Abstract
Background
Lower activation of higher threshold (type-II) motor units (MUs) has been suggested in children compared with adults. We examined child–adult differences in discrete MU activation of the flexor carpi radialis (FCR).
Methods
Fifteen boys (10.2 ± 1.4 years), and 17 men (25.0 ± 2.7 years) completed 2 laboratory sessions. Following a habituation session, maximal voluntary isometric wrist flexion torque (MVIC) was determined before completing trapezoidal isometric contractions at 70%MVIC. Surface electromyography was captured by Delsys Trigno Galileo sensors and decomposed into individual MU action potential trains. Recruitment threshold (RT), and MU firing rates (MUFR) were calculated.
Results
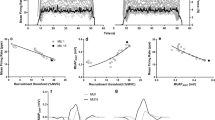
MVIC was significantly greater in men (10.19 ± 1.92 Nm) than in boys (4.33 ± 1.47 Nm) (p < 0.05), but not statistically different after accounting for differences in body size. Mean MUFR was not different between boys (17.41 ± 7.83 pps) and men (17.47 ± 7.64 pps). However, the MUFR–RT slope was significantly (p < 0.05) steeper (more negative) in boys, reflecting a progressively greater decrease in MUFR with increasing RT. Additionally, boys recruited more of their MUs early in the ramped contraction.
Conclusion
Compared with men, boys tended to recruit their MUs earlier and at a lower percentage of MVIC. This difference in MU recruitment may explain the greater decrease in MUFR with increasing RT in boys compared with men. Overall, these findings suggest an age-related difference in the neural strategy used to develop moderate–high torque in wrist flexors, where boys recruit more of their MUs earlier in the force gradation process, possibly resulting in a narrower recruitment range.




Similar content being viewed by others
Data availability
Data are available from the corresponding author upon reasonable request.
Abbreviations
- CV:
-
Coefficient of variation
- EMGRMS :
-
Root mean square of the electromyography signal
- ECR:
-
Extensor Carpi Radialis
- FCR:
-
Flexor Carpi Radialis
- FDI:
-
First dorsal interossei
- LBM:
-
Lean body mass
- MU:
-
Motor unit
- MUAP:
-
Motor-unit action potential
- MUFR:
-
Motor-unit firing rates
- MVIC:
-
Maximal voluntary isometric contraction
- P–P:
-
Peak to peak
- PHV:
-
Peak height velocity
- RT:
-
Recruitment threshold
- sEMG:
-
Electromyography
- STA:
-
Spike trigger average
References
Aagaard P, Simonsen EB, Andersen JL, Magnusson P, Dyhre-poulsen P, Dyhre-Poulsen P (2002) Neural adaptation to resistance training: changes in evoked V-wave and H-reflex responses. J Appl Physiol 92:2309–2318. https://doi.org/10.1152/japplphysiol.01185
Blimkie, C. J. R. (1989). Age- and sex-associated variation in strength during childhood: anthropometric, morphologic, neurological, biomechanical, genetic and physical activity correlates. In D. Gisolfi, C., Lamb (Eds.), Perspectives in exercise science and sports, Benchmark, (pp. 99-157).
Chalchat E, Piponnier E, Bontemps B, Julian V, Bocock O, Duclos M, Ratel S, Martin V (2019) Characteristics of motor unit recruitment in boys and men at maximal and submaximal force levels. Exp Brain Res 237:1289–1302. https://doi.org/10.1007/s00221-019-05508-z
Clamann HP (1993) Motor unit recruitment and the gradation of muscle force. Phys Ther 73(12):830–843. https://doi.org/10.1093/ptj/73.12.830
De Ste Croix MBA, Armstrong N, Welsman J (1999) Concentric isokinetic leg strength in pre-teen, teenage and adult males and females. Biol Sport 16(2):75–86
DeLuca C, Contessa P (2012) Hierarchical control of motor units in voluntary contractions. J Neurophysiol 107:178–195. https://doi.org/10.1152/jn.00961.2010.-For
DeLuca C, Erim Z (1994) Common drive of motor units in regulation of muscle force. Trends Neurosci 17(7):299–305
DeLuca C, Hostage E (2010) Relationship between firing rate and recruitment threshold of motoneurons in voluntary isometric contractions. J Neurophysiol 104:1034–1046. https://doi.org/10.1152/jn.01018.2009
Dobrzynska, Z., and Celichowski, J. (2016). Changes in contractile properties and action potentials of motor units in the rat medial gastrocnemius muscle during maturation. Journal of Physiology and Pharmacology, 67(1), 139–150. http://jpp.krakow.pl/journal/archive/02_16/pdf/139_02_16_article.pdf
Dotan R, Mitchell C, Cohen R, Gabriel D, Klentrou P, Falk B (2013) Explosive sport training and torque kinetics in children. Appl Physiol Nutr Metab 38(7):740–745. https://doi.org/10.1139/apnm-2012-0330
Dotan R, Mitchell C, Cohen R, Klentrou P, Gabriel D, Falk B (2012) Child—adult differences in muscle activation—a review. Pediatr Exerc Sci 24:2–21
Esbjörnsson M, Dahlström M, Gierup J, Jansson E (2021) Muscle fiber size in healthy children and adults in relation to sex and fiber types. Muscle Nerve 63:586–592. https://doi.org/10.1002/mus.27151
Falk B, Usselman C, Dotan R, Brunton L, Klentrou P, Shaw J, Gabriel D (2009) Child–adult differences in muscle strength and activation pattern during isometric elbow flexion and extension. Appl Physiol Nutr Metab 34(4):609–615. https://doi.org/10.1139/H09-020
Farina D, Merletti R, Enoka RM, Amann M, Blain GM, Proctor LT, Sebranek JJ, Pegelow DF, Dempsey JA (2004) The extraction of neural strategies from the surface EMG Neural Control of Movement The extraction of neural strategies from the surface EMG. J Appl Physiol 96:1486–1495. https://doi.org/10.1152/japplphysiol.01070.2003
Frost G, Bar-Or OO, Dowling J, Dyson K (2002) Explaining differences in the metabolic cost and efficiency of treadmill locomotion in children. J Sports Sci 20(6):451–461. https://doi.org/10.1080/02640410252925125
Frost G, Dowling J, Dyson K, Bar-Or O (1997) Cocontraction in three age groups of children during treadmill locomotion. J Electromyogr Kinesiol 7(3):179–186
Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, Zijdenbos A, Paus T, Evans AC, Rapoport JL (1999) Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci 2(10):861–863. https://doi.org/10.1038/13158
Groeschel S, Vollmer B, King MD, Connelly A (2010) Developmental changes in cerebral grey and white matter volume from infancy to adulthood. Int J Dev Neurosci 28(6):481–489. https://doi.org/10.1016/j.ijdevneu.2010.06.004
Grosset JF, Mora I, Lambertz D, Pérot C (2008) Voluntary activation of the triceps surae in prepubertal children. J Electromyogr Kinesiol 18:455–465. https://doi.org/10.1016/j.jelekin.2006.11.002
Henneman E (1957) Relation between size of neurons and their susceptibility to discharge. Science 126:1345–1347. https://doi.org/10.1126/science.126.3287.1345
Herda TJ, Miller JD, Wray ME, Sterczala AJ, Dimmick HL, Trevino MA (2019) Motor unit firing rates of the first dorsal interosseous differ between male and female children aged 8–10 years. Hum Mov Sci 66:416–424. https://doi.org/10.1016/j.humov.2019.04.011
Herda TJ, Parra ME, Miller JD, Sterczala AJ, Kelly MR (2020) Measuring the accuracies of motor unit firing times and action potential waveforms derived from surface electromyographic decomposition. J Electromyogr Kinesiol 52:102421. https://doi.org/10.1016/j.jelekin.2020.102421
Hug F, Laplaud D, Savin B, Grélot L (2003) Occurrence of electromyographic and ventilatory thresholds in professional road cyclists. Eur J Appl Physiol 90(5–6):643–646. https://doi.org/10.1007/s00421-003-0949-5
Huttenlocher PR, Dabholkar AS (1997) Regional differences in synaptogenesis in human cerebral cortex. J Comp Neurol 387(2):167–178
Kannas T, Kellis E, Arampatzi F (2010) Medial gastrocnemius architectural properties during isometric contractions in boys and men. Pediatr Exerc Sci 22(1):152–164
Koh THHG, Eyre JA (1988) Maturation of corticospinal tracts assessed by electromagnetic stimulation of the motor cortex. Arch Dis Child 63(11):1347–1352. https://doi.org/10.1136/adc.63.11.1347
Kumar N, Oliver JL, Lloyd RS, Pedley JS, Radnor JM (2021) The influence of growth, maturation and resistance training on muscle-tendon and neuromuscular adaptations: a narrative review. Sports 9(59):9050059
Lambertz D, Mora I, Grosset JF, Perot C (2003) Evaluation of musculotendinous stiffness in prepubertal children and adults, taking into account muscle activity. J Appl Physiol 95(1):64–72. https://doi.org/10.1152/japplphysiol.00885.2002
Lexell J, Sjostrom M, Nordlund A, Taylor CC (1992) Growth and development of human muscle: a quantitative morphological study of whole vastus lateralis from childhood to adult age. Muscle Nerve 15:404–409
Long D, Dotan R, Pitt B, McKinlay B, O’Brien TD, Tokuno C, Falk B (2017) The electromyographic threshold in girls and women. Pediatr Exerc Sci 29(1):84–93. https://doi.org/10.1123/pes.2016-0056
McIntosh JS, Ringqvist M, Schmidt EM (1985) Fiber type composition of monkey forearm muscle. Anat Rec 211(4):403–409. https://doi.org/10.1002/ar.1092110405
Metaxas T, Mandroukas A, Michailidis Y, Koutlianos N, Christoulas K, Ekblom B (2019) Correlation of fber-Type composition and sprint performance in Youth Soccer players. J Strength Cond Res 33(10):2629–2634. https://doi.org/10.1519/JSC.0000000000003320
Miller JD, Sterczala AJ, Trevino MA, Wray ME, Dimmick HL, Herda TJ (2019) Motor unit action potential amplitudes and firing rates during repetitive muscle actions of the first dorsal interosseous in children and adults. Eur J Appl Physiol 119(4):1007–1018. https://doi.org/10.1007/s00421-019-04090-0
Moritani T, DeVries H (1978) Re-examination of the relationship between the surface integrated electromyogram (IEMG) and force of isometric contraction. Am J Phys Med 57:263–277
O’Brien TD, Reeves ND, Baltzopoulos V, Jones DA, Maganaris CN (2010) In vivo measurements of muscle specific tension in adults and children. Exp Physiol 95(1):202–210. https://doi.org/10.1113/expphysiol.2009.048967
Oliver JL, Smith PM (2010) Neural control of leg stiffness during hopping in boys and men. J Electromyogr Kinesiol 20(5):973–979. https://doi.org/10.1016/j.jelekin.2010.03.011
Parra ME, Miller JD, Sterczala AJ, Trevino MA, Dimmick HL, Herda TJ (2020) Differences in the firing rate versus recruitment threshold relationships of the vastus lateralis in children ages 7–10 years and adults. Hum Mov Sci 72:102650. https://doi.org/10.1016/j.humov.2020.102650
Pfefferbaum A, Mathalon DH, Sullivan EV, Rawles JM, Zipursky RB, Lim KO (1994) A quantitative magnetic resonance imaging study of changes in brain morphology from infance to late adulthood. JAMA Neurol 51(9):874–887. https://doi.org/10.1001/archneur.1994.00540210046012
Pitt B, Dotan R, Millar J, Long D, Tokuno C, O’Brien T, Falk B (2015) The electromyographic threshold in boys and men. Eur J Appl Physiol 115(6):1273–1281. https://doi.org/10.1007/s00421-015-3100-5
Radnor JM, Oliver JL, Waugh CM, Myer GD, Lloyd RS (2020) The influence of maturity status on muscle architecture in school-aged boys. Pediatr Exerc Sci 32(2):89–96. https://doi.org/10.1123/pes.2019-0201
Sterczala AJ, Miller JD, Trevino MA, Dimmick HL, Herda TJ (2018) Differences in the motor unit firing rates and amplitudes in relation to recruitment thresholds during submaximal contractions of the first dorsal interosseous between chronically resistance-trained and physically active men. Appl Physiol Nutr Metab 43(8):759–768. https://doi.org/10.1139/apnm-2017-0646
Tanner JM (1962) Growth at adolescence, 2nd edn. Blackwell Scientific Publications, Oxford
Weide G, Huijing PA, Maas JC, Becher JG, Harlaar J, Jaspers RT (2015) Medial gastrocnemius muscle growth during adolescence is mediated by increased fascicle diameter rather than by longitudinal fascicle growth. J Anat 226:530–541. https://doi.org/10.1111/joa.12306
Wernbom M, Aagaard P (2019) Muscle fibre activation and fatigue with low-load blood flow restricted resistance exercise—an integrative physiology review. Acta Physiol 228(1):1–48. https://doi.org/10.1111/apha.13302
Woods S, Dotan R, Jenicek N, Falk B (2020) Isometric-based EMG threshold in girls and women. Eur J Appl Physiol 120(4):907–914. https://doi.org/10.1007/s00421-020-04331-7
Woods S, Dotan R, Jenicek N, Maynard J, Gabriel D, Tokuno C, Falk B (2019) Isometric-based test improves EMG-threshold determination in boys vs men. Eur J Appl Physiol 119(9):1971–1979. https://doi.org/10.1007/s00421-019-04185-8
Woods S, O’Mahoney C, Maynard J, Dotan R, Tenenbaum G, Filho E, Falk B (2022) Increase in volitional muscle activation from childhood to adulthood: a systematic review and meta-analysis. Med Sci Sports Exerc 54(5):789–799. https://doi.org/10.1249/MSS.0000000000002853
Woods S, O’Mahoney C, Mckiel A, Natale L, Falk B (2023) Child-Adult differences in antagonist muscle coactivation: a systematic review. J Electromyogr Kinesiol 26:102727. https://doi.org/10.1016/j.jelekin.2022.102727
Acknowledgements
We would like to thank all the participants for their time, effort and hard work. Additionally, we would like to thank the parents/guardians for making it all happen, even when faced with COVID-related limitations and hardships.
Funding
This work was supported by the Natural Science and Engineering Research Council of Canada (NSERC grants to B. Falk and to M. Holmes). S Woods and A McKiel were supported by Ontario Graduate Scholarship.
Author information
Authors and Affiliations
Contributions
SW, MH, DG and BF conceived and designed research. SW, AM conducted experiments. SW, TH, DG and BF analyzed data and wrote the manuscript. TH and PK commented on previous versions. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Communicated by Toshio Moritani.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Woods, S., McKiel, A., Herda, T. et al. Different discrete motor-unit activation patterns in the flexor carpi radialis in boys and men. Eur J Appl Physiol (2024). https://doi.org/10.1007/s00421-024-05417-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00421-024-05417-2