Abstract
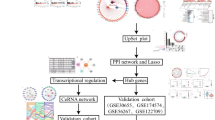
To elucidate the induction of ferroptotic pathways and the transcriptional modulation of pivotal genes in the context of hemorrhagic shock. The R software was used to analyze the GSE64711 dataset, isolating genes relevant to ferroptosis. Enrichment analyses and protein interaction networks were assembled. Using WGCNA hub genes were identified and intersected with ferroptosis-related genes, highlighting hub genes CD44 and MAPK14. In a rat hemorrhagic shock model, cardiac ROS, Fe2+, MDA, and GSH levels were assessed. Key ferroptotic proteins (SLC7A11/GPX4) in myocardial tissues were examined via western blot. Hub genes, CD44 and MAPK14, expressions were confirmed through immunohistochemistry. Analyzing the GSE64711 dataset revealed 337 differentially expressed genes, including 12 linked to ferroptosis. Enrichment analysis highlighted pathways closely related to ferroptosis. Using Genemania, we found these genes mainly affect ROS metabolism and oxidative stress response. WGCNA identified CD44 and MAPK14 as hub genes. Rat myocardial tissue validation showed significant cardiac damage and elevated ROS and MDA levels, and decreased GSH levels in the hemorrhagic shock model. The ferroptotic pathway SLC7A11/GPX4 was activated, and immunohistochemistry showed a significant increase in the expression levels of CD44 and MAPK14 in the hemorrhagic shock rat model. We demonstrated the presence of tissue ferroptosis in hemorrhagic shock by combining bioinformatics analysis with in vivo experimentation. Specifically, we observed the activation of the SLC7A11/GPX4 ferroptotic pathway. Further, CD44 and MAPK14 were identified as hub genes in hemorrhagic shock.







Similar content being viewed by others
Data availability
The article contains the data supporting the findings of this study.
References
Cannon JW (2018) Hemorrhagic shock. N Engl J Med 378(4):370–379
Ahmad FB, Anderson RN (2021) The leading causes of death in the US for 2020. JAMA 325(18):1829–1830
Rhee P, Joseph B, Pandit V, Aziz H, Vercruysse G, Kulvatunyou N, Friese RS (2014) Increasing trauma deaths in the United States. Ann Surg 260(1):13–21
Moreira MA, Irigoyen MC, Saad KR, Saad PF, Koike MK, Montero EF, Martins JL (2016) N-acetylcysteine reduces the renal oxidative stress and apoptosis induced by hemorrhagic shock. J Surg Res 203(1):113–120
Zhang LM, Zhang DX, Fu L, Li Y, Wang XP, Qi MM, Li CC, Song PP, Wang XD, Kong XJ (2019) Carbon monoxide-releasing molecule-3 protects against cortical pyroptosis induced by hemorrhagic shock and resuscitation via mitochondrial regulation. Free Radic Biol Med 141:299–309
Kleinveld DJB, Wirtz MR, van den Brink DP, Maas MAW, Roelofs J, Goslings JC, Hollmann MW, Juffermans NP (2019) Use of a high platelet-to-RBC ratio of 2:1 is more effective in correcting trauma-induced coagulopathy than a ratio of 1:1 in a rat multiple trauma transfusion model. Intensive Care Med Exp 7(Suppl 1):42
Malgras B, Prunet B, Lesaffre X, Boddaert G, Travers S, Cungi PJ, Hornez E, Barbier O, Lefort H, Beaume S et al (2017) Damage control: concept and implementation. J Visc Surg 154(Suppl 1):S19–S29
Sung CW, Sun JT, Huang EP, Shin SD, Song KJ, Hong KJ, Jamaluddin SF, Son DN, Hsieh MJ, Ma MH et al (2022) Association between prehospital fluid resuscitation with crystalloids and outcome of trauma patients in Asia by a cross-national multicenter cohort study. Sci Rep 12(1):4100
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS et al (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149(5):1060–1072
Xl L, Gy Z (2022) Ferroptosis in sepsis: the mechanism, the role and the therapeutic potential. Front Immunol 13:956361
Yan HF, Tuo QZ, Yin QZ, Lei P (2020) The pathological role of ferroptosis in ischemia/reperfusion-related injury. Zool Res 41(3):220–230
Mao C, Liu X, Zhang Y, Lei G, Yan Y, Lee H, Koppula P, Wu S, Zhuang L, Fang B et al (2021) DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer. Nature 593(7860):586–590
Jiang X, Stockwell BR, Conrad M (2021) Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol 22(4):266–282
Conrad M, Pratt DA (2019) The chemical basis of ferroptosis. Nat Chem Biol 15(12):1137–1147
Liu J, Kang R, Tang D (2022) Signaling pathways and defense mechanisms of ferroptosis. FEBS J 289(22):7038–7050
Scortegagna M, Ding K, Oktay Y, Gaur A, Thurmond F, Yan LJ, Marck BT, Matsumoto AM, Shelton JM, Richardson JA et al (2003) Multiple organ pathology, metabolic abnormalities and impaired homeostasis of reactive oxygen species in Epas1−/− mice. Nat Genet 35(4):331–340
van der Pol A, van Gilst WH, Voors AA, van der Meer P (2019) Treating oxidative stress in heart failure: past, present and future. Eur J Heart Fail 21(4):425–435
Tang D, Chen X, Kang R, Kroemer G (2021) Ferroptosis: molecular mechanisms and health implications. Cell Res 31(2):107–125
Rixen D, Siegel JH (2005) Bench-to-bedside review: oxygen debt and its metabolic correlates as quantifiers of the severity of hemorrhagic and post-traumatic shock. Crit Care 9(5):441–453
Duan C, Kuang L, Xiang X, Zhang J, Zhu Y, Wu Y, Yan Q, Liu L, Li T (2020) Activated Drp1-mediated mitochondrial ROS influence the gut microbiome and intestinal barrier after hemorrhagic shock. Aging (Albany NY) 12(2):1397–1416
Vanzant EL, Hilton RE, Lopez CM, Zhang J, Ungaro RF, Gentile LF, Szpila BE, Maier RV, Cuschieri J, Bihorac A et al (2015) Advanced age is associated with worsened outcomes and a unique genomic response in severely injured patients with hemorrhagic shock. Crit Care 19(1):77
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 102(43):15545–15550
Liberzon A, Subramanian A, Pinchback R, Thorvaldsdottir H, Tamayo P, Mesirov JP (2011) Molecular signatures database (MSigDB) 3.0. Bioinformatics 27(12):1739–1740
Franz M, Rodriguez H, Lopes C, Zuberi K, Montojo J, Bader GD, Morris Q (2018) GeneMANIA update 2018. Nucleic Acids Res 46(W1):W60–W64
Abdullah S, Karim M, Legendre M, Rodriguez L, Friedman J, Cotton-Betteridge A, Drury R, Packer J, Guidry C, Duchesne J et al (2021) Hemorrhagic shock and resuscitation causes glycocalyx shedding and endothelial oxidative stress preferentially in the lung and intestinal vasculature. Shock 56(5):803–812
Yao H, Gao Y, Han J, Wang Y, Cai J, Rui Y, Ge X (2022) MKK4 knockdown plays a protective role in hemorrhagic shock-induced liver injury through the JNK pathway. Oxid Med Cell Longev 2022:5074153
Liu R, Wang SM, Liu XQ, Guo SJ, Wang HB, Hu S, Zhou FQ, Sheng ZY (2016) Pyruvate alleviates lipid peroxidation and multiple-organ dysfunction in rats with hemorrhagic shock. Am J Emerg Med 34(3):525–530
Jia B, Ye J, Gan L, Li R, Zhang M, Sun D, Weng L, Xiong Y, Xu J, Zhang P et al (2022) Mitochondrial antioxidant SkQ1 decreases inflammation following hemorrhagic shock by protecting myocardial mitochondria. Front Physiol 13:1047909
Moon DH, Kang DY, Haam SJ, Yumoto T, Tsukahara K, Yamada T, Nakao A, Lee S (2019) Hydrogen gas inhalation ameliorates lung injury after hemorrhagic shock and resuscitation. J Thorac Dis 11(4):1519–1527
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascon S, Hatzios SK, Kagan VE et al (2017) Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171(2):273–285
Liang D, Minikes AM, Jiang X (2022) Ferroptosis at the intersection of lipid metabolism and cellular signaling. Mol Cell 82(12):2215–2227
Fang JY, Richardson BC (2005) The MAPK signalling pathways and colorectal cancer. Lancet Oncol 6(5):322–327
Li L, Hao Y, Zhao Y, Wang H, Zhao X, Jiang Y, Gao F (2018) Ferroptosis is associated with oxygen-glucose deprivation/reoxygenation-induced Sertoli cell death. Int J Mol Med 41(5):3051–3062
Yang L, Jiang L, Sun X, Li J, Wang N, Liu X, Yao X, Zhang C, Deng H, Wang S et al (2022) DEHP induces ferroptosis in testes via p38alpha-lipid ROS circulation and destroys the BTB integrity. Food Chem Toxicol 164:113046
Han S, Wang Y, Ma J, Wang Z, Wang HD, Yuan Q (2020) Sulforaphene inhibits esophageal cancer progression via suppressing SCD and CDH3 expression, and activating the GADD45B-MAP2K3-p38-p53 feedback loop. Cell Death Dis 11(8):713
Liu T, Jiang L, Tavana O, Gu W (2019) The deubiquitylase OTUB1 mediates ferroptosis via stabilization of SLC7A11. Cancer Res 79(8):1913–1924
Lei G, Zhuang L, Gan B (2022) Targeting ferroptosis as a vulnerability in cancer. Nat Rev Cancer 22(7):381–396
Muller S, Sindikubwabo F, Caneque T, Lafon A, Versini A, Lombard B, Loew D, Wu TD, Ginestier C, Charafe-Jauffret E et al (2020) CD44 regulates epigenetic plasticity by mediating iron endocytosis. Nat Chem 12(10):929–938
Mees ST, Gwinner M, Marx K, Faendrich F, Schroeder J, Haier J, Kahlke V (2008) Influence of sex and age on morphological organ damage after hemorrhagic shock. Shock 29(6):670–674
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 82274283), the High-level Public Healh Technical Personnel Construction Project Training Plan (No. XKGG-01-030), the Research on Prevention and Treatment of 2019-nCov with Traditional Chinese Medicine (No. 2020YFC0841500), and Beijing Hospitals Authority Youth Programme (Code: QML20231002).
Author information
Authors and Affiliations
Contributions
All authors listed have significantly contributed to the development and the writing of this article. HRY and SSH provided ideas, conducted data retrieval and carried out the experiment. YD and YCW suggested for the analysis. CXZ, YHH, YTJ and FYL visualized the data and advised on the study. YHG designed and supervised the study.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The animal experimental protocol was approved by the Animal Ethics Committee of Beijing Hospital of Traditional Chinese Medicine, Capital Medical University (Approval No. BJTCM-R-20220501).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ye, H., He, S., Du, Y. et al. Involvement of CD44 and MAPK14-mediated ferroptosis in hemorrhagic shock. Apoptosis 29, 154–168 (2024). https://doi.org/10.1007/s10495-023-01894-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01894-6