Abstract
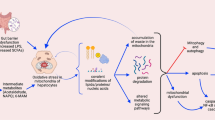
The liver is known to possess remarkable regenerative potential, but persistent inflammation or severe acute injury can lead to liver fibrosis and incomplete regeneration, ultimately resulting in liver failure. Recent studies have shown that the axis of two types of CXCL12 receptors, CXCR4 and CXCR7, plays a crucial role in liver fibrosis and regeneration. The present study aimed to investigate the regulatory factors involved in CXCR4 expression in injured liver. Immunohistochemical screening of liver tissue samples collected during liver transplantation revealed a reciprocal expression pattern between CXCR4 and MeCP2. An in vitro system involving cultured cell lines and H2O2 treatment was established to study the impact of oxidative stress on signaling pathways and epigenetic alterations that affect CXCR4 mRNA expression. Operating through distinct signaling pathways, H2O2 treatment induced a dose-dependent increase in CXCR4 expression in both hepatocyte- and intrahepatic cholangiocyte-derived cells. Treatment of the cells with trichostatin and azacytidine modulated CXCR4 expression in hepatocytes by modifying the methylation status of CpG dinucleotides located in a pair of TA repeats adjacent to the TATA box of the CXCR4 gene promoter. Only MeCP2 bound to oligonucleotides representing the TATA box region when the cytosine residues within the sequence were methylated, as revealed by electrophoretic mobility shift assay (EMSA). Methylation-specific PCR analysis of microdissected samples revealed a correlation between the loss of CpG methylation and the upregulation of CXCR4 in injured hepatocytes, replicating the findings from the in vitro study. Besides the conventional MEK/ERK and NF-κB signaling pathways that activate CXCR4 in intrahepatic cholangiocytes, the unique epigenetic modifications observed in hepatocytes might also contribute to a shift in the CXCR4–CXCR7 balance towards CXCR4, leading to irreversible liver injury and fibrosis. This study highlights the importance of epigenetic modifications in regulating CXCR4 expression in liver injury and fibrosis.








Similar content being viewed by others
Data availability
All data is available upon request.
References
Darwanto A, Kitazawa R, Mori K, Kondo T, Kitazawa S (2008) MeCP2 expression and promoter methylation of cyclin D1 gene are associated with cyclin D1 expression in developing rat epididymal duct. Acta Histochem Cytochem 41(5):135–142. https://doi.org/10.1267/ahc.08025
Dikici I, Mehmetoglu I, Dikici N, Bitirgen M, Kurban S (2005) Investigation of oxidative stress and some antioxidants in patients with acute and chronic viral hepatitis B and the effect of interferon-alpha treatment. Clin Biochem 38(12):1141–1144. https://doi.org/10.1016/j.clinbiochem.2005.10.006
Ding BS, Cao Z, Lis R, Nolan DJ, Guo P, Simons M, Penfold ME, Shido K, Rabbany SY, Rafii S (2014) Divergent angiocrine signals from vascular niche balance liver regeneration and fibrosis. Nature 505(7481):97–102. https://doi.org/10.1038/nature12681
Douzinas EE, Livaditi O, Tasoulis M-K, Prigouris P, Bakos D, Goutas N, Vlachodimitropoulos D, Andrianakis I, Betrosian A, Tsoukalas GD (2012) Nitrosative and oxidative stresses contribute to post-ischemic liver injury following severe hemorrhagic shock: the role of hypoxemic resuscitation. PLoS ONE 7(3):e32968. https://doi.org/10.1371/journal.pone.0032968
Foglia B, Cannito S, Bocca C, Parola M, Novo E (2019) ERK pathway in activated, myofibroblast-like, hepatic stellate cells: a critical signaling crossroad sustaining liver fibrosis. Int J Mol Sci 20(11):2700. https://doi.org/10.3390/ijms20112700
García-Ruiz C, Fernández-Checa JC (2018) Mitochondrial oxidative stress and antioxidants balance in fatty liver disease. Hepatol Commun 2(12):1425–1439. https://doi.org/10.1002/hep4.1271
Haraguchi R, Kohara Y, Matsubayashi K, Kitazawa R, Kitazawa S (2020) New insights into the pathogenesis of diabetic nephropathy: proximal renal tubules are primary target of oxidative stress in diabetic kidney. Acta Histochem Cytochem 53(2):21–31. https://doi.org/10.1267/ahc.20008
Hong F, Tuyama A, Lee TF, Loke J, Agarwal R, Cheng X, Garg A, Fiel MI, Schwartz M, Walewski J, Branch A, Schecter AD, Bansal MB (2009) Hepatic stellate cells express functional CXCR4: role in stromal cell-derived factor-1α-mediated stellate cell activation. Hepatology 49(6):2055–2067. https://doi.org/10.1002/hep.22890
Huang C-Y, Lee C-Y, Chen M-Y, Yang W-H, Chen Y-H, Chang C-H, Hsu H-C, Fong Y-C, Tang C-H (2009) Stromal cell-derived factor-1/CXCR4 enhanced motility of human osteosarcoma cells involves MEK1/2, ERK and NF-κB-dependent pathways. J Cell Physiol 221(1):204–212. https://doi.org/10.1002/jcp.21846
Kang N, Choi SY, Kim BN, Yeo CD, Park CK, Kim YK, Kim TJ, Lee SB, Lee SH, Park JY, Park MS, Yim HW, Kim SJ (2019) Hypoxia-induced cancer stemness acquisition is associated with CXCR4 activation by its aberrant promoter demethylation. BMC Cancer 19(1):148. https://doi.org/10.1186/s12885-019-5360-7
Kisseleva T, Brenner DA (2007) Role of hepatic stellate cells in fibrogenesis and the reversal of fibrosis. J Gastroenterol Hepatol 22(s1):S73–S78. https://doi.org/10.1111/j.1440-1746.2006.04658.x
Kitazawa R, Kitazawa S (2007) Methylation status of a single CpG locus 3 bases upstream of TATA-box of receptor activator of nuclear factor-kappaB ligand (RANKL) gene promoter modulates cell- and tissue-specific RANKL expression and osteoclastogenesis. Mol Endocrinol 21(1):148–158. https://doi.org/10.1210/me.2006-0205
Klose RJ, Sarraf SA, Schmiedeberg L, Mcdermott SM, Stancheva I, Bird AP (2005) DNA binding selectivity of MeCP2 due to a requirement for A/T sequences adjacent to methyl-CpG. Mol Cell 19(5):667–678. https://doi.org/10.1016/j.molcel.2005.07.021
Kubarek Ł, Jagodzinski PP (2007) Epigenetic up-regulation of CXCR4 and CXCL12 expression by 17 β-estradiol and tamoxifen is associated with formation of DNA methyltransferase 3B4 splice variant in Ishikawa endometrial adenocarcinoma cells. FEBS Lett 581(7):1441–1448. https://doi.org/10.1016/j.febslet.2007.02.070
Lozano-Sepulveda SA (2015) Oxidative stress modulation in hepatitis C virus infected cells. World J Hepatol 7(29):2880. https://doi.org/10.4254/wjh.v7.i29.2880
Maroni P, Bendinelli P, Matteucci E, Desiderio MA (2007) HGF induces CXCR4 and CXCL12-mediated tumor invasion through Ets1 and NF-kappaB. Carcinogenesis 28(2):267–279. https://doi.org/10.1093/carcin/bgl129
Miyaoka Y, Ebato K, Kato H, Arakawa S, Shimizu S, Miyajima A (2012) Hypertrophy and unconventional cell division of hepatocytes underlie liver regeneration. Curr Biol 22(13):1166–1175. https://doi.org/10.1016/j.cub.2012.05.016
Mo H, Wu Q, Miao J, Luo C, Hong X, Wang Y, Tang L, Hou FF, Liu Y, Zhou L (2017) C-X-C chemokine receptor type 4 plays a crucial role in mediating oxidative stress-induced podocyte injury. Antioxid Redox Signal 27(6):345–362. https://doi.org/10.1089/ars.2016.6758
Mori K, Kitazawa R, Kondo T, Mori M, Hamada Y, Nishida M, Minami Y, Haraguchi R, Takahashi Y, Kitazawa S (2014) Diabetic osteopenia by decreased β-catenin signaling is partly induced by epigenetic derepression of sFRP-4 gene. PLoS ONE 9(7):e102797. https://doi.org/10.1371/journal.pone.0102797
Nakagawa M, Kitazawa R, Kuwahara N, Yoshida K, Haraguchi R, Kitazawa S (2013) Efficient genetic analysis of microdissected samples by agarose-bead method: alterations of β-catenin gene in fundic gland polyp and heterotopic gastric mucosa of duodenum. Acta Histochem Cytochem 46(1):19–24. https://doi.org/10.1267/ahc.12022
Nieto N (2006) Oxidative-stress and IL-6 mediate the fibrogenic effects of rodent Kupffer cells on stellate cells. Hepatology 44(6):1487–1501. https://doi.org/10.1002/hep.21427
Nishikawa Y, Tokusashi Y, Kadohama T, Nishimori H, Ogawa K (1996) Hepatocytic cells form bile duct-like structures within a three-dimensional collagen gel matrix. Exp Cell Res 223(2):357–371. https://doi.org/10.1006/excr.1996.0091
Nishikawa Y, Sone M, Nagahama Y, Kumagai E, Doi Y, Omori Y, Yoshioka T, Tokairin T, Yoshida M, Yamamoto Y, Ito A, Sugiyama T, Enomoto K (2013) Tumor necrosis factor-α promotes bile ductular transdifferentiation of mature rat hepatocytes in vitro. J Cell Biochem 114(4):831–843. https://doi.org/10.1002/jcb.24424
Qin D, Liu S, Lu Y, Yan Y, Zhang J, Cao S, Chen M, Chen N, Huang W, Wang L, Chen X, Zhang L (2022) Lgr5+ cell fate regulation by coordination of metabolic nuclear receptors during liver repair. Theranostics 12(14):6130–6142. https://doi.org/10.7150/thno.74194
Rouach RNaCRaH, Nordmann R, Ribière C, Rouach H (1992) Implication of free radical mechanisms in ethanol-induced cellular injury. Free Radic Biol Med 12(3):219–240
Tan HK, Yates E, Lilly K, Dhanda AD (2020) Oxidative stress in alcohol-related liver disease. World J Hepatol 12(7):332–349. https://doi.org/10.4254/wjh.v12.i7.332
Tsuchida T, Friedman SL (2017) Mechanisms of hepatic stellate cell activation. Nat Rev Gastroenterol Hepatol 14(7):397–411. https://doi.org/10.1038/nrgastro.2017.38
Villanueva-Paz M, Morán L, López-Alcántara N, Freixo C, Andrade RJ, Lucena MI, Cubero FJ (2021) Oxidative stress in drug-induced liver injury (DILI): from mechanisms to biomarkers for use in clinical practice. Antioxidants 10(3):390. https://doi.org/10.3390/antiox10030390
Yeung Y, Lau DK, Chionh F, Tran H, Tse JWT, Weickhardt AJ, Nikfarjam M, Scott AM, Tebbutt NC, Mariadason JM (2017) K-Ras mutation and amplification status is predictive of resistance and high basal pAKT is predictive of sensitivity to everolimus in biliary tract cancer cell lines. Mol Oncol 11(9):1130–1142. https://doi.org/10.1002/1878-0261.12078
Acknowledgements
We express our appreciation to Mss. Yuki Takaoka and Mariko Hashimoto for their valuable assistance.
Funding
This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan (20H03458 and 21K19354 to S. K.).
Author information
Authors and Affiliations
Contributions
SK participated in the experimental design and managed whole project. CI participated in all experiments. RH participated in MSP assays. RK participated in cell culture studies. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This study was approved by the ethics committee of Ehime University of Medical School (approval no. 2307003).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ito, C., Haraguchi, R., Ogawa, K. et al. Demethylation in promoter region of severely damaged hepatocytes enhances chemokine receptor CXCR4 gene expression. Histochem Cell Biol 160, 407–418 (2023). https://doi.org/10.1007/s00418-023-02229-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-023-02229-x