Abstract
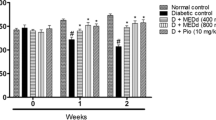
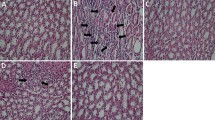
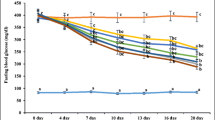
Diabetes is a chronic metabolic disorder characterized by chronic hyperglycemia which causes secondary pathophysiological changes in multiple organ systems. Clinically used oralhypoglycemic agents are associated with a lot of side effects and high cost of treatment. As per ethnobotanical relevance, traditional medicines and natural products offer a valuable alternative to the oralhypoglycemic drugs. This study was hypothesized to evaluate the antidiabetic and nephroprotective activities of Mentha aquatica in streptozotocin (STZ)-induced diabetic rats. Diabetes was induced by a single intraperitoneal injection of STZ at a dosage of 40 mg/kg bw. At the end of the study, overnight-fasted rats were dissected, and the blood and kidney samples were analyzed for biochemical and histopathological analysis. Oral administration of aqueous extract of leaves of Mentha aquatica (AELMA) at a dose of 100 mg/kg bw/day for 90 days significantly decreased the level of fasting blood glucose, HbA1c, TC, TG, plasma urea, creatinine, urine albumin, and kidney lipid peroxidation and increased the body weight, insulin, HDL cholesterol, plasma albumin, urinary urea, urinary creatinine, and antioxidant enzyme activities. The present study demonstrates that aqueous extract leaves of Mentha aquatica exert significant antidiabetic activity by stimulating secretion of insulin and nephroprotective potential activity by reducing the lipid peroxidation and enhancing the scavenging ability of antioxidant defense system in the body.






Similar content being viewed by others
References
Arif MI, Rafiq M, Ghaffar A (2009) Host plants of cotton mealybug (Phenacoccus solenopsis): a new menace to cotton agroecosystem of Punjab, Pakistan. Int J Agric Biol 11(2):163–167
Atanasov AG, Waltenberger B, Pferschy-Wenzig EM, Linder T, Wawrosch C, Uhrin P, Temml V, Wang L, Schwaiger S, Heiss EH, Rollinger JM (2015) Discovery and resupply of pharmacologically active plant-derived natural products: a review. Biotechnol Adv 33(8):1582–1614. https://doi.org/10.1016/j.biotechadv.2015.08.001
Chen YM, Miner JH (2012) Glomerular basement membrane and related glomerular disease. Transl Res 160(4):291–297. https://doi.org/10.1016/j.trsl.2012.03.004
Conforti F, Sosa S, Marrelli M, Menichini F, Statti GA, Uzunov D, Tubaro A, Menichini F, Della Loggia R (2008) In vivo anti-inflammatory and in vitro antioxidant activities of Mediterranean dietary plants. J Ethnopharmacol 116(1):144–151. https://doi.org/10.1016/j.jep.2007.11.015
Dorman HD, Koşar M, Kahlos K, Holm Y, Hiltunen R (2003) Antioxidant properties and composition of aqueous extracts from Mentha species, hybrids, varieties, and cultivars. J Agric Food Chem 51(16):4563–4569. https://doi.org/10.1021/jf034108k
Forbes JM, Coughlan MT, Cooper ME (2008) Oxidative stress as a major culprit in kidney disease in diabetes. Diabetes 57(6):1446–1454. https://doi.org/10.2337/db08-0057
Fraga CG, Leibovitz BE, Tappel AL (1998) Lipid peroxidation measured as thiobarbituric acid-reactive substances in tissue slices: characterization and comparison with homogenates and microsomes. Free Radic Biol Med 4(3):155–161. https://doi.org/10.1016/0891-5849(88)90023-8
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases the first enzymatic step in mercapturic acid formation. J Biol Chem 249(22):7130–7139
Harbone JB (1998) A guide to modern techniques of plant analysis-phytochemical methods. pp. 253-262. https://doi.org/10.1016/j.jep.2011.12.022
International Diabetes Federation-IDF (2017) Diabetes Atlas 8th Edition Global fact sheet
Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometer assay of superoxide dismutase. Indian J Biochem Biophys 21:130–132
Konda PY, Dasari S, Konanki S, Nagarajan P (2019) In vivo antihyperglycemic, antihyperlipidemic, antioxidative stress and antioxidant potential activities of Syzygium paniculatum Gaertn. In Streptozotocin-induced diabetic rats. Heliyon 5(3):1–22. https://doi.org/10.1016/j.heliyon.2019.e01373
Kulkarni YA, Garud MS (2016) Bauhinia variegata (Caesalpiniaceae) leaf extract: an effective treatment option in type I and type II diabetes. Biomed Pharmacother 83:122–129. https://doi.org/10.1016/j.biopha.2016.06.025
Kumar MJ, Prabhakar Y, Saritha M, Tilak TK, Nabi SA, Ali MS, Peddanna N, Rao CA (2016) Effect of flavonoid rich fraction of Andrographis echioides in streptozotocin-induced diabetic rats. J Pharm Chem 1(10):16–20
Mimica-Dukić N, Božin B, Soković M, Mihajlović B, Matavulj M (2003) Antimicrobial and antioxidant activities of three Mentha species essential oils. Planta Med 69(05):413–419. https://doi.org/10.1055/s-2003-39704
Miyazawa M, Watanabe H, Umemoto K, Kameoka H (1998) Inhibition of acetylcholine esterase activity by essential oils of Mentha species. J Agric Food Chem 46(9):3431–3434. https://doi.org/10.1021/jf9707041
Olsen HT, Stafford GI, Van Staden J, Christensen SB, Jäger AK (2008) Isolation of the MAO-inhibitor naringenin from Mentha aquatica L. J Ethnopharmacol 117(3):500–502. https://doi.org/10.1016/j.jep.2008.02.015
Prabhakar Y, Ali MS, Kumar MJ, Tilak TK, Rao CA (2013) Evaluation of antioxidant activities of aqueous extract of stem bark of Boswellia ovalifoliolata in streptozotocin induced diabetic rats. J Pharm Chem 7:19–24
Pruthia R, Pitchera D, Dawnayb A (2012) UK Renal Registry 14th annual report: chapter 9 biochemical variables amongst UK adult dialysis patients in 2010: national and centre specific analyses. Nephron Clin Pract 120:175–210. https://doi.org/10.1159/000342852
Ramya N, Peddanna K, Prabhakar YK, Apparao C (2014) Evaluation of anti-hyperglycemic activity of Narengi crenulata leaf in STZ induced diabetic rats. Asian J Biomed Pharm 4(39):35–39
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra W (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179(4073):588–590
Sinha AK (1972) Colorimetric assay of catalase. Anal Biochem 47(2):389–394. https://doi.org/10.1016/0003-2697(72)90132-7
Stafford GI, Pedersen ME, van Staden J, Jäger AK (2008) Review on plants with CNS-effects used in traditional south African medicine against mental diseases. J Ethnopharmacol 119(3):513–537. https://doi.org/10.1016/j.jep.2008.08.010
Tang W, Martin KA, Hwa J (2012) Aldose reductase, oxidative stress, and diabetic mellitus. Front Pharmacol 3(87):1–8. https://doi.org/10.3389/fphar.2012.00087
Wang J, Wang F, Yun H, Zhang H, Zhang Q (2012) Effect and mechanism of fucoidan derivatives from Laminaria japonica in experimental adenine-induced chronic kidney disease. J Ethnopharmacol 139(3):807–813
Acknowledgments
We would like to acknowledge the anonymous referees for helpful comments and suggestions.
Author information
Authors and Affiliations
Contributions
PN designed the study and wrote the original draft. YKP, JYE, SD, RK, and KKJ performed the experiments, are involved in data curation, and wrote the manuscript. YKP and PN reviewed and edited the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yellanur Konda, P., Egi, J.Y., Dasari, S. et al. Ameliorative effects of Mentha aquatica on diabetic and nephroprotective potential activities in STZ-induced renal injury. Comp Clin Pathol 29, 189–199 (2020). https://doi.org/10.1007/s00580-019-03042-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-019-03042-6