Abstract
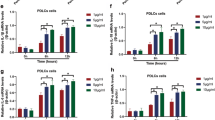
Diabetes mellitus (DM), characterized by production and accumulation of advanced glycation end products (AGEs), induces and promotes chronic inflammation in tissues, including periodontal tissue. Increasing amount of epidemiological and experimental evidence demonstrated that more extensive inflammatory reaction and bone resorption occurred in periodontal tissues in diabetic patients with periodontitis, which is speculated to be related to NLRP3 inflammasome. NLRP10 is the only NOD-like receptor protein lacking leucine-rich repeats, suggesting that NLRP10 may be a regulatory protein. The aim of this study was to investigate the regulatory role of NLRP10 on NLRP1 and NLRP3 inflammasome in human periodontal ligament cells (HPDLCs) under AGEs treatment. Expression of NLRP10 in HPDLCs stimulated with 100 ug/mL AGEs for 24 h was observed. Detection of TRIM31 is conducted, and in TRIM31-overexpressed HPDLCs, the interaction between NLRP10 with TRIM31 as well as NLRP10 with ubiquitination were explored by immunoprecipitation. Under AGEs stimulation, the activation of reactive oxidative stress (ROS) and inflammatory signaling pathway (NF-κB, MAPK pathway) was detected by biomedical microscope and western blot (WB), respectively. After stimulation with AGEs for 24 h with or without silencing NLRP10, inflammatory cytokines (IL-6 and IL-1β), NF-κB, MAPK pathway, ROS, and components of inflammasome were assessed. In HPDLCs, we found AGEs induced NLRP10 and inhibited TRIM31. TRIM31 overexpression significantly enhanced interaction between TRIM31 and NLRP10, then induced proteasomal degradation of NLRP10. Moreover, under AGEs stimulation, NLRP10 positively regulates NLRP1, NLRP3 inflammasomes by activating NF-κB, MAPK pathway, and increasing ROS, finally promoting the expression of inflammatory cytokines. Together, we, for the first time, confirmed that NLRP10 could promote inflammatory response induced by AGEs in HPDLCs via activation of NF-κB, and MAPK pathway and increasing ROS.





Similar content being viewed by others
Data Availability
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.
References
Slots J. Periodontitis: Facts, fallacies and the future. Periodontol 2000. 2017;75(1):7–23. https://doi.org/10.1111/prd.12221.
Kwon T, Lamster IB, Levin L. Current concepts in the management of periodontitis. Int Dent J. 2021;71(6):462–76. https://doi.org/10.1111/idj.12630.
Bianchi S, Mancini L, Torge D, Cristiano L, Mattei A, Varvara G, et al. Bio-Morphological reaction of human periodontal ligament fibroblasts to different types of dentinal derivates: In vitro study. Int J Mol Sci. 2021;22(16):8681. https://doi.org/10.3390/ijms22168681.
Li Y, Ling J, Jiang Q. Inflammasomes in alveolar bone loss. Front Immunol. 2021;12:691013. https://doi.org/10.3389/fimmu.2021.691013.
Beertsen W, Mcculloch C, Sodek J. The periodontal ligament: a unique, multifunctional connective tissue. Periodontol. 2000;1997(13):20–40. https://doi.org/10.1111/j.1600-0757.1997.tb00094.x.
Kocher T, König J, Borgnakke WS, Pink C, Meisel P. Periodontal complications of hyperglycemia/diabetes mellitus: epidemiologic complexity and clinical challenge. Periodontol 2000. 2018;78(1):59–97. https://doi.org/10.1111/prd.12235.
Polak D, Shapira L. An update on the evidence for pathogenic mechanisms that may link periodontitis and diabetes. J Clin Periodontol. 2018;45(2):150–66. https://doi.org/10.1111/jcpe.12803.
Shin JJ, Lee EK, Park TJ, Kim W. Damage-associated molecular patterns and their pathological relevance in diabetes mellitus. Ageing Res Rev. 2015;24(Pt A):66–76. https://doi.org/10.1016/j.arr.2015.06.004.
Zindel J, Kubes P. DAMPs, PAMPs, and LAMPs in immunity and sterile inflammation. Annu Rev Pathol. 2020;15:493–518. https://doi.org/10.1146/annurev-pathmechdis-012419-032847.
Singh R, Barden A, Mori T, Beilin L. Advanced glycation end-products: a review. Diabetologia. 2001;44:129–46. https://doi.org/10.1007/s001250051591.
Sruthi CR, Raghu KG. Advanced glycation end products and their adverse effects: the role of autophagy. J Biochem Mol Toxic. 2021;35:e22710. https://doi.org/10.1002/jbt.22710.
Yi X, Zhang L, Lu W, Tan X, Yue J, Wang P, et al. The effect of NLRP inflammasome on the regulation of AGEs-induced inflammatory response in human periodontal ligament cells. J Periodontal Res. 2019;54(6):681–9. https://doi.org/10.1111/jre.12677.
Mathews RJ, Sprakes MB, McDermott MF. NOD-like receptors and inflammation. Arthritis Res Ther. 2008;10(6):228. https://doi.org/10.1186/ar2525.
Rathinam VAK, Fitzgerald KA. Inflammasome complexes: emerging mechanisms and effector functions. Cell. 2016;165(4):792–800. https://doi.org/10.1016/j.cell.2016.03.046.
Ising C, Venegas C, Zhang S, Scheiblich H, Schmidt SV, Vieira-Saecker A, et al. NLRP3 inflammasome activation drives tau pathology. Nature. 2019;575(7784):669–73. https://doi.org/10.1038/s41586-019-1769-z.
Wang D, Zhang Y, Xu X, Wu J, Peng Y, Li J, et al. YAP promotes the activation of NLRP3 inflammasome via blocking K27-linked polyubiquitination of NLRP3. Nat Commun. 2021;12(1):2674. https://doi.org/10.1038/s41467-021-22987-3.
Li H, Zhong X, Chen Z, Li W. Suppression of NLRP3 inflammasome improves alveolar bone defect healing in diabetic rats. J Orthop Surg Res. 2019;14(1):167. https://doi.org/10.1186/s13018-019-1215-9.
Guan X, Guan Y, Shi C, Zhu X, He Y, Wei Z, et al. Estrogen deficiency aggravates apical periodontitis by regulating NLRP3/caspase-1/IL-1β axis. Am J Transl Res. 2020;12(2):660–71.
Kaur G, Bagam P, Pinkston R, Singh DP, Batra S. Cigarette smoke-induced inflammation: NLRP10-mediated mechanisms. Toxicology. 2018;398–399:52–67. https://doi.org/10.1016/j.tox.2018.02.010.
Damm A, Lautz K, Kufer TA. Roles of NLRP10 in innate and adaptive immunity. Microbes Infect. 2013;15(6–7):516–23. https://doi.org/10.1016/j.micinf.2013.03.008.
Murphy N, Grehan B, Lynch MA. Glial uptake of amyloid beta induces NLRP3 inflammasome formation via cathepsin-dependent degradation of NLRP10. Neuromol Med. 2014;16(1):205–15. https://doi.org/10.1007/s12017-013-8274-6.
Lautz K, Damm A, Menning M, Wenger J, Adam AC, Zigrino P, et al. NLRP10 enhances Shigella-induced pro-inflammatory responses. Cell Microbiol. 2012;14(10):1568–83. https://doi.org/10.1111/j.1462-5822.2012.01822.x.
Wang L, Li X, Song Y, Zhang L, Ye L, Zhou X, et al. NELL1 augments osteogenesis and inhibits inflammation of human periodontal ligament stem cells induced by BMP9. J Periodontol. 2022;93(7):977–87. https://doi.org/10.1002/JPER.20-0517.
Hiraiwa H, Sakai T, Mitsuyama H, Hamada T, Yamamoto R, Omachi T, et al. Inflammatory effect of advanced glycation end products on human meniscal cells from osteoarthritic knees. Inflamm Res. 2011;60(11):1039–48. https://doi.org/10.1007/s00011-011-0365-y.
Cheng A, Dong Y, Zhu F, Liu Y, Hou FF, Nie J. AGE-LDL activates toll like receptor 4 pathway and promotes inflammatory cytokines production in renal tubular epithelial cells. Int J Biol Sci. 2013;9(1):94–107. https://doi.org/10.7150/ijbs.5246.
Li DX, Deng TZ, Lv J, Ke J. Advanced glycation end products (AGEs) and their receptor (RAGE) induce apoptosis of periodontal ligament fibroblasts. Braz J Med Biol Res. 2014;47(12):1036–43. https://doi.org/10.1590/1414-431X20143996.
Xu W, Lu Y, Yue J, Lu W, Zhou W, Zhou X, et al. Occlusal trauma inhibits osteoblast differentiation and bone formation through IKK-NF-κB signaling. J Periodontol. 2020;91(5):683–92. https://doi.org/10.1002/JPER.18-0710.
Chaudhuri J, Bains Y, Guha S, Kahn A, Hall D, Bose N, et al. The role of advanced glycation end products in aging and metabolic diseases: bridging association and causality. Cell Metab. 2018;28(3):337–52. https://doi.org/10.1016/j.cmet.2018.08.014.
Dudley DT, Pang L, Decker SJ, Bridges AJ, Saltiel AR. A synthetic inhibitor of the mitogen-activated protein-kinase cascade. P Natl Acad Sci USA. 1995;92(17):7686–9. https://doi.org/10.1073/pnas.92.17.7686.
Zhou Q, Ren X, Bischoff D, Weisgerber DW, Yamaguchi DT, Miller TA, et al. Nonmineralized and mineralized collagen scaffolds induce differential osteogenic signaling pathways in human mesenchymal stem cells. Adv Healthc Mater. 2017;6(23):1700641. https://doi.org/10.1002/adhm.201700641.
Valdivieso ÁG, Dugour AV, Sotomayor V, Clauzure M, Figueroa JM, Santa-Coloma TA. N-acetyl cysteine reverts the proinflammatory state induced by cigarette smoke extract in lung Calu-3 cells. Redox Biol. 2018;16:294–302. https://doi.org/10.1016/j.redox.2018.03.006.
Spagnuolo G, D’Antò V, Cosentino C, Schmalz G, Schweikl H, Rengo S. Effect of N-acetyl-l-cysteine on ROS production and cell death caused by HEMA in human primary gingival fibroblasts. Biomaterials. 2006;27(9):1803–9. https://doi.org/10.1016/j.biomaterials.2005.10.022.
Swanson KV, Deng M, Ting JP. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol. 2019;19:477–89. https://doi.org/10.1038/s41577-019-0165-0.
Song H, Liu B, Huai W, Yu Z, Wang W, Zhao J, et al. The E3 ubiquitin ligase TRIM31 attenuates NLRP3 inflammasome activation by promoting proteasomal degradation of NLRP3. Nat Commun. 2016. https://doi.org/10.1038/ncomms13727.
Esposito D, Koliopoulos MG, Rittinger K. Structural determinants of TRIM protein function. Biochem Soc T. 2017;45(1):183–91. https://doi.org/10.1042/BST20160325.
Graves DT, Ding Z, Yang Y. The impact of diabetes on periodontal diseases. Periodontol 2000. 2019;82(1):214–24. https://doi.org/10.1111/prd.12318.
Akram Z, Alqahtani F, Alqahtani M, Al-Kheraif AA, Javed F. Levels of advanced glycation end products in gingival crevicular fluid of chronic periodontitis patients with and without type-2 diabetes mellitus. J Periodontol. 2019;91(3):396–402. https://doi.org/10.1002/JPER.19-0209.
Zizzi A, Tirabassi G, Aspriello SD, Piemontese M, Rubini C, Lucarini G. Gingival advanced glycation end-products in diabetes mellitus-associated chronic periodontitis: an immunohistochemical study. J Periodontal Res. 2013;48(3):293–301. https://doi.org/10.1111/jre.12007.
Chen F, Jin Y. Periodontal tissue engineering and regeneration: current approaches and expanding opportunities. Tissue Eng Part B Rev. 2010;16(2):219–55. https://doi.org/10.1089/ten.teb.2009.0562.
Lu WL, Zhang L, Song DZ, Yi XW, Xu WZ, Ye L, et al. NLRP6 suppresses the inflammatory response of human periodontal ligament cells by inhibiting NF-κB and ERK signal pathways. Int Endod J. 2019;52(7):999–1009. https://doi.org/10.1111/iej.13091.
Zhang S, Liu Y, Wang X, An N, Ouyang X. STAT1/SOCS1/3 are involved in the Inflammation-Regulating effect of GAS6/AXL in periodontal ligament cells induced by porphyromonas gingivalis lipopolysaccharide in vitro. J Immunol Res. 2021;2021:9577695. https://doi.org/10.1155/2021/9577695.
Zheng J, Chen S, Albiero ML, Vieira GHA, Wang J, Feng JQ, et al. Diabetes activates periodontal ligament fibroblasts via NF-κB in vivo. J Dent Res. 2018;97(5):580–8. https://doi.org/10.1177/0022034518755697.
Motta V, Soares F, Sun T, Philpott DJ. NOD-like receptors: versatile cytosolic sentinels. Physiol Rev. 2015;95(1):149–78. https://doi.org/10.1152/physrev.00009.2014.
Leemans JC, Kors L, Anders H, Florquin S. Pattern recognition receptors and the inflammasome in kidney disease. Nat Rev Nephrol. 2014;10(7):398–414. https://doi.org/10.1038/nrneph.2014.91.
Lim RR, Wieser ME, Ganga RR, Barathi VA, Lakshminarayanan R, Mohan RR, et al. NOD-like receptors in the eye: uncovering its role in diabetic retinopathy. Int J Mol Sci. 2020;21(3):899. https://doi.org/10.3390/ijms21030899.
Eisenbarth SC, Williams A, Colegio OR, Meng H, Strowig T, Rongvaux A, et al. NLRP10 is a NOD-like receptor essential to initiate adaptive immunity by dendritic cells. Nature. 2012;484(7395):510–3. https://doi.org/10.1038/nature11012.
Kong X, Yuan Z, Cheng J. The function of NOD-like receptors in central nervous system diseases. J Neurosci Res. 2017;95(8):1565–73. https://doi.org/10.1002/jnr.24004.
Jie YZLT. Post-translational regulation of inflammasomes. Cell Mol Immunol. 2017;14(1):65–79. https://doi.org/10.1038/cmi.2016.29.
Juliana C, Fernandes-Alnemri T, Kang S, Farias A, Qin F, Alnemri ES. Non-transcriptional priming and deubiquitination regulate NLRP3 inflammasome activation. J Biol Chem. 2012;287(43):36617–22. https://doi.org/10.1074/jbc.M112.407130.
Wu X, Lu M, Ding S, Zhong Q. Tripartite motif 31 alleviates IL-1ß secretion via promoting the ubiquitination of pyrin domain domains-containing protein 3 in human periodontal ligament fibroblasts. Odontology. 2020;108(3):424–32. https://doi.org/10.1007/s10266-020-00519-7.
Wang Y, Hasegawa M, Imamura R, Kinoshita T, Kondo C, Konaka K, et al. PYNOD, a novel Apaf-1/CED4-like protein is an inhibitor of ASC and caspase-1. Int Immunol. 2004;16(6):777–86. https://doi.org/10.1093/intimm/dxh081.
Li Z, Shui S, Han X, Yan L. NLRP10 ablation protects against ischemia/reperfusion-associated brain injury by suppression of neuroinflammation. Exp Cell Res. 2020;389(2):111912. https://doi.org/10.1016/j.yexcr.2020.111912.
Xu W, Lu Q, Qu M, Fan R, Leng S, Wang L, et al. Wnt4 regulates bone metabolism through IKK-NF-κB and ROCK signaling under occlusal traumatic periodontitis. J Periodontal Res. 2022;57(3):461–9. https://doi.org/10.1111/jre.12975.
Mirza N, Sowa AS, Lautz K, Kufer TA. NLRP10 affects the stability of abin-1 to control inflammatory responses. J Immunol. 2019;202(1):218–27. https://doi.org/10.4049/jimmunol.1800334.
Lee S, Choi B. Involvement of NLRP10 in IL-1α induction of oral epithelial cells by periodontal pathogens. Innate Immun-London. 2017;23(7):569–77. https://doi.org/10.1177/1753425917722610.
Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature. 2000;404(6779):787–90. https://doi.org/10.1038/35008121.
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
This work was supported by National Natural Science Foundation of China. (No. 82170946[L.Z] and No. 81970936 [D.M.H]).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors deny any conflicts of interest related to this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yi, X., Song, Y., Xu, J. et al. NLRP10 promotes AGEs-induced NLRP1 and NLRP3 inflammasome activation via ROS/MAPK/NF-κB signaling in human periodontal ligament cells. Odontology 112, 100–111 (2024). https://doi.org/10.1007/s10266-023-00813-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-023-00813-0