Abstract
An actinomycete strain, 2603PH03T, was isolated from a mangrove rhizosphere soil sample collected in Wenchang, China. Phylogenetic analysis of the 16S rRNA gene sequence of strain 2603PH03T indicated high similarity to Verrucosispora gifthornensis DSM 44337T (99.4%), Verrucosispora andamanensis (99.4%), Verrucosispora fiedleri MG-37T (99.4%) and Verrucosispora maris AB18-032T (99.4%). The cell wall was found to contain meso-diaminopimelic acid and glycine. The major menaquinones were identified as MK-9(H4), MK-9(H6) and MK-9(H8), with MK-9(H2), MK-10(H2), MK-9(H10) and MK-10(H6) as minor components. The characteristic whole cell sugars were found to be xylose and mannose. The phospholipid profile was found to contain phosphatidylethanolamine, diphosphatidylglycerol, phosphatidylinositol mannoside, phosphatidylinositol, phosphatidylserine and an unidentified phospholipid. The DNA G+C content was determined to be 70.1 mol%. The results of physiological and biochemical tests and low DNA-DNA relatedness readily distinguished the isolate from the closely related species. On the basis of these phenotypic and genotypic data, strain 2603PH03T is concluded to represent a novel species of the genus Verrucosispora, for which the name Verrucosispora rhizosphaerae sp. nov. is proposed. The type strain is 2603PH03T (=CCTCC AA 2016023T = DSM 45673T).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Verrucosispora was established by Rheims et al. (1998) as a member of the family Micromonosporaceae. Members of the genus Verrucosispora form a well-developed substrate mycelium, lacking aerial mycelium or sporangia. The major menaquinones are MK-9(H4) and MK-9(H6), meso-diaminopimelic acid is the diagnostic diamino acid of the peptidoglycan. The phenotypic, chemotaxonomic and phylogenetic characteristics of the genus (Goodfellow et al. 2012; Stackebrandt 2012) and genus-specific primers (Xie et al. 2011) allow the genus to be distinguished from other genera classified in the family Micromonosporaceae. There is considerable interest in members of the genus Verrucosispora as they are a source of novel antibiotics including, for example, abyssomicins (Bister et al. 2004) and Proximicin A (Schneider et al. 2008) from Verrucosispora maris (Goodfellow et al. 2012), gifhornenolones A and B from Verrucosispora gifhornensis (Shirai et al. 2010), proximicins A-C from Verrucosispora fiedleri MG-37 (Fiedler et al. 2008), Thiocoraline A from Verrucosispora sp. WMMA107 (Wyche et al. 2011), Butrepyrazinone from Verrucosispora sp. K51G (Kyeremeh et al. 2014) and Brevianamide F from Verrucosispora sp. MS100047 (Huang et al. 2016).
In the course of investigating mangrove as an actinomycete resource in Hainan Province, China (Hong et al. 2009), strain 2603PH03T was isolated from a mangrove rhizosphere soil sample. We present here a polyphasic taxonomic characterisation of strain 2603PH03T.
Materials and methods
Isolation and maintenance of isolate
A mangrove rhizosphere soil sample was collected in Wenchang, Hainan, China (GPS: N19°36.506′, E110°47.746′). The soil sample was air dried at room temperature for a week. The dried soil sample (0.1 g) was added to 0.9 mL of sterile water. The resultant 10−1 dilution was initially ribolised with a FastPrep-Instrument for 2 s at a speed of 4.0 m/s, and then diluted to 10−2 and 10−3 (Xie et al. 2011). The soil suspensions were spread on the surfaces of agar plates of PH medium. The medium consisted of l-arabinose (1.0 g), salicin (1.0 g), l-phenylalamine (0.1 g), l-histidine (0.1 g) and 15 g agar, supplemented with base mineral salts (Na2HPO4, 0.8 g; KH2PO4, 0.2 g; MgSO4·7H2O, 0.2 g; CaCl2·2H2O, 0.2 g; FeCl3·6H2O, 5.0 mg; Na2MoO4·2H2O, 1 mg), potassium dichromate (30 ml/l), novobiocin (5 mg/l) and nystatin (30 mg/l), in 1.0 L distilled water at pH 7.4. After 4 weeks of aerobic incubation at 28 °C, the isolate, which formed a vivid orange yellow colony, was transferred and purified on yeast extract-malt extract (ISP 2) agar (Shirling and Gottlieb 1966) and maintained as working cultures on ATCC 172 medium (http://www.atcc.org).
The reference strains V. gifhornensis DSM 44337T, V. maris AB18-032T and V. fiedleri MG-37T were obtained from Prof Michael Goodfellow (University of Newcastle, UK). Reference strains were cultured under the same conditions as strain 2603PH03T in comparative tests.
Phenotypic characteristics
Cultural characteristics of strain 2603PH03T and the reference strains were determined following growth on tap-water agar, Czapek’s agar (Raper and Fennell 1965), GYM agar (Ochi 1987), ATCC 172 medium, M 8 agar (Castiglione et al. 2008), modified Bennett agar and ISP 1–7 media for 14–21 days at 28 °C. The ISCC-NBS colour charts were used to determine the designations of colony colours (Kelly1964). The morphological characteristics of strain M4I47T were assessed by scanning electron microscopy (Zeiss, Evo18) of 21-day-old cultures grown on ISP 2 medium. The Gram reaction was performed according to Gregersen (1978) by using KOH for cell lysis. The pH (4–11) and NaCl (0–10%) tolerance for growth were determined on ISP 2 medium for 14–21 days at 28 °C. Allantoin hydrolysis was carried out by the method of Gordon (1967). Tests for the degradation (%, w/v) of adenine, elastin, gelatin, guanine, starch, l-tyrosine, uric acid, xanthine and xylan used modified Bennett’s agar as the basal medium (Tan et al. 2006). Carbon source utilisation was tested by using ISP 9 medium (Shirling and Gottlieb 1966) supplemented with 1% (final concentration) carbon source. The utilisation of amino acids as nitrogen source was tested as described by Williams et al. (1983). The other physiological and biochemical characteristics of strain 2603PH03T and the reference strains were tested by using media and methods described by Williams et al. (1983) and Kämpfer et al. (1991).
Chemotaxonomy
Biomass for molecular systematic and most of the chemotaxonomic studies was obtained and washed after growing in yeast extract malt extract broth (ISP 2) at 28 °C for 7–14 days on a rotary shaker (220 rpm). Cell wall amino acid and whole cell sugars were analysed as the procedure of Lechevalier and Lechevalier (1980). The N-acyl group of the muramic acid in the peptidoglycan was determined by the method of Uchida and Aida (1977). The presence of mycolic acids was determined by the method of Minnikin et al. (1975). Phospholipids in cells were extracted and identified by the method of Minnikin et al. (1984). Fatty acids were extracted by the method of Sasser (1990) and the composition was determined by Sherlock Microbial Identification System (MIDI). The fatty acid methyl esters were identified by using the Microbial Identification software package (Sherlock Version 6.0; MIDI database: ACTIN6). Menaquinones were extracted according to Minnikin et al. (1984) and analysed by an established HPLC procedure (Wang et al. 2011).
Phylogenetic analyses
Genomic DNA extraction, PCR-mediated amplification of the 16S rRNA gene and sequencing of the PCR products were carried out as described by Nakajima et al. (1999). The 16S rRNA gene sequence of strain 2603PH03T was aligned with multiple sequences obtained from the GenBank/EMBL/DDBJ databases using CLUSTAL-X software (Version 2.1; Larkin et al. 2007). Alignment was manually verified and adjusted prior to the construction of phylogenetic trees. The phylogenetic trees were generated with the neighbor joining (Saitou and Nei 1987), maximum-likelihood (Felsenstein 1981) and maximum-parsimony tree-making algorithms (Kluge and Farris 1969) using MEGA version 6.0 software (Tamura et al. 2013). Phylogenetic distances were calculated with Kimura’s 2-parameter model (Kimura 1980) and the stability of the tree topologies was evaluated by bootstrap analysis (Felsenstein 1985) based on 1000 resamplings. Salinispora arenicola CNB-643T was used as an outgroup. The values for the 16S rRNA gene sequence similarities between strains were determined using the EzBioCloud (Yoon et al. 2016).
DNA relatedness studies
Genomic DNA of strain 2603PH03T was obtained as described by Pospiech and Neumann (1995). The DNA G+C content of strain 2603PH03T was determined by the HPLC method (Mesbah et al. 1989a, b). The level of DNA relatedness between strain 2603PH03T and the related strains were measured on nylon membranes using the method described by Wang et al. (2011).
Results and discussion
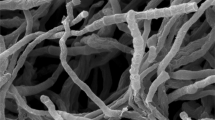
The morphological properties of strain 2603PH03T are consistent with its classification as a member of the genus Verrucosispora (Rheims et al. 1998). Strain 2603PH03T was observed to produce well-developed and branched substrate mycelium on ISP 2 medium, with colonies approximately 0.3–0.4 μm in diameter, but no aerial hyphae. Single unevenly warty–surfaced spores are formed on the substrate hypha, with a diameter of approximate 0.6–0.8 μm (Supplementary Fig. S1). Good growth was observed on ISP 1, ISP 2, M 8,ATCC 172, GYM and modified Bennett agar; moderate growth was observed on ISP 5, ISP 6, ISP 7 and tap water agar; poor growth was observed on ISP 3 and ISP 4 media (Supplementary Table S1). The colour of the substrate hyphae was vivid orange yellow to strong orange yellow. No soluble pigment was produced on any of the ISP media tested. Other physiological characteristics are given in the type strain description and Table 1. In addition, the strain is positive for adenine, starch, casein, urea, gelatin, xanthine, elastin, l-tyrosine hydrolysis and nitrate reduction, but negative for allantoin, aesculin, arbutin, guanine, xylan hydrolysis and H2S production. d-glucose, l-arabinose, l-fucose, d-fructose, d-mannose, d(+)-melibose, d(+)-arabinose, lactose, d-xylose, maltose, frucose sucrose, melezitose, turanose, d-raffinose, sorbitol, adonitol, glycerol, maltitriose, amygdalin, erythritol, ethanol, arbutin and salicin can be utilised as sole carbon sources but d(+)-galactose, l-rhamnose, d-mannitol, d(+)-trehalose, cellobiose, l-ribose and dulcitol are not. l-alanine, l-arginine, l-histidine, l-phenylalanine, l-serine, l-threonine, l-tyrosine and l-proline are utilised as sole nitrogen source but l-cysteine, l-glycine, l-methionine, l-valine, l-asparagine, and l-glutamic acid are not.
The cell wall of the novel isolate was found to contain meso-diaminopimelic acid and glycine. The whole cell sugars were identified as xylose and mannose. The acyl type of the cell wall peptidoglycan was found to be glycolyl. Mycolic acids were not detected. The major menaquinones (>5%) were identified as MK-9(H4) (65.0%), MK-9(H6) (17.6%) and MK-9(H8) (9.6%), with MK-9(H2) (2.6%), MK-10(H2) (2.4%) and MK-9(H10) (2.3%) MK-10(H6) (0.5%) as minor components. The characteristic phospholipids were identified as phosphatidylethanolamine (PE), diphosphatidylglycerol (DPG), phosphatidylinositol mannosides (PIMs), phosphatidylinositol (PI), phosphatidylserine (PS) and an unidentified phospholipid (PL), corresponding to phospholipid type PII of Lechevalier et al. (1977) (Supplementary Fig. S2). Significant cellular fatty acids were identified as iso-C16:0 (21.4%), iso-C15:0 (20.7%), iso-C18:0 (14.4%), C17:1ω10c (8.8%) C16:0 (8.6%), C17:0 (5.4%), C15:0 (5.1%), anteiso-C15:0 (3.9%), anteiso-C17:0 (2.6%), C18:0 (2.5%), iso-C14:0 (1.9%), anteiso-C16:0 (1.1%), C18:1ω9c (1.1%), iso-C17:0 (1.0%)10-methyl-C16:0 (1.0%), C14:0 (0.4%) and C16:1ω9c (0.3%). The G+C content of the DNA was determined to be 70.1 mol%.
The almost complete 16S rRNA gene sequence of strain 2603PH03T (1475 nt, GenBank accession number HQ123438) was compared with sequences of representatives of the genus Verrucosispora. The 16S rRNA gene sequence similarities of strain 2603PH03T with the type strains of V. gifthornensis, Verrucosispora andamanensis (Supong et al. 2013), V. fiedleri (Goodfellow et al. 2013), V. maris (Goodfellow et al. 2012), Verrucosispora wenchangensis (Xie et al. 2012), Verrucosispora sediminis (Dai et al. 2010), Verrucosispora lutea (Liao et al. 2009) and Verrucosispora qiuiae (Xi et al. 2012) were 99.4, 99.4, 99.4, 99.2, 99.1, 99.0 and 99.0%, respectively. Based on 16S rRNA gene sequence analysis, strain 2603PH03T formed a phyletic line on the periphery of the 16S rRNA gene subclade comprised of the type strains of V. gifhornensis, V. fiedleri and V. maris, a relationship which was supported by all of the tree-making algorithms and by a 93% bootstrap value (Fig. 1; Supplementary Fig. S3a and b). Further study showed that the DNA relatedness between strain 2603PH03T and the type strains of V. gifthornensis, V. fiedleri, V. maris were 21.8 ± 2.0, 16.6 ± 4.5, and 18.9 ± 4.0%, respectively, all of which are below the 70% threshold value proposed by Wayne et al. (1987) as the key marker for the identification of a novel prokaryotic species.
Neighbour-joining phylogenetic tree (Saitou and Nei 1987), based on almost-complete 16S rRNA gene sequences (1407 nt), showing the relationships between strain 2603PH03T and other members of the genus Verrucosispora. Salinispora arenicola CNB-643T was used as an outgroup. Numbers at branch points indicate bootstrap percentages (based on 1000 replicates); only values >50% are indicated. Bar 0.002 substitutions per nucleotide position. Asterisks indicate branches of the tree that were also found maximum-likelihood (Felsenstein 1981) and maximum-parsimony methods (Kluge and Farris)
The characteristics shown in Table 1 indicated that strain 2603PH03T has some different physiological and biochemical characteristics compared to its closely related phylogenetic neighbours, and can be distinguished on the basis of its inability to degrade allantoin, capacity to grow in fructose and sorbitol as sole carbon source and inability to use l-valine as sole nitrogen source. It is evident from the phenotypic, chemotaxonomic, genotypic and phylogenetic data presented above that strain 2603PH03T can be distinguished from previously described Verrucosispora species. Therefore, strain 2603PH03T is concluded to represent a novel species of the genus Verrucosispora, for which the name Verrucosispora rhizosphaerae sp. nov. is proposed. The Digital Protologue database TaxoNumber (Rosselló-Móra et al. 2017) for strain 2603PH03T is TA00249.
Description of Verrucosispora rhizosphaerae sp. nov.
Verrucosispora rhizosphaerae (rhi.zo.sphae’rae. Gr. n. rhiza, a root; L. n. sphaera, a ball, sphere; N.L. fem. n. rhizosphaera, rhizosphere; N.L. gen. n. rhizosphaerae, of the rhizosphere, pertaining to the soil from which the type strain was isolated).
Aerobic, Gram-positive, mesophilic actinomycete that forms well-developed and branched substrate hyphae; aerial mycelium and spore vesicles are not formed. Single spores are formed on the substrate hyphae. Grows well on ISP 1, ISP 2, M 8,ATCC 172, GYM and modified Bennett media agar. The substrate hyphae are vivid orange yellow to strong orange yellow. The pH range for growth is 7-10, with an optimum at 7. The maximum NaCl concentration for growth is 10%. Unable to degrade allantoin. Can grow using fructose and sorbitol as sole carbon source. Unable to use l-valine as sole nitrogen source. The cell wall contains meso-diaminopimelic acid and glycine. The whole cell sugars are xylose and mannose. The acyl type of the cell wall peptidoglycan is glycolyl. Mycolic acids are not present. The major menaquinones (>5%) are MK-9(H4), MK-9(H6) and MK-9(H8) (9.6%). The major fatty acids (>5%) are iso-C16:0, iso-C15:0, iso-C18:0, C17:1ω10c, C16:0, C17:0 and C15:0. The characteristic phospholipids are phosphatidylethanolamine, diphosphatidylglycerol, phosphatidylinositol mannoside, phosphatidylinositol, phosphatidylserine and an unidentified phospholipid. The G+C content of the DNA of the type strain is 70.1 mol%.
The type strain, 2603PH03T (=CCTCC AA 2016023T = DSM 45673), was isolated from a mangrove rhizosphere soil sample that was collected in Wenchang, Hainan, China. The GenBank/EMBL/DDJB accession number for the 16S rRNA gene sequence of the type strain 2603PH03T is HQ123438.
References
Bister B, Bischoff D, Ströbele M, Riedlinger J, Reicke A, Wolter F, Bull AT, Zähner H, Fiedler HP, Süssmuth RD (2004) Abyssomicin C-A polycyclicantibiotic from a marine Verrucosispora strain as an inhibitor of the p-aminobenzoic acid/tetrahydrofolate biosynthesis pathway. Angew Chem Int Ed Engl 43:2574–2576
Castiglione F, Lazzarini A, Carrano L, Corti E, Ciciliato I, Gastaldo L, Candiani P, Losi D, Marinelli F, Selva E (2008) Determining the structure and mode of action of microbisporicin, a potent lantibiotic active against multiresistant pathogens. Chem Biol 15:22–31
Dai HQ, Wang J, Xin YH, Pei G, Tang SK, Ren B, Ward A, Ruan JS, Li WJ, Zhang LX (2010) Verrucosispora sediminis sp. nov., a novel cyclodipeptide-producing actinomycete from the South China Sea. Int J Syst Evol Microbiol 60:1807–1812
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–789
Fiedler HP, Bruntner C, Riedlinger J, Bull AT, Knutsen G, Goodfellow M, Jones A, Maldonado L, Pathom-aree W, Beil W, Schneider K, Keller S, Sussmuth RD (2008) Proximicin A, B and C, novel aminofuran antibiotic and anticancer compounds isolated from marine strains of the actinomycete Verrucosispora. J Antibiot 61(3):158–163
Goodfellow M, Stach JE, Brown R, Bonda AN, Jones AL, Mexson J, Fiedler HP, Zucchi TD, Bull AT (2012) Verrucosispora maris sp. nov., a novel deep-sea actinomycete isolated from a marine sediment which produces abyssomicins. Antonie Van Leeuwenhoek 101:185–193
Goodfellow M, Brown R, Ahmed L, Pathom-Aree W, Bull AT, Jones AL, Stach JE, Zucchi TD, Zhang L, Wang J (2013) Verrucosispora fiedleri sp. nov., an actinomycete isolated from a fjord sediment which synthesizes proximicins. Antonie Van Leeuwenhoek 103(3):493–502
Gordon RE (1967) The taxonomy of soil bacteria. In: Gray TRG, Parkinson D (eds) Ecology of soil bacteria. Liverpool University Press, Liverpool, pp 293–321
Gregersen T (1978) Rapid method for distinction of Gram-negative from Gram-positive bacteria. Eur J Appl Microbiol 5:123–127
Hong K, Gao AH, Xie QY, Gao H, Zhuang L, Lin HP, Yu HP, Li J, Yao XS, Goodfellow M, Ruan JS (2009) Actinomycetes for marine drug discovery isolated from mangrove soils and plants in China. Mar Drugs 7:24–44
Huang P, Xie F, Ren B, Wang Q, Wang J, Wang Q, Abdel-Mageed WM, Liu M, Han J, Oyeleye A, Shen J, Song F, Dai H, Liu X, Zhang L (2016) Anti-MRSA and anti-TB metabolites from marine-derived Verrucosispora sp. MS100047. Appl Microbiol Biotechnol 100(17):7437–7447
Kämpfer P, Kroppenstedt RM, Dott W (1991) A numerical classification of the genera Streptomyces and Streptoverticillium using miniaturized physiological tests. J Gen Microbiol 137:1831–1891
Kelly KL (1964) Inter-society color council–national bureau of standards color name charts illustrated with centroid colors. US Government Printing Office, Washington
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kluge AG, Farris FS (1969) Quantitative phyletics and the evolution of anurans. Syst Zool 18:1–32
Kyeremeh K, Acquah KS, Camas M, Tabudravu J, Houssen W, Deng H, Jaspars M (2014) Butrepyrazinone, a new pyrazinone with an unusual methylation pattern from a Ghanaian Verrucosispora sp. K51G. Mar Drugs 12(10):5197–5208
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Lechevalier MP, Lechevalier HA (1980) The chemotaxonomy of actinomycetes. In: Dietz A, Thayer J (eds) Actinomycete taxonomy (Special Publication No. 6). Society for Industrial Microbiology, Arlington, pp 227–291
Lechevalier MP, BiévreC De, Lechevalier HA (1977) Chemotaxonomy of aerobic actinomycetes: phospholipid composition. Biochem Syst Ecol 5:249–260
Liao ZL, Tang SK, Guo L, Zhang YQ, Tian XP, Jiang CL, Xu LH, Li WJ (2009) Verrucosispora lutea sp. nov., isolated from a mangrove sediment sample. Int J Syst Evol Microbiol 59:2269–2273
Mesbah M, Premachandran U, Whitman WB (1989a) Precise measurement of the G+C content of deoxyribonucleic acid by high-performance liquid chromatography. Int J Syst Bacteriol 39:159–167
Mesbah M, Premachandran U, Whitman WB (1989b) Precise measurement of the G+C content of deoxyribonucleic acid by high-performance liquid chromatography. Int J Syst Bacteriol 39:159–167
Minnikin DE, Alshamaony L, Goodfellow M (1975) Differentiation of Mycobacterium, Nocardia and related taxa by thin-layer-chromatographic analyses of whole-cell methanolysates. J Gen Microbiol 88:200–204
Minnikin DE, O’Donnell AG, Goodfellow M, AldersonG Athalye M, Schaal A, Parlett JK (1984) An integrated procedure for the extraction of isoprenoid quinones and polar lipids. J Microbiol Methods 2:233–241
Nakajima Y, Kitpreechavanich V, Suzuki K, Kudo T (1999) Microbispora caponicas sp. nov., a new species of the genus Microbispora isolated from Thai soil. Int J Syst Bacteriol 49:1761–1767
Ochi K (1987) Metabolic initiation of differentiation and secondary metabolism by Streptomyces griseus: significance of the stringent response (ppGpp) and GTP content in relation to A factor. J Bacteriol 169:3608–3616
Pospiech A, Neumann B (1995) A versatile quick-prep of genomic DNA from gram-positive bacteria. Trends Gene 11:217–218
Raper KB, Fennell DI (1965) The Genus Aspergillus. Williams and Wilkins, Balti-more, p 686
Rheims H, Schumann P, Rohde M, Stackebrandt E (1998) Verrucosispora gifhornensis gen. nov., sp. nov., a new member of the actinobacterial family Micromonosporaceae. Int J Syst Bacteriol 48:1119–1127
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenetic tree. Mol Biol Evol 4:406–425
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids. Technical Note 101. Newark, DE: Microbial ID
Schneider K, Keller S, Wolter FE, Röglin L, Beil W, Seitz O, Nicholson G, Bruntner C, Riedlinger J, Fiedler HP, Süssmuth RD (2008) Proximicins A, B, and C-antitumor furan analogues of netropsin from the marine actinomycete Verrucosispora induce upregulation of p53 and the cyclin kinase inhibitor p21. Angew Chem Int Ed Engl 47(17):3258–3261
Shirai M, Okuda M, Motohashi K, Imoto M, Furihata K, Matsuo Y, Katsuta A, Shizuri Y, Seto H (2010) Terpenoids produced by actinomycetes: isolation, structural elucidation and biosynthesis of new diterpenes, gifhornenolones A and B from Verrucosispora gifhornensis YM28-088. J Antibiot 63(5):245–250
Shirling EB, Gottlieb D (1966) Methods for characterization of Streptomyces species. Int J Syst Bacteriol 16:313–340
Stackebrandt E (2012) Genus XVI. Verrucosispora Rheims, Schumann, Rohde, Stackebrandt, 1998. In: Goodfellow M, Kampfer P, Busse H-J, Trujillo ME, Suzuki K-I, Ludwig W, Whitman WB (eds) Bergey’s manual of systematic bacteriology. The actinobacteria, Part B, vol 5, 2nd edn. Springer, New York, pp 1124–1127
Supong K, Suriyachadkun C, Suwanborirux K, Pittayakhajonwut P, Thawai C (2013) Verrucosispora andamanensis sp. nov., isolated from a marine sponge. Int J Syst Evol Microbiol 63:3970–3974
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Tan GYA, Robinson S, Lacey E, Goodfellow M (2006) Evaluation of Amycolatopsis diversity in soil using genus-specific primers and novel selective media. Syst Appl Microbiol 29:557–569
Uchida K, Aida A (1977) Acyl type of bacterial cell wall: its simple identification by colorimetricmethod. J Gen Appl Microbiol 23:249–260
Wang C, Xu XX, Qu Z, Wang HL, Lin HP, Xie QY, Ruan JS, Hong K (2011) Micromonospora rhizosphaerae sp. nov., isolated from mangrove rhizosphere soil. Int J Syst Evol Microbiol 61:320–324
Wayne LG, Brenner DJ, Colwell RR, Grimont PAD, Kandler O, Krichevsky MI, Moore LH et al (1987) International 21 Committee on Systematic Bacteriology. Report of the ad hoc committee on reconciliation 22 of approaches to bacterial systematics. Int J Syst Bacteriol 37:463–464
Williams ST, Goodfellow M, Alderson G, Wellington EMH, Sneath PHA, Sackin MJ (1983) Numerical classification of Streptomyces and related genera. J Gen Microbiol 129:1743–1813
Wyche TP, Hou Y, Braun D, Cohen HC, Xiong MP, Bugni TS (2011) First natural analogs of the cytotoxic thiodepsipeptide thiocoraline A from a marine Verrucosispora sp. J Org Chem 76(16):6542–6547
Xi L, Zhang L, Ruan J, Huang Y (2012) Verrucosispora qiuiae sp. nov., isolated from mangrove swamp, and emended description of the genus Verrucosispora. Int J Syst Evol Microbiol 62:1564–1569
Xie QY, Hong K, Goodfellow M (2011) Genus-specific primers targeting the 16S rRNA gene for PCR detection of members of the genus Verrucosispora. Antonie Van Leeuwenhoek 100:117–128
Xie QY, Lin HP, Li L, Brown R, Goodfellow M, Deng Z, Hong K (2012) Verrucosispora wenchangensis sp. nov., isolated from mangrove soil. Antonie Van Leeuwenhoek 102:1–7
Yoon SH, Ha SM, Kwon S, Lim J, Kim Y, Seo H, Chun J (2016) Introducing EzBioCloud: a taxonomically united database of 16S rRNA and whole genome assemblies. Int J Syst Evol Microbiol 66:5430–5436
Acknowledgements
This investigation was co-supported by the Open Research Fund Program of Guangxi Key Lab of Mangrove Conservation and Utilization, Natural Science Foundation of Hainan Province of China under Grant No. 20153143, Central Public-interest Scientific Institution Basal Research Fund for Innovative Research Team Program of CATAS (No. 17CXTD-15), and Special Fund for Agro-Scientific Research in the Public Interest (201303117).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involving animal and human rights
This article does not contain any studies with animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Xie, Qy., Bao, Xd., Ma, Qy. et al. Verrucosispora rhizosphaerae sp. nov., isolated from mangrove rhizosphere soil. Antonie van Leeuwenhoek 111, 125–133 (2018). https://doi.org/10.1007/s10482-017-0933-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-017-0933-4