, Timaná de la Flor M, Ulloa Ulloa C, Vacas Cruz O, Newton AC (2012) Evaluación del estado de conservación de los bosques montanos en los Andes tropicales. Ecosistemas 21:148–166
Terborgh J (1985) The role of ecotones in the distribution of Andean birds. Ecology 66:1237–1246. https://doi.org/10.2307/1939177
Tinoco BA, Graham CH, Aguilar JM, Schleuning M (2017) Effects of hummingbird morphology on specialization in pollination networks vary with resource availability. Oikos 126:52–60. https://doi.org/10.1111/oik.02998
Tracewski L, Butchart SHM, Di Marco M, Ficetola GF, Rondinini C, Symes A, Wheatley H, Beresford AE, Buchanan GM (2016) Toward quantification of the impact of 21st-century deforestation on the extinction risk of terrestrial vertebrates. Conserv Biol 30:1070–1079. https://doi.org/10.1111/cobi.12715
Trujillo G, Ahumada JA (2005) Artificial nest experiments in a fragmented Neotropical cloud forest. Ornitol Neotrop 16:53–63
Tung Ho LS, Ane C, Lachlan R, Tarpinian K, Feldman R, Yu Q, van der Bijl W, Maspons J, Vos R (2020) Package “phylolm”: phylogenetic linear regression. Version 2(6):2
Vetter D, Hansbauer MM, Vegvari Z, Storch I (2011) Predictors of forest fragmentation sensitivity in Neotropical vertebrates: a quantitative review. Ecography 34:1–8. https://doi.org/10.1111/j.1600-0587.2010.06453.x
Vetter D, Rucker G, Storch I (2013) A meta-analysis of tropical forest edge effects on bird nest predation risk: edge effects in avian nest predation. Biol Conserv 159:382–395. https://doi.org/10.1016/j.biocon.2012.12.023
Visco DM, Sherry TW (2015) Increased abundance, but reduced nest predation in the chestnut-backed antbird in Costa Rican rainforest fragments: surprising impacts of a pervasive snake species. Biol Conserv 188:22–31. https://doi.org/10.1016/j.biocon.2015.01.015
Visco DM, Michel NL, Boyle WA, Sigel BJ, Woltmann S, Sherry TW (2015) Patterns and causes of understory bird declines in human-disturbed tropical forest landscapes: a case study from Central America. Biol Conserv 191:117–129. https://doi.org/10.1016/j.biocon.2015.05.018
Walther BA (2002a) Grounded ground birds and surfing canopy birds: Variation of foraging stratum breadth observed in Neotropical forest birds and tested with simulation models using boundary constraints. Auk 119:658–675. https://doi.org/10.1093/auk/119.3.658
Walther BA (2002b) Vertical stratification and use of vegetation and light habitats by Neotropical forest birds. J Ornithol 143:64–81. https://doi.org/10.1007/BF02465460
Watson JEM, Shanahan DF, Di Marco M, Allan J, Laurance WF, Sanderson EW, Mackey B, Venter O (2016) Catastrophic declines in wilderness areas undermine global environment targets. Curr Biol 26:2929–2934. https://doi.org/10.1016/j.cub.2016.08.049
Weeks BC, O’Brien BK, Chu JJ, Claramunt S, Sheard C, Tobias JA (2022) Morphological adaptations linked to flight efficiency and aerial lifestyle determine natal dispersal distance in birds. Funct Ecol 36:1681–1689. https://doi.org/10.1111/1365-2435.14056
Weeks TL, Betts MG, Pfeifer M, Wolf C, Banks-Leite C, Barbaro L, Barlow J, Cerezo A, Kennedy CM, Kormann UG, Marsh CJ, Olivier PI, Phalan BT, Possingham HP, Wood EM, Tobias JA (2023) Climate-driven variation in dispersal ability predicts responses to forest fragmentation in birds. Nat Ecol Evol. https://doi.org/10.1038/s41559-023-02077-x
Weinstein BG, Graham CH (2017) Persistent bill and corolla matching despite shifting temporal resources in tropical hummingbird-plant interactions. Ecol Lett 20:326–335. https://doi.org/10.1111/ele.12730
Woltmann S, Kreiser BR, Sherry TW (2012) Fine-scale genetic population structure of an understory rainforest bird in Costa Rica. Conserv Geneti 13:925–935. https://doi.org/10.1007/s10592-012-0341-2
Young BE, Sherry TW, Sigel BJ, Woltmann S (2008) Nesting success of Costa Rican lowland rain forest birds in response to edge and isolation effects. Biotropica 40:615–622. https://doi.org/10.1111/j.1744-7429.2008.00406.x
Acknowledgements
This work would not be possible without the heroic efforts of many field technicians, including Julio Bermúdez, Edwin Munera, Duván García-Ramírez, Felipe Castro, María Fernanda Restrepo, and Carolina Revelo. We also thank the staff at Serraniagua, in particular Cristhian Cardona, for their help with field logistics and communicating with landowners. Dr. Elisa Barreto coded the multi-species occupancy model used to generate the response variables. Drs. Andrés Cuervo and Oscar Johnson provided bill height measurements for specimens in the UNAL and LSU collections, respectively. Dr. Oscar Murillo helped to obtain research permits for this work.
Funding
Funding for this project was provided by an Animal Behavior Society grant, a Tinker Foundation Grant, and the Katherine Ordway Endowment for Ecosystem Conservation. No funder had input into the content of the manuscript nor required their approval prior to submission.
Author information
Authors and Affiliations
Contributions
HJ, GJCZ and SKR helped conceive of the idea, HJ collected the field data, and HJ and MJB-D developed the analytical methods and analyzed the data. SKR and GL contributed substantial field gear, field data, and funding. The first draft of the manuscript was written by HHJ and all authors commented on drafts of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Animal capture and manipulation protocols were approved by the Institutional Animal Care and Use Committee of the University of Florida (permit #201709853). Research in Colombia was conducted under the research permit of the Universidad del Valle (permit #1070). Permission to work in forest fragments was obtained from private landowners prior to visiting each site.
Additional information
Communicated by David Hawksworth.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jones, H.H., Bedoya-Durán, M.J., Colorado Z., G.J. et al. Dietary and habitat specialization, eye size, clutch size, and aerial lifestyle predict avian fragmentation sensitivity in an Andean biodiversity hotpot. Biodivers Conserv 32, 4057–4081 (2023). https://doi.org/10.1007/s10531-023-02682-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02682-z