Abstract
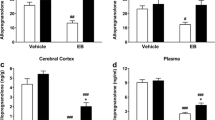
Previously, we found out that in ovariectomized female rats, estrogen and progesterone produce a memory deficit which is reverted by the intrahippocampal administration of allopregnanolone. Here, we study the possible interplay between allopregnanolone and hippocampal serotonergic activity. Ovariectomized rats injected subcutaneously with estrogen and progesterone were subsequently injected in the dorsal hippocampus with vehicle, allopregnanolone alone or allopregnanolone shortly after 8OH-DPAT, a predominantly 5HT1A-7 receptor agonist. Then, the subjects were sequentially tested in: (1) an inhibitory avoidance task and (2) K+-evoked [3H]-serotonin ex vivo release through superfusion experiments. Allopregnanolone increased the K+-evoked [3H]-serotonin release compared to control. 8OH-DPAT infusions reversed the effects of allopregnanolone on memory and K+-evoked [3H]-serotonin release. These results suggest that allopregnanolone memory improvement could be mediated, at least in part, through modulation of the hippocampal serotonergic system reactivity.



Similar content being viewed by others
References
Birzniece V, Bäckström T, Johansson IM, Lindblad C, Lundgren P, Löfgren M, Olsson T, Ragagnin G, Taube M, Turkmen S, Wahlström G, Wang MD, Wihlbäck AC, Zhu D (2006) Neuroactive steroid effects on cognitive functions with a focus on the serotonin and GABA systems. Brain Res Rev 51:212–239. https://doi.org/10.1016/j.brainresrev.2005.11.001
Buhot MC, Martin S, Segu L (2000) Role of serotonin in memory impairment. Ann Med 32:210–221. https://doi.org/10.3109/07853890008998828
Caligaris L, Astrada J, Taleisnik S (1972) Effect of oestrogen and progesterone on the release of luteinizing hormone in pseudopregnant rats. Acta Endocrinol (Copenh) 70:163–166
Cammarota M, Bevilaqua L, Rossato J, Lima R, Medina JH, Izquierdo I (2008) Parallel memory processing by the CA1 region of the dorsal hippocampus and the basolateral amygdala. Proc Natl Acad Sci USA 105:10279–10284. https://doi.org/10.1073/pnas.0805284105
Carli M, Tranchina S, Samanin R (1992) 8-Hydroxy-2-(di-n-propylamino) tetralin, a 5-HT1A receptor agonist, impairs performance in a passive avoidance task. Eur J Pharmacol 211:227–234. https://doi.org/10.1016/0014-2999(92)90533-A
Dabrowska J, Brylinski M (2006) Stereoselectivity of 8-OH-DPAT toward the serotonin 5-HT1A receptor: biochemical and molecular modeling study. Biochem Pharmacol 72(4):498–511. https://doi.org/10.1016/j.bcp.2006.05.008
Devall AJ, Santos JM, Fry JP, Honour JW, Brandão ML, Lovick TA (2015) Elevation of brain allopregnanolone rather than 5-HT release by short term, low dose fluoxetine treatment prevents the estrous cycle-linked increase in stress sensitivity in female rats. Eur Neuropsychopharmacol 25(1):113–123. https://doi.org/10.1016/j.euroneuro.2014.11.017
Díaz-Véliz G, Urresta F, Dussaubat N, Mora S (1994) Progesterone effects on the acquisition of conditioned avoidance responses and other motoric behaviors in intact and ovariectomized rats. Psychoneuroendocrinology 19:387–394. https://doi.org/10.1016/j.pbb.2007.05.017
Do Rego JL, Seong JY, Burel D, Leprince J, Luu-The V, Tsutsui K, Tonon MC, Pelletier G, Vaudry H (2009) Neurosteroid biosynthesis: enzymatic pathways and neuroendocrine regulation by neurotransmitters and neuropeptides. Front Neuroendocrinol 30:259–301. https://doi.org/10.1016/j.yfrne.2009.05.006
Escudero C, Giuliani F, Casas S, Bazzocchini V, Yunes R, Cabrera R (2012) Allopregnanolone prevents memory impairment: effect on mRNA expression and enzymatic activity of hippocampal 3-α hydroxysteroid oxide-reductase. Brain Res Bull 87:280–285. https://doi.org/10.1016/j.brainresbull.2011.11.019
Evans J, Sun Y, McGregor A, Connor B (2012) Allopregnanolone regulates neurogenesis and depressive/anxiety-like behaviour in a social isolation rodent model of chronic stress. Neuropharmacol 63:1315–1326. https://doi.org/10.1016/j.neuropharm.2012.08.012
Fozard J, Saxena P (1991) Serotonin: molecular biology, receptors and functional effects. Birkhauser, Basel
Freund TF, Gulyas AI, Acsady L, Gorcs T, Toth K (1990) Serotonergic control of the hippocampus via local inhibitory interneurons. Proc Natl Acad Sci 87:8501–8505
Frick K, Kim J, Tuscher J, Fortress A (2015) Sex steroid hormones matter for learning and memory: estrogenic regulation of hippocampal function in male and female rodents. Learn Mem 22:472–493. https://doi.org/10.1101/lm.037267.114
Frye CA (2009) Neurosteroids’ effects and mechanisms for social, cognitive, emotional, and physical functions. Psychoneuroendocrinology 34:143–161. https://doi.org/10.1016/j.psyneuen.2009.07.005
Frye CA, Paris JJ, Rhodes ME (2009) Increasing 3alpha, 5alpha-THP following inhibition of neurosteroid biosynthesis in the ventral tegmental area reinstates anti-anxiety, social, and sexual behavior of naturally receptive rats. Reproduction 137:119–128. https://doi.org/10.1530/REP-08-0250
Giuliani FA, Yunes R, Mohn CE, Laconi M, Rettori V, Cabrera R (2011) Allopregnanolone induces LHRH and glutamate release through NMDA receptor modulation. Endocrine 40:21–26. https://doi.org/10.1007/s12020-011-9451-8
Glikmann-Johnston Y, Saling MM, Reutens DC, Stout JC (2015) Hippocampal 5-HT1A receptor and spatial learning and memory. Front Pharmacol 6:289. https://doi.org/10.3389/fphar.2015.00289
Hedlund PB, Sutcliffe G (2004) Functional, molecular and pharmacological advances in 5-HT7 receptor research. Cell 25:481–486. https://doi.org/10.1016/j.tips.2004.07.002
Jez JL, Penning TM (2001) The aldo-keto reductase (AKR) superfamily: an update. Chem Biol Interact 130:499–525. https://doi.org/10.1016/S0009-2797(00)00295-7
Kaura V, Ingram CD, Gartside SE, Young AH, Judge SJ (2007) The progesterone metabolite allopregnanolone potentiates GABAA receptor-mediated inhibition of 5-HT neuronal activity. European Neuropsychopharmacol 17:108–115. https://doi.org/10.1016/j.euroneuro.2006.02.006
Lesch K, Waider J (2012) Serotonin in the modulation of neural plasticity and networks: implications for neurodevelopmental disorders. Neuron 76(1):175–191. https://doi.org/10.1016/j.neuron.2012.09.013
Liy-Salmeron G, Meneses A (2007) Role of 5-HT1–7 receptors in short- and long-term memory for an autoshaping task: Intrahippocampal manipulations. Brain Res 1147:140–147. https://doi.org/10.1016/j.brainres.2007.02.007
Lu NZ, Bethea CL (2002) Ovarian steroid regulation of 5-HT1A receptor binding and G protein activation in female monkeys. Neuropsychopharmacol 27:12–24. https://doi.org/10.1016/S0893-133X(01)00423-7
Mason JP, Dring LG, Caldwell J (1995) Pharmacokinetics of the 5-hydroxytryptamine1A agonist 8-hydroxy-2-(N,N-di-n-propylamino)tetralin (8-OHDPAT) in the rat after intravenous and oral administration. Xenobiotica 25(12):1371–1380
Meneses A (2007) Do serotonin (1-7) receptors modulate short and long-term memory? Neurobiol Learn Mem 87:561–572. https://doi.org/10.1016/j.nlm.2006.12.005
Meneses A, Perez-Garcia G (2007) 5-HT (1A) receptors and memory. Neurosci Biobehav Rev 31:705–727. https://doi.org/10.1016/j.neubiorev.2007.02.001
Myhrer T (2003) Neurotransmitter systems involved in learning and memory in the rat: a meta-analysis based on studies of four behavioral tasks. Brain Res Brain Res Rev 41:268–287. https://doi.org/10.1016/S0165-0173(02)00268-0
Nanfaro F, Cabrera R, Bazzocchini V, Laconi M, Yunes R (2010) Pregnenolone sulfate infused in lateral septum of male rats impairs novel object recognition memory. Pharmacol Rep. 62(2):265–272
Ogren S, Eriksson T, Elvander-Tottie E, D’Addario C, Ekström J, Svenningsson P, Meister B, Kehr J, Stiedl O (2008) The role of 5-HT(1A) receptors in learning and memory. Behav Brain Res. 195(1):54–77. https://doi.org/10.1016/j.bbr.2008.02.023
Osborne LM, Gispen F, Sanyal A, Yenokyan G, Meilman S, Payne JL (2017) Lower allopregnanolone during pregnancy predicts postpartum depression: an exploratory study. Psychoneuroendocrinology. 79:116–121. https://doi.org/10.1016/j.psyneuen.2017.02.012
Pinna G, Rasmusson AM (2012) Upregulation of neurosteroid biosynthesis as a pharmacological strategy to improve behavioral deficits in a putative mouse model of PTSD. J Neuroendocrinol. 24(1):102–116. https://doi.org/10.1111/j.1365-2826.2011.02234.x
Porcu P, Barron AM, Frye CA, Walf AA, Yang SY, He XY, Morrow AL, Panzica GC, Melcang RC (2016) Neurosteroidogenesis today: novel targets for neuroactive steroid synthesis and action and their relevance for translational research. J Neuroendocrinol. 28(2):12351. https://doi.org/10.1111/jne.12351
Prakapenka A, Hiroi R, Quihuis A, Carson C, Pate S, Berns-Leone C, Fox C, Sirianni R, Bimonte-Nelson H (2018) Contrasting effects of individual versus combined estrogen and progestogen regimens as working memory load increases in middle-aged ovariectomized rats: one plus one does not equal two. Neurobiol Aging 64:1–14. https://doi.org/10.1016/j.neurobiolaging.2017.11.015
Rebas E, Radzik T, Boczek T, Zylinska L (2017) Calcium-engaged mechanisms of nongenomic action of neurosteroids. Curr Neuropharmacol. 15(8):1174–1191. https://doi.org/10.2174/1570159X15666170329091935
Roozbehi A, Sharafi MT, Karimi F, Kamali A (2017) The impact of using different doses of progesterone on memory performance. Bratisl Med J 118(7):405–407
Ruat M, Traiffort E, Leurs R, Tardivel-Lacombe J, Diaz J, Arrang JM, Schwartz JC (1993) Molecular cloning, characterization, and localization of a high-affinity serotonin receptor (5-HT7) activating cAMP formation. Proc Natl Acad Sci USA 90(18):8547–8551
Tuem KB, Atey TM (2017) Neuroactive steroids: receptor interactions and responses. Front Neurol. 8:442. https://doi.org/10.3389/fneur.2017.00442
Tuscher J, Fortress A, Kim J, Frick K (2015) Regulation of object recognition and object placement by ovarian sex steroid hormones. Behav Brain Res 15(285):140–157. https://doi.org/10.1016/j.bbr.2014.08.001
Vahid-Ansari F, Daigle M, Manzini MC et al (2017) Abrogated Freud-1/Cc2d1a Repression of 5-HT1A autoreceptors induces fluoxetine-resistant anxiety/depression-like behavior. J Neurosci. 37(49):11967–11978. https://doi.org/10.1523/JNEUROSCI.1668-17.2017
Walf AA, Rhodes ME, Frye CA (2006) Ovarian steroids enhance object recognition in naturally cycling and ovariectomized, hormone-primed rats. Neurobiol Learn Mem 86:35–46. https://doi.org/10.1016/j.nlm.2006.01.004
Acknowledgements
This study was financially supported by the Grant PIP 11220100100126/11 of the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina, and the Grant 113/07 of the Universidad de Mendoza, Argentina. We wish to thank Mr N. Persia (IMBECU-CONICET) for his technical assistance and Ms. A. Byrne for language assistance. Finally, we are also grateful to V. Bazzocchini and S. García for their kind assistance during some experiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Escudero, C., Giuliani, F., Bernedo, M.M. et al. Allopregnanolone reversion of estrogen and progesterone memory impairment: interplay with serotonin release. J Neural Transm 126, 1321–1328 (2019). https://doi.org/10.1007/s00702-019-02052-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-019-02052-6