Abstract
Objectives
To assess short- and long-term repeatability of T2 relaxation time measurements of the knee articular cartilage.
Materials and methods
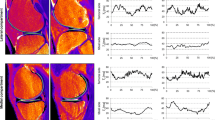
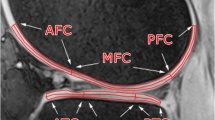
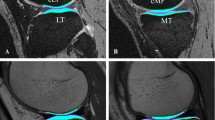
The right knees of nine asymptomatic volunteers (age 30–38 years, five male, four female) were imaged at 1.5 T in three sessions 1 and 2 weeks apart. To observe short-term repeatability, the measurements were repeated three times within one of the three imaging sessions for each volunteer. T2 relaxation time was mapped using a multi-slice multi-echo spin echo sequence in axial and sagittal planes. Cartilage was manually segmented and repeatability, as measured by root-mean-square coefficient of variation (CVRMS) was evaluated both for the entire bulk cartilage of each joint surface in the slice and separately for each region of interest (ROI) at different topographical locations and separately for the superficial and deep half of each ROI.
Results
For bulk T2, the long-term repeatability was 3.2, 5.4, and 3.7 %, and the short-term reproducibility was 3.9, 3.9, and 3.4 % for bulk femoral, tibial, and patellar cartilage, respectively. There were no significant differences between long-term and short-term repeatability in superficial or deep cartilage when comparing CVRMS values (p = 0.338 and 0.700, respectively). For individual ROIs, the repeatability varied between 2.5 and 22.2 % depending on the topographical location.
Conclusion
The current results show mostly good repeatability. However, there were remarkable variations of T2 between bulk cartilage and different ROIs, bulk cartilage showing better repeatability. With careful patient positioning T2 can be accurately determined for different cartilage surfaces.

Similar content being viewed by others
References
Felson DT (2009) Developments in the clinical understanding of osteoarthritis. Arthritis Res Ther 11:203
Buckwalter JA, Mankin HJ (1998) Articular cartilage: degeneration and osteoarthritis, repair, regeneration, and transplantation. Instr Course Lect 47:487–504
Eckstein F, Burstein D, Link TM (2006) Quantitative MRI of cartilage and bone: degenerative changes in osteoarthritis. NMR Biomed 19:822–854
Fragonas E, Mlynarik V, Jellus V, Micali F, Piras A, Toffanin R, Rizzo R, Vittur F (1998) Correlation between biochemical composition and magnetic resonance appearance of articular cartilage. Osteoarthr Cartil 6:24–32
Lüsse S, Claassen H, Gehrke T, Hassenpflug J, Schünke M, Heller M, Glüer C- (2000) Evaluation of water content by spatially resolved transverse relaxation times of human articular cartilage. Magn Reson Imaging 18:423–430
Nieminen MT, Töyräs J, Rieppo J, Hakumäki JM, Silvennoinen J, Helminen HJ, Jurvelin JS (2000) Quantitative MR microscopy of enzymatically degraded articular cartilage. Magn Reson Med 43:676–681
Grunder W, Wagner M, Werner A (1998) MR-microscopic visualization of anisotropic internal cartilage structures using the magic angle technique. Magn Reson Med 39:376–382
Nieminen MT, Rieppo J, Töyräs J, Hakumäki JM, Silvennoinen J, Hyttinen MM, Helminen HJ, Jurvelin JS (2001) T2 relaxation reveals spatial collagen architecture in articular cartilage: a comparative quantitative MRI and polarized light microscopic study. Magn Reson Med 46:487–493
Xia Y, Moody JB, Alhadlaq H (2002) Orientational dependence of T2 relaxation in articular cartilage: a microscopic MRI (μMRI) study. Magn Reson Med 48:460–469
Nieminen MT, Töyräs J, Laasanen MS, Silvennoinen J, Helminen HJ, Jurvelin JS (2004) Prediction of biomechanical properties of articular cartilage with quantitative magnetic resonance imaging. J Biomech 37:321–328
Wayne JS, Kraft KA, Shields KJ, Yin C, Owen JR, Disler DG (2003) MR imaging of normal and matrix-depleted cartilage: correlation with biomechanical function and biochemical composition. Radiology 228:493–499
Kurkijärvi JE, Nissi MJ, Kiviranta I, Jurvelin JS, Nieminen MT (2004) Delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) and T2 characteristics of human knee articular cartilage: topographical variation and relationships to mechanical properties. Magn Reson Med 52:41–46
Lammentausta E, Kiviranta P, Nissi MJ, Laasanen MS, Kiviranta I, Nieminen MT, Jurvelin JS (2006) T2 relaxation time and delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) of human patellar cartilage at 1.5 T and 9.4 T: relationships with tissue mechanical properties. J Orthop Res 24:366–374
Nissi MJ, Töyräs J, Laasanen MS, Rieppo J, Saarakkala S, Lappalainen R, Jurvelin JS, Nieminen MT (2004) Proteoglycan and collagen sensitive MRI evaluation of normal and degenerated articular cartilage. J Orthop Res 22:557–564
David-Vaudey E, Ghosh S, Ries M, Majumdar S (2004) T2 relaxation time measurements in osteoarthritis. Magn Reson Imaging 22:673–682
Dunn TC, Lu Y, Jin H, Ries MD, Majumdar S (2004) T2 relaxation time of cartilage at MR imaging: comparison with severity of knee osteoarthritis. Radiology 232:592–598
Liebl H, Joseph G, Nevitt MC, Singh N, Heilmeier U, Karupppasamy S, Jungmann PM, McCulloch CE, Lynch JA, Lane NE, Link TM (2015) Early T2 changes predict onset of radiographic knee osteoarthritis: data from the osteoarthritis initiative. Ann Rheum Dis 74:1353–1359
Glaser C, Mendlik T, Dinges J, Weber J, Stahl R, Trumm C, Reiser M (2006) Global and regional reproducibility of T2 relaxation time measurements in human patellar cartilage. Magn Reson Med 56:527–534
Koff MF, Parratte S, Amrami KK, Kaufman KR (2009) Examiner repeatability of patellar cartilage T2 values. Magn Reson Imaging 27:131–136
Horng A, Raya JG, Weber J, Weckbach S, Nixdorf S, Reiser MF, Glaser C (2010) Reproducibility of T2 mapping in healthy and osteoarthritic human tibial cartilage: precise enough to detect true degenerative change? In: Proceedings of the 22nd European Congress of Radiology, Vienna, p 10
Atkinson G, Nevill AM (1998) Statistical methods for assessing measurement error (reliability) in variables relevant to sports medicine. Sports Med 26:217–238
Fleiss JL (1986) The design and analysis of clinical experiments. Wiley, New York
Glaser C, Horng A, Mendlik T, Weckbach S, Hoffmann R-, Wagner S, Raya JG, Horger W, Reiser M (2007) T2 relaxation time in patellar cartilage—global and regional reproducibility at 1.5 Tesla and 3 Tesla. RöFo 179:146–152
Jordan JM, Sowers MF, Messier SP, Bradley J, Arangio G, Katz JN, Losina E, Rovati L, Bachtell N, Cooper C, Spector T, Zhang W, Gardiner J, Wahba M (2011) Methodologic issues in clinical trials for prevention or risk reduction in osteoarthritis. Osteoarth Cartil 19:500–508
Meyer JS (2008) Musculoskeletal imaging at 3 T: imaging and optimization. Pediatr Radiol 38:S243–S245
Waterton JC, Solloway S, Foster JE, Keen MC, Gandy S, Middleton BJ, Maciewicz RA, Watt I, Dieppe PA, Taylor CJ (2000) Diurnal variation in the femoral articular cartilage of the knee in young adult humans. Magn Reson Med 43:126–132
Nishii T, Kuroda K, Matsuoka Y, Sahara T, Yoshikawa H (2008) Change in knee cartilage T2 in response to mechanical loading. J Magn Reson Imaging 28:175–180
Souza RB, Kumar D, Calixto N, Singh J, Schooler J, Subburaj K, Li X, Link TM, Majumdar S (2014) Response of knee cartilage T1rho and T2 relaxation times to invivo mechanical loading in individuals with and without knee osteoarthritis. Osteoarthr Cartil 22:1367–1376
Mamisch TC, Trattnig S, Quirbach S, Marlovits S, White LM, Welsch GH (2010) Quantitative T2 mapping of knee cartilage: differentiation of healthy control cartilage and cartilage repair tissue in the knee with unloading—initial results. Radiology 254:818–826
Hannila I, Räinä SS, Tervonen O, Ojala R, Nieminen MT (2009) Topographical variation of T2 relaxation time in the young adult knee cartilage at 1.5 T. Osteoarthr Cartil 17:1570–1575
Brem MH, Lang PK, Neumann G, Schlechtweg PM, Schneider E, Jackson R, Yu J, Eaton CB, Hennig FF, Yoshioka H, Pappas G, Duryea J (2009) Magnetic resonance image segmentation using semi-automated software for quantification of knee articular cartilage—initial evaluation of a technique for paired scans. Skelet Radiol 38:505–511
Mosher TJ, Zhang Z, Reddy R, Boudhar S, Milestone BN, Morrison WB, Kwoh CK, Eckstein F, Witschey WR, Borthakur A (2011) Knee articular cartilage damage in osteoarthritis: analysis of MR image biomarker reproducibility in ACRIN-PA 4001 multicenter trial. Radiology 258:832–842
Multanen J, Rauvala E, Lammentausta E, Ojala R, Kiviranta I, Häkkinen A, Nieminen MT, Heinonen A (2009) Reproducibility of imaging human knee cartilage by delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) at 1.5 Tesla. Osteoarthr Cartil 17:559–564
Xia Y (2000) Magic-angle effect in magnetic resonance imaging of articular cartilage: a review. Investig Radiol 35:602–621
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
All volunteer studies in this work were approved by local ethics committee. The manuscript does not report clinical studies or contain patient data.
Rights and permissions
About this article
Cite this article
Hannila, I., Lammentausta, E., Tervonen, O. et al. The repeatability of T2 relaxation time measurement of human knee articular cartilage. Magn Reson Mater Phy 28, 547–553 (2015). https://doi.org/10.1007/s10334-015-0494-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-015-0494-3