Abstract
Background
Thyroid carcinoma is a very rare tumor in the pediatric age group, accounting for only 1.5–3% of childhood carcinomas in the United States and Europe. We aimed to identify the risk of a second malignancy among pediatric thyroid cancer survivors.
Methods
The cohort analysis consisted of pediatric cancer patients aged less than 20 years, diagnosed with a primary thyroid cancer, identified by site code ICD-0-3: C739, and reported to the SEER 9 database between 1973 and 2013. They were followed up until December 31, 2013; the end of the study period, or up to death if earlier.
Results
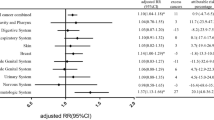
Out of 1769 patients diagnosed primarily with thyroid carcinoma, 42 patients had a total of 45 incidences of subsequent malignancies. The mean age of patients at the initial diagnosis of thyroid cancer was 16 years. Females (90.5%) had a significantly higher incidence of second malignancies (SM) than males (9.5%). The overall Standardized Incidence Ratio (SIR) of SM in the study patients was higher than expected (SIR = 1.48). Some specific sites showed significantly higher incidences: the salivary glands (SIR = 33.95), the gum and other parts of the mouth [excluding the lips, tongue, salivary glands and floor of the mouth] (SIR = 24.53)*** and the kidneys (SIR = 5.72). The overall risk of SM in patients who had received radioactive iodine was higher than expected (SIR = 4.41). The cumulative incidence of SM after treatment of thyroid cancer in children increases steadily over 40 years (11.92%).
Conclusions
Race, gender, histological subtypes, and radioactive iodine are potentially significant prognostic factors for the development of SM among pediatric thyroid cancer survivors. Identification of underlying mechanisms that raise the risk of SM is important for both treatment and follow-up strategies.


Similar content being viewed by others
References
Greenlee RT, Hill-Harmon MB, Murray T et al (2001) Cancer statistics. CA Cancer J Clin 51:15–36
Klein Hesselink MS, Nies M, Bocca G et al (2016) Pediatric differentiated thyroid carcinoma in The Netherlands: a nationwide follow-up study. J Clin Endocrinol Metab 101:2031–2039. https://doi.org/10.1210/jc.2015-3290
Vaisman F, Corbo R, Vaisman M (2011) Thyroid carcinoma in children and adolescents—systematic review of the literature. J Thyroid Res 2011:1–7. https://doi.org/10.4061/2011/845362
Francis GL, Waguespack SG, Bauer AJ et al (2015) Management guidelines for children with thyroid nodules and differentiated thyroid cancer. Thyroid 25:716–759. https://doi.org/10.1089/thy.2014.0460
Rapkin L, Pashankar FD (2012) Management of thyroid carcinoma in children and young adults. J Pediatr Hematol Oncol 34:S39–S46. https://doi.org/10.1097/MPH.0b013e31824e37a6
Cho YY, Lim J, Oh C-M et al (2015) Elevated risks of subsequent primary malignancies in patients with thyroid cancer: a nationwide, population-based study in Korea. Cancer 121:259–268. https://doi.org/10.1002/cncr.29025
Leslie Bernstein JGG (1999) Carcinomas and Other Malignant Epithelial Neoplasms. In: Ries LAG, Smith MA, Gurney JG, Linet M, Tamra T, Young JL BG (eds) (ed) Cancer Incid. Surviv. among Child. Adolesc. United States SEER Progr. 1975-1995, Natl. Cancer Institute, SEER Program.NIH Pub. No. 99-4649. Bethesda, MD, p. 139–148
Delbert A, Fisher AG (2008) Thyroid disorders in childhood and adolescence. In: Sperling MA (ed) Pediatr. Endocrinol., 3rd ed. pp 227–253
Stanley JA, Aruldhas MM, Chandrasekaran M et al (2012) Androgen receptor expression in human thyroid cancer tissues: a potential mechanism underlying the gender bias in the incidence of thyroid cancers. J Steroid Biochem Mol Biol 130:105–124. https://doi.org/10.1016/j.jsbmb.2012.02.004
Rahbari R, Zhang L, Kebebew E (2010) Thyroid cancer gender disparity. Future Oncol 6:1771–1779. https://doi.org/10.2217/fon.10.127
Edgren G, Liang L, Adami H-O et al (2012) Enigmatic sex disparities in cancer incidence. Eur J Epidemiol 27:187–196. https://doi.org/10.1007/s10654-011-9647-5
Al-Qahtani K, Tunio M, Al Asiri M et al (2016) Tall cell variant papillary thyroid carcinoma in Saudi patients. A clinicopathological and outcomes analysis. Saudi Med J 37:1220–1224. https://doi.org/10.15537/smj.2016.11.15480
Berthe E, Henry-Amar M, Michels J-J et al (2004) Risk of second primary cancer following differentiated thyroid cancer. Eur J Nucl Med Mol Imaging 31:685–691. https://doi.org/10.1007/s00259-003-1448-y
Hakala TT, Sand JA, Jukkola A et al (2016) Increased risk of certain second primary malignancies in patients treated for well-differentiated thyroid cancer. Int J Clin Oncol 21:231–239. https://doi.org/10.1007/s10147-015-0904-6
Hall P, Holm LE, Lundell G (1990) Second primary tumors following thyroid cancer. A Swedish record-linkage study. Acta Oncol 29:869–873
Liou M-J, Tsang N-M, Hsueh C et al (2016) Therapeutic outcome of second primary malignancies in patients with well-differentiated thyroid cancer. Int J Endocrinol 2016:1–7. https://doi.org/10.1155/2016/9570171
Lu C-H, Lee K-D, Chen P-T et al (2013) Second primary malignancies following thyroid cancer: a population-based study in Taiwan. Eur J Endocrinol 169:577–585. https://doi.org/10.1530/EJE-13-0309
Verkooijen RBT, Smit JWA, Romijn JA et al (2006) The incidence of second primary tumors in thyroid cancer patients is increased, but not related to treatment of thyroid cancer. Eur J Endocrinol 155:801–806. https://doi.org/10.1530/eje.1.02300
Brown AP, Chen J, Hitchcock YJ et al (2008) The risk of second primary malignancies up to three decades after the treatment of differentiated thyroid cancer. J Clin Endocrinol Metab 93:504–515. https://doi.org/10.1210/jc.2007-1154
Canchola AJ, Horn-Ross PL, Purdie DM (2006) Risk of second primary malignancies in women with papillary thyroid cancer. Am J Epidemiol 163:521–527. https://doi.org/10.1093/aje/kwj072
Kim C, Bi X, Pan D et al (2013) The risk of second cancers after diagnosis of primary thyroid cancer is elevated in thyroid microcarcinomas. Thyroid 23:575–582. https://doi.org/10.1089/thy.2011.0406
Neglia JP, Friedman DL, Yasui Y et al (2001) Second malignant neoplasms in five-year survivors of childhood cancer: childhood cancer survivor study. J Natl Cancer Inst 93:618–629
Meadows AT, Friedman DL, Neglia JP et al (2009) Second neoplasms in survivors of childhood cancer: findings from the childhood cancer survivor study cohort. J Clin Oncol 27:2356–2362. https://doi.org/10.1200/JCO.2008.21.1920
Meadows AT (1989) Second malignant neoplasms in childhood cancer survivors. J Assoc Pediatr Oncol Nurses 6:7–11
Uprety D, Khanal A, Arjyal L et al (2016) The risk of secondary primary malignancy in early stage differentiated thyroid cancer: a US population-based study. Acta Oncol (Madr) 55:1375–1377. https://doi.org/10.1080/0284186X.2016.1196829
Teng C-J, Hu Y-W, Chen S-C et al (2016) Use of radioactive iodine for thyroid cancer and risk of second primary malignancy: a nationwide population-based study. J Natl Cancer Inst 108:djv314. https://doi.org/10.1093/jnci/djv314
Lang BH-H, Wong IOL, Wong KP et al (2012) Risk of second primary malignancy in differentiated thyroid carcinoma treated with radioactive iodine therapy. Surgery 151:844–850. https://doi.org/10.1016/j.surg.2011.12.019
Iyer NG, Morris LGT, Tuttle RM et al (2011) Rising incidence of second cancers in patients with low-risk (T1N0) thyroid cancer who receive radioactive iodine therapy. Cancer 117:4439–4446. https://doi.org/10.1002/cncr.26070
Fallahi B, Adabi K, Majidi M et al (2011) Incidence of second primary malignancies during a long-term surveillance of patients with differentiated thyroid carcinoma in relation to radioiodine treatment. Clin Nucl Med 36:277–282. https://doi.org/10.1097/RLU.0b013e31820a9fe3
Marti JL, Jain KS, Morris LGT (2015) Increased risk of second primary malignancy in pediatric and young adult patients treated with radioactive iodine for differentiated thyroid cancer. Thyroid 25:681–687. https://doi.org/10.1089/thy.2015.0067
Lee SL (2010) Complications of radioactive iodine treatment of thyroid carcinoma. J Natl Compr Canc Netw 8:1277–1286
Klubo-Gwiezdzinska J, Van Nostrand D, Burman KD et al (2010) Salivary gland malignancy and radioiodine therapy for thyroid cancer. Thyroid 20:647–651. https://doi.org/10.1089/thy.2009.0466
Pappo AS, Furman WL, Schultz KA et al (2015) Rare tumors in children: progress through collaboration. J Clin Oncol 33:3047–3054. https://doi.org/10.1200/JCO.2014.59.3632
Yu JB, Gross CP, Wilson LD et al (2009) NCI SEER public-use data: applications and limitations in oncology research. Oncology (Williston Park) 23:288–295
Acknowledgements
We would like to express our thanks to Denise S. Schwartz, DVM, MS, Ph.D. (School of Veterinary Medicine and Animal Science, the University of Sao Paulo) for English editing and to Ali S. Hadi (Department of Mathematics and Actuarial Science, American University in Cairo) for help with fitting the Cox Proportional Hazards Models. Also, we would like to thank Mohamed-Ismail Rakha for his efforts in revising the manuscript.
Funding
ASA is supported by Grant 57147166 from The German Academic Exchange Service (DAAD).
Author information
Authors and Affiliations
Contributions
WMR designed the concept of the paper. WMR and MHA conducted the analysis of the data and have access to them. WMR, MHA, MS, MAR, MI, AE, MAA, WZ and ASA contributed to data interpretation and writing of the paper. All authors revised and agreed to the content of the paper.
Corresponding author
Ethics declarations
Conflict of interest
No financial interests are to be disclosed by authors.
About this article
Cite this article
Adly, M.H., Sobhy, M., Rezk, M.A. et al. Risk of second malignancies among survivors of pediatric thyroid cancer. Int J Clin Oncol 23, 625–633 (2018). https://doi.org/10.1007/s10147-018-1256-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-018-1256-9