Abstract
Background
The aim of this multicentre study was to analyse the outcomes of biosynthetic absorbable poly-4-hydroxybutyrate (P4HB) prosthesis implantation in patients undergoing ventral hernia repair (VHR) in the context of different degrees of contamination.
Methods
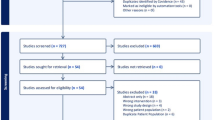
From May 2016 to December 2021, a multicentre retrospective analysis of patients who underwent elective or urgent hernia repair with P4HB prosthesis was performed in seven hospitals in Spain and Portugal. Patients with a postoperative follow-up of less than 20 months and those within the theoretical period of prosthesis resorption were excluded from the study. Regarding the degree of contamination, patients were assessed according to the modified Ventral Hernia Working Group (VHWG) classification. Epidemiological data, hernia characteristics, surgical and postoperative variables (Clavien–Dindo classification) of these patients were analyzed. Risk factors related to long-term recurrence were studied by a multivariate analysis.
Results
In 236 cases of P4HB prosthesis implantation, repair in cases of Grade 3 was the most frequent (49.1%), followed by Grade 2 in 42.3% of cases and Grade 1 in 8.4%. The most frequent complications were Grade 1, with the majority occurring during the first year. The overall rate of surgical site occurrences (SSO) was 30%. The hernia recurrence rate was 14.4% (n = 34), with a mean postoperative follow-up time of 41 months (22–61). The multivariate analysis showed that the onlay location of the mesh (OR 1.07; CI 1.42–2.70, p = 0.004) was a significant independent risk factor for recurrence.
Conclusions
The use of a P4HB bioresorbable mesh for the VHR with different degrees of contamination leads to favourable results overall, with an acceptable rate of hernia recurrence. The onlay location of the P4HB prosthesis is the main factor in recurrence in both elective and emergency settings.
Similar content being viewed by others
Data availability
Not applicable.
References
Miserez M, Jairam AP, Boersema GSA et al (2019) Resorbable synthetic meshes for abdominal wall defects in preclinical setting: a literature review. J Surg Res 237:67–75
Martin DP, Badhwar A, Shah DV et al (2013) Characterization of poly-4-hydroxybutyrate mesh for hernia repair applications. J Surg Res 184:766–773
Buell JF, Sigmon D, Ducoin C et al (2017) Initial experience with biologic polymer scaffold (poly-4-hydroxybuturate) in complex abdominal wall reconstruction. Ann Surg 266:185–188
Scott JR, Deeken CR, Martindale RG et al (2016) Evaluation of a fully absorbable poly-4-hydroxybutyrate/absorbable barrier composite mesh in a porcine model of ventral hernia repair. Surg Endosc 30:3691–3701
Lake SP, Stoikes NFN, Badhwar A et al (2019) Contamination of hybrid hernia meshes compared to bioresorbable Phasix™ Mesh in a rabbit subcutaneous implant inoculation model. Ann Med Surg 46:12–16
Williams SF, Rizk S, Martin DP (2013) Poly-4-hydroxybutyrate (P4HB): a new generation of resorbable medical devices for tissue repair and regeneration. Biomed Tech 58:439–452
Kanters AE, Krpata DM, Blatnik JA et al (2012) Modified hernia grading scale to stratify surgical site occurrence after open ventral hernia repairs. J Am Coll Surg 215(6):787–879
Baldan N, Munegato G, Di Leo A et al (2020) Lessons learned from 227 biological meshes used for the surgical treatment of ventral abdominal defects. Hernia 24(1):57–65
Van Rooijen MMJ, Jairam A, Tollens T et al (2020) Outcomes of a new slowly resorbable biosynthetic mesh (Phasix™) in potentially contaminated incisional hernias: a prospective, multi-center, single-arm trial. Int J Surg 83:31–36
Charleux-Muller D, Hurel R, Fabacher T et al (2021) Slowly absorbable mesh in contaminated incisional hernia repair: results of a French multicenter study. Hernia 25:1051–1059
Deeken CR, Chen DC, Lopez-Cano M et al (2023) Fully resorbable poly-4-hydroxybutyrate (P4HB) mesh for soft tissue repair and reconstruction: a scoping review. Front Surg. https://doi.org/10.3389/fsurg.2023.1157661
Muysoms FE, Miserez M, Berrevoet F et al (2009) Classification of primary and incisional abdominal wall hernias. Hernia 13:407–414
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications. A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Atema JJ, de Vries FE, Boermeester MA (2016) Systematic review and meta-analysis of the repair of potentially contaminated and contaminated abdominal wall defects. Am J Surg 212:982–995
Claessen JJM, Timmer AS, Atema JJ et al (2021) Outcomes of mid-term and long-term degradable biosynthetic meshes in single-stage open complex abdominal wall reconstruction. Hernia 25:1647–1657
Rognoni C, Cuccurullo D, Borsoi L et al (2020) Clinical outcomes and quality of life associated with the use of a biosynthetic mesh for complex ventral hernia repair: analysis of the “Italian Hernia Club” registry. Sci Rep 10:10706
Mellia JA, Othman S, Naga HI et al (2020) Outcomes of Poly-4-Hydroxybutyrate mesh in ventral hernia repair: a systematic review and pooled analysis. Plast Reconstr Surg Glob Open 8(12):e3158
Plymale MA, Davenport DL, Dugan A et al (2018) Ventral hernia repair with poly-4-hydroxybutyrate mesh. Surg Endosc 32:1689–1694
Roth JS, Anthone GJ, Selzer DJ et al (2018) Prospective evaluation of poly-4-hydroxybutyrate mesh in CDC class I/high-risk ventral and incisional hernia repair: 18-month follow-up. Surg Endosc 32:1929–1936
Layer T, Benammi S, Dubuisson V et al (2023) Incisional hernia repair with a slowly absorbable P4HB mesh: what happens after the mesh disappears? A retrospective longitudinal clinical study. Hernia 27:387–394
Bueno-Lledo J, Ceno M, Perez-Alonso C et al (2021) Biosynthetic resorbable prosthesis is useful in single-stage management of chronic mesh infection after abdominal wall hernia repair. World J Surg 45:443–450
Talwar AA, Perry NJ, McAuliffe PB et al (2022) Shifting the goalpost in ventral hernia care: 5-year outcomes after ventral hernia repair with poly-4-hydroxybutyrate mesh. Hernia 26:1635–1643. https://doi.org/10.1007/s10029-022-02674-y
Holihan JL, Hannon C, Goodenough C et al (2017) Ventral hernia repair: a meta-analysis of randomized controlled trials. Surg Infect (Larchmt) 18(6):647–658. https://doi.org/10.1089/sur.2017.029
Plymale MA, Davenport DL, Walsh-Blackmore S et al (2020) Costs and complications associated with infected mesh for ventral hernia repair. Surg Infect (Larchmt) 21(4):344–349
Levy AS, Bernstein JL, Premaratne ID et al (2020) Poly-4-hydroxybutyrate (Phasix™) mesh onlay in complex abdominal wall repair. Surg Endosc 35(5):2049–2058. https://doi.org/10.1007/s00464-020-07601-9
Itani KM, Rosen M, Vargo D, et al. RICH Study Group. Prospective study of single-stage repair of contaminated hernias using a biologic porcine tissue matrix: the RICH Study. Surgery 2012;152(3):498–505. https://doi.org/10.1016/j.surg.2012.04.008
Bueno-Lledó J, Ceno M, Pérez-Alonso C et al (2020) Abdominal wall reconstruction with biosynthetic absorbable mesh after infected prosthesis explantation: single stage is better than two-stage approach of chronic mesh infection. Hernia 25(4):1005–1012
Köckerling F, Alam NN, Antoniou SA et al (2018) What is the evidence for the use of biologic or biosynthetic meshes in abdominal wall reconstruction? Hernia 22:249–269
Warren J, Desai SS, Boswell ND et al (2020) Safety and efficacy of synthetic mesh for ventral hernia repair in a contaminated field. J Am Coll Surg 230:405–413
Birolini C, de Miranda JS, Tanaka EY et al (2020) The use of synthetic mesh in contaminated and infected abdominal wall repairs: challenging the dogma. A long-term prospective clinical trial. Hernia 24:307–323
Pakula A, Skinner R (2020) Outcomes of open complex ventral hernia repairs with retromuscular placement of poly-4-hydroxybutyrate bioabsorbable mesh. Surg Innov 27:32–37
Schecter SC, Imhof L, Lasker MV et al (2022) Single-stage abdominal wall reconstruction in contaminated and dirty wounds is safe: a single center experience. Surg Endosc 36:5766–5771
Acknowledgements
We would like to thank to Santiago Bonafé, Providencia Garcia-Pastor, Victoria Pareja and Omar Carreño of our surgical group for their helpful discussions.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
All authors have participated to this recollection of data.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Bueno-Lledó has been speaker for BD surgical and had grants with BD surgical (workshops). These disclosures do not impact the topics reported herein. All other authors declare no disclosures relevant to this article or research, and have no conflicts of interest or financial ties to disclose. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Informed consent
Authors declare that a written informed consent was obtained from all patients.
Human and animal rights
All participants gave their permission to participate to the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bueno-Lledó, J., Porrero-Guerrero, B., Ferreira, F. et al. Long-term results with biosynthetic absorbable P4HB mesh in ventral abdominal wall repair: a multicentre analysis. Hernia (2024). https://doi.org/10.1007/s10029-024-02981-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10029-024-02981-6