Abstract
This work demonstrates the inhibition effects of single-stranded (ssDNA) on the oxidase-like activity of a mixed-valence state cerium-based metal-organic framework, denoted as MVC-MOF. The MVC-MOF was synthesized by partial oxidation of cerium(III) which leads to the presence of both Ce(III) and Ce(IV) ions. The latter endows the MVC-MOF with a typical oxidase-like activity. However, on addition of ssDNA, the catalytic activity of the MVC-MOF is inhibited because it binds the MVC-MOF and thereby shield its active sites. This prevents the access of substrates. The inhibition by ssDNA depends on its length but not its sequence. By contrast, negligible changes in the oxidase-mimicking activity are observed if double-stranded DNA (dsDNA) is added. By employing a thymine-rich ssDNA (T-ssDNA) as a model DNA, a colorimetric assay was developed for the determination of Hg(II). This ion binds to T-ssDNA and causes the formation of T-dsDNA. Hence, the oxidase-mimicking activity is compromised. By using the oxidase substrate 3,3′,5,5′-tetramethylbenzidine that gives a colored product in the presence of oxygen, the assay has a linear response that covers 0.05 to 6 μM Hg(II) with a detection limit of 10.5 nM, and exhibits high selectivity over other metal ions. The assay was successfully applied to the determination of Hg(II) in environmental water samples.

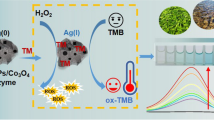
Schematic of the inhibition effect of ssDNA on the oxidase-like activity of MVC-MOF that converts colorless TMB to oxTMB with blue color in the presence of oxygen, and its application in the construction of a colorimetric assay for Hg(II) determination.







Similar content being viewed by others
References
Li J-R, Sculley J, Zhou H-C (2012) Metal-organic frameworks for separations. Chem Rev 112:869–932
Zheng H, Zhang Y, Liu L, Wan W, Guo P, Nyström AM, Zou X (2016) One-pot synthesis of metal-organic frameworks with encapsulated target molecules and their applications for controlled drug delivery. J Am Chem Soc 138:962–968
Liu D, Lu K, Poon C, Lin W (2014) Metal-organic frameworks as sensory materials and imaging agents. Inorg Chem 53:1916–1924
Tan H, Li Q, Zhou Z, Ma C, Song Y, Xu F, Wang L (2015) A sensitive fluorescent assay for thiamine based on metal-organic frameworks with intrinsic peroxidase-like activity. Anal Chim Acta 856:90–95
Ai L, Li L, Zhang C, Fu J, Jiang J (2013) MIL-53(Fe): a metal-organic framework with intrinsic peroxidase-like catalytic activity for colorimetric biosensing. Chem Eur J 19:15105–15108
Wang N, Sun J, Chen L, Fan H, Ai S (2015) A Cu2(OH)3Cl-CeO2 nanocomposite with peroxidase-like activity, and its application to the determination of hydrogen peroxide, glucose and cholesterol. Microchim Acta 182:1733–1738
Lu Y, Yu J, Ye W, Yao X, Zhou P, Zhang H, Zhao S, Jia L (2016) Spectrophotometric determination of mercury(II) ions based on their stimulation effect on the peroxidase-like activity of molybdenum disulfide nanosheets. Microchim Acta 183:2481–2489
Zhao Y, Qiang H, Chen Z (2017) Colorimetric determination of Hg(II) based on a visually detectable signal amplification induced by a Cu@Au-Hg trimetallic amalgam with peroxidase-like activity. Microchim Acta 184:107–115
Zhang J-W, Zhang H-T, Du Z-Y, Wang X, Yu S-H, Jiang H-L (2014) Water-stable metal-organic frameworks with intrinsic peroxidase-like catalytic activity as a colorimetric biosensing platform. Chem Commun 50:1092–1094
Xiong Y, Chen S, Ye F, Su L, Zhang C, Shen S, Zhao S (2015) Synthesis of a mixed valence state Ce-MOF as an oxidase mimetic for the colorimetric detection of biothiols. Chem Commun 51:4635–4638
Cho EJ, Lee J-W, Ellington AD (2009) Applications of aptamers as sensors. Annu Rev Anal Chem 2:241–264
Goulko AA, Li F, Chris Le X (2009) Bioanalytical applications of aptamer and molecular-beacon probes in fluorescence-affinity assays. TrAC Trends Anal Chem 28:878–892
Wang H, Yang R, Yang L, Tan W (2009) Nucleic acid conjugated nanomaterials for enhanced molecular recognition. ACS Nano 3:2451–2460
Wang G, Wang Y, Chen L, Choo J (2010) Nanomaterial-assisted aptamers for optical sensing. Biosens Bioelectron 25:1859–1868
Pautler R, Kelly EY, Huang P-JJ, Cao J, Liu B, Liu J (2013) Attaching DNA to nanoceria: regulating oxidase activity and fluorescence quenching. ACS Appl Mater Interfaces 5:6820–6825
Ono A, Togashi H (2004) Highly selective oligonucleotide-based sensor for mercury(II) in aqueous solutions. Angew Chem Int Ed 43:4300–4302
Li Q, Wang C, Tan H, Tang G, Gao J, Chen C-H (2016) A turn on fluorescent sensor based on lanthanide coordination polymer nanoparticles for the detection of mercury(II) in biological fluids. RSC Adv 6:17811–17817
Tan H, Wu J, Chen Y (2014) Terbium(III) based coordination polymer microparticles as a luminescent probe for ascorbic acid. Microchim Acta 181:1–7
Tan H, Liu B, Chen Y (2012) Lanthanide coordination polymer nanoparticles for sensing of mercury(II) by photoinduced electron rransfer. ACS Nano 6:10505–10511
Liu YL, Zhao XJ, Yang XX, Li YF (2013) A nanosized metal-organic framework of Fe-MIL-88NH2 as a novel peroxidase mimic used for colorimetric detection of glucose. Analyst 138:4526–4531
Liu M, Zhang Q, Zhao H, Chen S, Yu H, Zhang Y, Quan X (2011) Controllable oxidative DNA cleavage-dependent regulation of graphene/DNA interaction. Chem Commun 47:4084–4086
Chen X, Zhao T, Zou J (2009) A novel mimetic peroxidase catalyst by using magnetite-containing silica nanoparticles as carriers. Microchim Acta 164:93–99
Heckert EG, Karakoti AS, Seal S, Self WT (2008) The role of cerium redox state in the SOD mimetic activity of nanoceria. Biomaterials 29:2705–2709
Yu P, Hayes SA, O’Keefe TJ, O’Keefe MJ, Stoffer JO (2006) The phase stability of cerium species in aqueous systems: II. The systems. Equilibrium considerations and pourbaix diagram calculations. J Electrochem Soc 153:C74–C79
Wang Y, Yang F, Yang X (2010) Colorimetric detection of mercury(II) ion using unmodified silver nanoparticles and mercury-specific oligonucleotides. ACS Appl Mater Interfaces 2:339–342
Tang G, Gao J, Wang C, Tan H (2017) Luminescent lanthanide coordination polymer as a platform for DNA colorimetric detection. Sensors Actuators B Chem 244:571–576
Wang G-Y, Song C, Kong D-M, Ruan W-J, Chang Z, Li Y (2014) Two luminescent metal-organic frameworks for the sensing of nitroaromatic explosives and DNA strands. J Mater Chem A 2:2213–2220
Miyake Y, Togashi H, Tashiro M, Yamaguchi H, Oda S, Kudo M, Tanaka Y, Kondo Y, Sawa R, Fujimoto T, Machinami T, Ono A (2006) MercuryII-mediated formation of thymine-HgII-thymine base pairs in DNA duplexes. J Am Chem Soc 128:2172–2173
Li T, Dong S, Wang E (2009) Label-free colorimetric detection of aqueous mercury ion (Hg2+) using Hg2+-modulated G-quadruplex-based DNAzymes. Anal Chem 81:2144–2149
Wang Y, Geng F, Cheng Q, Xu H, Xu M (2011) Oligonucleotide-based label-free Hg2+ assay with a monomer-excimer fluorescence switch. Analyst 136:4284–4288
Zhang L, Li T, Li B, Li J, Wang E (2010) Carbon nanotube-DNA hybrid fluorescent sensor for sensitive and selective detection of mercury(II) ion. Chem Commun 46:1476–1478
Oh BN, Park S, Ren J, Jang YJ, Kim SK, Kim J (2011) Label-free emission assay of mercuric ions using DNA duplexes of poly(dT). Dalton Trans 40:6494–6499
Xu J-P, Song Z-G, Fang Y, Mei J, Jia L, Qin AJ, Sun JZ, Ji J, Tang BZ (2010) Label-free fluorescence detection of mercury(II) and glutathione based on Hg2+-DNA complexes stimulating aggregation-induced emission of a tetraphenylethene derivative. Analyst 135:3002–3007
He S, Song B, Li D, Zhu C, Qi W, Wen Y, Wang L, Song S, Fang H, Fan C (2010) A graphene nanoprobe for rapid, sensitive, and multicolor fluorescent DNA analysis. Adv Funct Mater 20:453–459
Clever GH, Kaul C, Carell T (2007) DNA-metal base pairs. Angew Chem Int Ed 46:6226–6236
Acknowledgements
This work was supported by the Natural Science Foundation of China (21765010, 21305054), the Specialized Research Fund for the Doctoral Program of Higher Education of China (20133604120002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 669 kb)
Rights and permissions
About this article
Cite this article
Wang, C., Tang, G. & Tan, H. Colorimetric determination of mercury(II) via the inhibition by ssDNA of the oxidase-like activity of a mixed valence state cerium-based metal-organic framework. Microchim Acta 185, 475 (2018). https://doi.org/10.1007/s00604-018-3011-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-3011-3