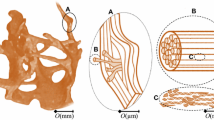
Abstract
As a living tissue, bone is subjected to internal evolutions of its trabecular architecture under normal everyday mechanical loadings leading to damage. The repeating bone remodeling cycle aims at repairing the damaged zones in order to maintain bone structural integrity; this activity of sensing the peak stress at locations where damage or microcracks have occurred, removing old bone and apposing new bone is achieved thanks to a complicated machinery at the cellular level involving specialized cells (osteocytes, osteoclasts, and osteoblasts). This work aims at developing an integrated remodeling-to-fracture model to simulate the bone remodeling process, considering trabecular bone anisotropy. The effective anisotropic continuum mechanical properties of the trabecular bone are derived from an initially discrete planar hexagonal structure representative of femur bone microstructure, relying on the asymptotic homogenization technique. This leads to scaling laws of the effective elastic properties of bone versus effective density at an intermediate mesoscopic scale. An evolution law for the local bone apparent density is formulated in the framework of the thermodynamics of irreversible processes, whereby the driving force for density evolutions is identified as the local strain energy density weighted by the locally accumulated microdamage. We adopt a classical nonlinear damage model for high cycle fatigue under purely elastic strains, where the assumed homogeneous damage is related to the number of cycles bone experiences. Based on this model, we simulate bone remodeling for the chosen initial microstructure, showing the influence of the external mechanical stimuli on the evolution of the density of bone and the incidence of this evolution on trabecular bone effective mechanical properties.











Similar content being viewed by others
Abbreviations
- B :
-
proportionality constant measuring the rate of adaptation process;
- \(B_{R}\) :
-
Set of beams within the reference unit cell;
- C :
-
Elastic damage stiffness tensor;
- \(\mathbf{C}^{0}\) :
-
Virgin (undamaged) elasticity stiffness tensor;
- D :
-
The fatigue damage variable;
- \(E_x ,E_y \) :
-
Homogenized Young’s moduli in x and y directions, respectively;
- \(E_s \) :
-
Young’s modulus of trabecular material;
- \(\mathbf{F}^{b}\) :
-
Resultant of forces at the nodes of a beam b;
- F \(_{e}\) :
-
Accommodation tensor;
- F \(_{g}\) :
-
Irreversible growth tensor;
- \(G_{xy}\) :
-
Homogenized shear modulus;
- \(G_{s}\) :
-
Shear modulus of trabeculae material;
- g:
-
Jacobian associated with the function transformation from Cartesian to curvilinear coordinates;
- \(\hbox {g}_{\mathrm{a}}\) :
-
Gravitational acceleration;
- I :
-
Identity tensor;
- J :
-
Jacobian of the total deformation gradient;
- \(J_{\mathrm{e}}\) :
-
Jacobian of the accommodation tensor;
- \(J_{g }\) :
-
Jacobian of the growth tensor;
- k :
-
Timoshenko shear correction factor;
- \(\mathbf{M}^{n}\) :
-
Bending moment at node n;
- \({N/N_ f} \) :
-
Proportion of fatigue life;
- N :
-
Daily loading cycle number;
- \(N_ f \) :
-
Number of cycles to failure;
- O, E :
-
Origin and end node of a beam, respectively;
- P :
-
Traction vector;
- R :
-
Position vector;
- \(\mathbf{S}^{\mathrm {i}}\) :
-
Stress vector;
- \(\mathbf{u}\left( {\hbox {u}_{\mathrm{x}} ,\hbox {u}_{\mathrm{y}} } \right) \) :
-
Displacement vector;
- v :
-
Test (vector) function;
- v \(^{\mathrm{n}}\) :
-
Virtual velocity field of node n;
- \(\nu _{xy} ,\nu _{yx} \) :
-
Homogenized in-plane Poisson’s ratio;
- \(\mathbf{w}^{n}\) :
-
Virtual rotational velocity of node n;
- \(\alpha ,\beta \) :
-
Material fatigue parameters of bone;
- \(\delta ^{ib}\) :
-
Shift factor for nodes belonging to a neighboring cell;
- \(\phi ^{O\left( b \right) }, \phi ^{E\left( b \right) }\) :
-
The two discrete nodal rotations
- \(\psi \) :
-
Strain energy density;
- \({[}{{\varvec{\upvarepsilon }} }{]}\) :
-
Elastic strain tensor;
- \(\rho _a \) :
-
Apparent density of trabecular bone;
- \(\rho _s \) :
-
Density of trabeculae material;
- \(\Gamma ^{s}\) :
-
Growth rate;
- \({\varvec{\Sigma }}(\hbox {u})\) :
-
Actual volumetric Eshelby stress tensor;
- \({{\varvec{\Sigma }} }_0 \) :
-
Threshold value for the volumetric Eshelby stress stimulus;
- \({{\varvec{\upsigma }} }\left( \mathbf{u} \right) \) :
-
Stress tensor;
- \(\lambda ^{i}\) :
-
Curvilinear Lagrangian coordinates suitable for a general parametrization of any material point
References
Ambrosi, D., Guana, F.: Stress modulated growth. Math. Mech. Solids 12(3), 319–342 (2007)
Aoubiza, B., Crolet, J.M., Meunier, A.: On the mechanical characterization of compact bone structure using the homogenization theory. J. Biomech. 29(12), 1539–1547 (1996)
Arnoux, P.J., Bonnoit, J., Chabrand, P., Jean, M., Pithioux, M.: Numerical damage models using a structural approach: application in bones and ligaments. Eur. Phys. J. Appl. Phys. 17, 65–73 (2002)
Beauprée, G.S., Orr, T.E., Carter, D.R.: An approach for time-dependent bone modeling and remodeling-application: a preliminary remodeling simulation. J. Orthop. Res. 8, 662–670 (1990)
Carter, D.R., Fyhrie, D.P., Whalen, R.T.: Trabecular bone density and loading history: regulation of connective tissue biology by mechanical energy. J. Biomech. XI 20, 785–794 (1987)
Chen, G., Pettet, G., Pearcy, M., McElwain, D.L.S.: Comparison of two numerical approaches for bone remodeling. Med. Eng. Phys. 29, 134–139 (2007)
Cowin, S.C., Van Buskirk, W.C.: Surface bone remodeling induced by a medullary pin. J. Biomech. 12(4), 269–276 (1979)
Doblaré, M., Garcıa, J.M.: Application of an anisotropic bone-remodelling model based on a damage-repair theory to the analysis of the proximal femur before and after total hip replacement. J. Biomech. 34, 1157–1170 (2001)
Fang, Z.: Image-Guided Modeling, Fabrication and Micro-mechanical Analysis of Bone and Heterogeneous Structure. Ph.D. Thesis, Drexel University (2005)
Fritsch, A., Dormieux, L., Hellmich, C.: Porous polycrystals built up by uniformly and axisymmetrically oriented needles: homogenization of elastic properties. C. R. Méc. 334, 151–157 (2006)
Ganghoffer, J.F.: Mechanical modeling of growth considering domain variation—part II: volumetric and surface growth involving Eshelby tensors. J. Mech. Phys. Solids 58(9), 1434–1459 (2010a)
Ganghoffer, J.F.: On Eshelby tensors in the context of the thermodynamics of open systems: application to volumetric growth. Int. J. Eng. Sci. 48(12), 2081–2098 (2010b)
Ganghoffer, J.F.: A contribution to the mechanics and thermodynamics of surface growth. Application to bone external remodeling. Int. J. Eng. Sci. 50, 166–191 (2012)
Gibson, L.J.: Biomechanics of cellular solids. J. Biomech. 38, 377–399 (2005)
Goda, I., Assidi, M., Ganghoffer, J.F.: Equivalent mechanical properties of textile monolayers from discrete asymptotic homogenization. J. Mech. Phys. Solids 61, 2537–2565 (2013)
Goda, I., Assidi, M., Belouettar, S., Ganghoffer, J.F.: A micropolar anisotropic constitutive model of cancellous bone from discrete homogenization. J. Mech. Behav. Biomed. Mater. 16, 87–108 (2012)
Hambli, R.: Numerical procedure for multiscale bone adaptation prediction based on neural networks and finite element simulation. Finite Elem. Anal. Des. 47, 835–842 (2011)
Hazelwood, S.J., Martin, R.B., Rashid, M.M., Rodrigo, J.J.: A mechanistic model for internal bone remodeling exhibits different dynamic responses in disuse and overload. J. Biomech. 34, 299–308 (2001)
Hecht, F., Pironneau, O., Le Hyaric, A., Ohtsuka, K.: “FreeFem++ Manual”, downloadable at http://www.freefem.org (2005)
Hellmich, C., Barthélémy, J.F., Dormieux, L.: Mineral-collagen interactions in elasticity of bone ultrastructure-a continuum micromechanics approach. Eur. J. Mech. A Solid. 23(5), 783–810 (2004)
Hellmich, C., Kober, C., Erdmann, B.: Micromechanics-based conversion of CT data into anisotropic elasticity tensors, applied to FE simulations of a mandible. Ann. Biomed. Eng. 36(1), 108–122 (2008)
Hellmich, C., Ulm, F.J.: Micromechanical model for ultrastructural stiffness of mineralized tissues. J. Eng. Mech. 128(8), 898–908 (2002)
Hernandez, C.J., Beaupre, G.S., Carter, D.R.: A model of mechanobiologic and metabolic influences on bone adaptation. J. Rehabil. Res. Dev. 37(2), 235–244 (2000)
Ju, J.: Damage mechanics of composite materials: constitutive modeling and computational algorithms. Technical report, DTIC document (1991)
Kowalczyk, P.: Simulation of orthotropic microstructure remodelling of cancellous bone. J. Biomech. 43, 563–569 (2010)
Lee, T.C., Noelke, L., McMahon, G.T., Mulville, J.P., Taylor, D.: Functional adaptation in bone. In: Pedersen, P., Bendsoe, M.P. (eds.) Synthesis in Bio Solid Mechanics, pp. 1–10. Kluwer, Dordrecht (1999)
Lemaitre, J.: How to use damage mechanics. Nucl. Eng. Des. 80, 233–245 (1984)
Lemaitre, J., Chaboche, J.L.: Aspect phenomenologique de la rupture par endommagement. J. de Mech. Appl. 2, 317–365 (1978)
Li, J., Li, H., Shi, L., Fok, A.S., Ucer, C., Devlin, H., Horner, K., Silikas, N.: A mathematical model for simulating the bone remodeling process under mechanical stimulus. Dent. Mater. 23, 1073–1078 (2007)
Martin, R.B.: A theory of fatigue damage accumulation and repair in cortical bone. J. Orthop. Res. 10, 818–825 (1992)
McNamara, L.M., Prendergast, P.J.: Bone remodelling algorithms incorporating both strain and microdamage stimuli. J. Biomech. 40, 1381–1391 (2007)
Mori, S., Burr, D.B.: Increased intracortical remodeling following fatigue damage. Bone 14, 103–109 (1993)
Prendergast, J., Huiskes, R.: Mathematical modeling of microdamage in bone remodeling and adaptation In: A. Odgaard, H. Weinans (Eds.), Bone Structure and Remodeling, Recent Advances in Human Biology2, World ScientiGc, Singapore 213–223 (1995)
Prendergast, P.J., Taylor, D.: Prediction of bone adaptation using damage accumulation. J. Biomech. 27, 1067–1076 (1994)
Qin, Y.X., Rubin, C.T., McLeod, K.J.: Nonlinear dependence of loading intensityand cycle number in the maintenance of bone mass and morphology. J. Orthop. Res. 16, 482–489 (1998)
Ramtani, S., Zidi, M.: Damaged-bone adaptation under steady homogeneous stress. J. Biomech. Eng. 124(3), 322–7 (2002)
Tobin, W.J.: The internal architecture of the femur and its clinical significance. J. Bone Joint Surg. A 37, 57–71 (1955)
Taylor, D., Hazenberg, J.G., Lee, T.C.: Living with cracks: damage and repair in human bone. Nat. Mater. 6(4), 263–268 (2007)
Wang, C., Zhang, C., Han, J., Wu, H., Fan, Y.: Simulated evolution of the vertebral body based on basic multicellular unit activities. J. Bone Min. Metab. 29(4), 466–76 (2011)
Watzky, A., Naili, S.: Orthotropic bone remodeling: case of plane stresses. Mech. Res. Commun. 31, 617–625 (2004)
Weinans, H., Huiskes, R., Grootenboer, H.J.: The behavior of adaptive bone remodeling simulation models. J. Biomech. 25, 1425–1441 (1992)
Wolff, J.: Das Gesetz transformation der Knochen. Hirschwald Verlag, Berlin (1892)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Goda, I., Ganghoffer, JF. Modeling of anisotropic remodeling of trabecular bone coupled to fracture. Arch Appl Mech 88, 2101–2121 (2018). https://doi.org/10.1007/s00419-018-1438-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00419-018-1438-y