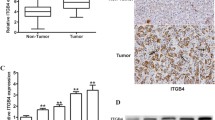
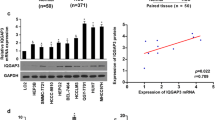
Abstract
In hepatocellular carcinomas (HCCs), the role of the cell surface protein V-set and immunoglobulin domain containing 1 (VSIG1), which is known as a specific marker of the gastric mucosa and testis, has not yet been determined. We examined VSIG1 immunohistochemical (IHC) expression in 105 consecutive samples provided by HCC patients, along with the IHC expression of three of the biomarkers known to be involved in the epithelial–mesenchymal transition (EMT): vimentin (VIM), and E- and N-cadherin (encoded by CDH1 and CDH2 genes). IHC subcellular localization of thyroid transcription factor 1 (TTF1), in which nuclear-to-cytoplasmic translocation is known to cause a lineage shift from lung to gastric-type adenocarcinoma, was also checked. The obtained data were validated using the miRNET program. In the examined HCC samples, VSIG1 expression was observed in the cytoplasm of normal hepatocytes and downregulated in 47 of the 105 HCCs (44.76%). In 29 cases (27.62%), VSIG1 was co-expressed with cytoplasmic TTF1. VSIG1 expression was positively correlated with both E-cadherin and N-cadherin and negatively correlated with VIM (p < 0.0001). The VSIG1+/E-cadherin+/N-cadherin-/VIM phenotype was seen in 13 cases (12.4%) and was characteristic of well-differentiated (G1/2) carcinomas diagnosed in pT1/2 stages. Like pulmonary carcinomas, simultaneous cytoplasmic positivity of HCC cells for VSIG1 and TTF1 may be a potential indicator of a lineage shift from conventional to gastric-type HCC. The E-cadherin/VSIG1 complex can help suppress tumor growth by limiting HCC dedifferentiation. The miRNET-based interaction between VSIG1/VIM/CDH1/CDH2 genes might be interconnected by miR-200b-3p, a central regulator of EMT which also targets VIM and VSIG1.




Similar content being viewed by others
Availability of data and material
Data are available upon request.
References
Amin MB, Edge S, Greene F, Byrd DR, Brookland RK, Washington MK et al (2017) AJCC Cancer Staging Manual, 8th edn. Springer International Publishing, New York
Bara T Jr, Gurzu S, Sugimura H, Bara T, Beleaua MA, Jung I (2017) A systematic review of the possible carcinogenic role of the aristolochic acid. Rom J Morphol Embryol 58:41–44
Bernal C, Silvano M, Tapponnier Y, Anand S, Angulo C, Altaba RA (2020) Functional pro-metastatic heterogeneity revealed by spiked-scRNAseq is shaped by cancer cell interactions and restricted by VSIG1. Cell Rep 33:108372. https://doi.org/10.1016/j.celrep.2020.108372
Braicu C, Burz C, Berindan-Neagoe I, Balacescu O, Graur F, Cristea V, Irimie A (2009) Hepatocellular carcinoma: tumorigenesis and prediction markers. Gastroenterol Res 2:191–199. https://doi.org/10.4021/gr2009.07.1304
Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Rodriguez IP, Chakravarthi BVSK, Varambally S (2017) UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia 19:649–658. https://doi.org/10.1016/j.neo.2017.05.002
Chao WT, Liu CH, Lai CR, Chen YJ, Chuang CM, Wang PH (2018) Alpha-fetoprotein-producing ovarian clear cell adenocarcinoma with fetal gut differentiation: a rare case report and literature review. J Ovarian Res 11:52. https://doi.org/10.1186/s13048-018-0426-8
Chen Y, Pan K, Li S, Xia J, Wang W, Chen J, Zhao J, Lu L, Wang D, Pan Q, Wang Q, Li Y, He J, Li Q (2012) Decreased expression of V-set and immunoglobulin domain containing 1 (VSIG1) is associated with poor prognosis in primary gastric cancer. J Surg Oncol 106:286–293. https://doi.org/10.1002/jso.22150
Costa RH, Kalinichenko VV, Lim L (2001) Transcription factors in mouse lung development and function. Am J Physiol Lung Cell Mol Physiol 280:L823–L838. https://doi.org/10.1152/ajplung.2001.280.5.L823
Delker DA, McGettigan BM, Kanth P, Pop S, Neklason DW, Bronner MP, Burt RW, Hagedorn CH (2014) RNA sequencing of sessile serrated colon polyps identifies differentially expressed genes and immunohistochemical markers. PLoS ONE 9:e88367. https://doi.org/10.1371/journal.pone.0088367
Denker BM, Nigam SK (1998) Molecular structure and assembly of the tight junction. Am J Physiol 274:F1–F9. https://doi.org/10.1152/ajprenal.1998.274.1.F1
Fan Y, Siklenka K, Arora SK, Ribeiro P, Kimmins S, Xia J (2016) miRNet - dissecting miRNA-target interactions and functional associations through network-based visual analysis. Nucleic Acids Res 44:W135-141. https://doi.org/10.1093/nar/gkw288
Gherman C, Braicu OL, Zanoaga O, Jurj A, Pileczki V, Maralani M, Drigla F, Braicu C, Budisan L, Achimas-Cadariu P, Berindan-Neagoe I (2016) Caffeic acid phenethyl ester activates pro-apoptotic and epithelial–mesenchymal transition-related genes in ovarian cancer cells A2780 and A2780cis. Mol Cell Biochem 413:189–198. https://doi.org/10.1007/s11010-015-2652-3
Gurzu S, Banias L, Bara T, Feher I, Bara T, Jung I (2018) The epithelial–mesenchymal transition pathway in two cases with gastric metastasis originating from breast carcinoma, one with a metachronous primary gastric cancer. Recent Pat Anticancer Drug Discov 13:118–124. https://doi.org/10.2174/2212798409666171101121108
Gurzu S, Ciortea D, Ember I, Jung I (2013) The possible role of Mena protein and its splicing-derived variants in embryogenesis, carcinogenesis, and tumor invasion: a systematic review of the literature. Biomed Res Int 2013:365192. https://doi.org/10.1155/2013/365192
Gurzu S, Kobori L, Fodor D, Jung I (2019) Epithelial mesenchymal and endothelial mesenchymal transitions in hepatocellular carcinoma: a review. BioMed Res Int 2019:2962580. https://doi.org/10.1155/2019/2962580
Inoue Y, Matsuura S, Yoshimura K, Iwashita Y, Kahyo T, Kawase A, Tanahashi M, Maeda M, Ogawa H, Inui N, Funai K, Shinmura K, Niwa H, Suda T, Sugimura H (2017) Characterization of V-set and immunoglobulin domain containing 1 exerting a tumor suppressor function in gastric, lung, and esophageal cancer cells. Cancer Sci 108:1701–1714. https://doi.org/10.1111/cas.13295
Kim E, Lee Y, Kim JS, Song BS, Kim SU, Huh JW, Lee SR, Kim SH, Hong Y, Chang KT (2010) Extracellular domain of V-set and immunoglobulin domain containing 1 (VSIG1) interacts with sertoli cell membrane protein, while its PDZ-binding motif forms a complex with ZO-1. Mol Cells 30:443–448. https://doi.org/10.1007/s10059-010-0138-4
Kim JH, Kim KJ, Rhee YY, Bae JM, Cho NY, Lee HS, Kang GH (2015) Gastric-type expression signature in serrated pathway-associated colorectal tumors. Hum Pathol 46:643–656. https://doi.org/10.1016/j.humpath.2015.01.003
Kovecsi A, Gurzu S, Szentirmay Z, Kovacs Z, Bara TJ, Jung I (2017) Paradoxical expression pattern of the epithelial mesenchymal transition-related biomarkers CD44, SLUG, N-cadherin and VSIG1/Glycoprotein A34 in gastrointestinal stromal tumors. World J Gastrointest Oncol 9:436–443. https://doi.org/10.4251/wjgo.v9.i11.436
Marra G (2021) An “expressionistic” look at serrated precancerous colorectal lesions. Diagn Pathol 16:4. https://doi.org/10.1186/s13000-020-01064-1
Meguro S, Yasuda M (2013) α-Fetoprotein-producing ovarian tumor in a postmenopausal woman with germ cell differentiation. Ann Diagn Pathol 17:140–144. https://doi.org/10.1016/j.anndiagpath.2011.07.010
Oidovsambuu O, Nyamsuren G, Liu S, Göring W, Engel W, Adham IM (2011) Adhesion protein VSIG1 is required for the proper differentiation of glandular gastric epithelia. PLoS ONE 6:e25908. https://doi.org/10.1371/journal.pone.0025908
Pasricha S, Grover S, Kamboj M, Bansal D, Batra U, Gupta G, Sharma A, Durga G, Jajodia A, Koyyala VP, Mehta A (2021) Hepatoid adenocarcinoma of lung: a diagnostic challenge—series of six cases with histopathological, predictive molecular and PD-L1 assessment. Indian J Pathol Microbiol 64:128–131. https://doi.org/10.4103/IJPM.IJPM_334_20
Sata Y, Nakajima T, Fukuyo M, Matsusaka K, Hata A, Morimoto J, Rahmutulla B, Ito Y, Suzuki H, Yoshino I, Kaneda A (2020) High expression of CXCL14 is a biomarker of lung adenocarcinoma with micropapillary pattern. Cancer Sci 111:2588–2597. https://doi.org/10.1111/cas.14456
Satala CB, Jung I, Kobori L, Kovacs Z, Fodor D, Szodorai R, Gurzu S (2020) Benefits of the 8th American joint committee on cancer system for hepatocellular carcinoma staging. J Gastrointest Cancer. https://doi.org/10.1007/s12029-020-00394-z
Scanlan MJ, Ritter G, Yin BWT, Williams C Jr, Cohen LS, Coplan KA, Fortunato SR, Frosina D, Lee SY, Murray AE, Chua R, Filonenko VV, Sato E, Old LJ, Jungbluth AA (2006) Glycoprotein A34, a novel target for antibody-based cancer immunotherapy. Cancer Immun 6:2
Snyder EL, Watanabe H, Magendantz M, Hoersch S, Chen TA, Wang DG, Crowley D, Whittaker CA, Meyerson M, Kimura S, Jacks T (2013) Nkx2-1 represses a latent gastric differentiation program in lung adenocarcinoma. Mol Cell 50:185–199. https://doi.org/10.1016/j.molcel.2013.02.018
Straub BK, Rickelt S, Zimbelmann R, Grund C, Kuhn C, Iken M, Ott M, Schirmacher P, Franke WW (2011) E–N-cadherin heterodimers define novel adherens junctions connecting endoderm-derived cells. J Cell Biol 195:873–887. https://doi.org/10.1083/jcb.201106023
Tong H, Liu X, Li T, Qiu W, Peng C, Shen B, Zhu Z (2020) NR1D2 accelerates hepatocellular carcinoma progression by driving the epithelial-to-mesenchymal transition. Onco Targets Ther 13:3931–3942. https://doi.org/10.2147/OTT.S237804
Turdean S, Gurzu S, Turcu M, Voidazan S, Sin A (2012) Current data in clinicopathological characteristics of primary hepatic tumors. Rom J Morphol Embryol 53:719–724
Wichelmann TA, Patel K, Malas A, James E (2020) A unique case of Alpha-Fetoprotein-negative hepatoid adenocarcinoma of the stomach with associated signet ring cell histological features and linitis plastica. Cureus 12:e11908. https://doi.org/10.7759/cureus.11908
Yang K, Jiang H, Li Q (2019) Primary pulmonary hepatoid adenocarcinoma: a case report and review of the literature. Medicine 98:e15053. https://doi.org/10.1097/MD.0000000000015053
Zeremski V, Mawrin C, Fischer T, Schalk E (2017) Diagnostic and therapeutic challenges in extragonadal yolk sac tumor with hepatoid differentiation: a case report. Mol Clin Oncol 6:79–82. https://doi.org/10.3892/mco.2016.1080
Zou M, Li Y, Dai Y, Sun L, Huang T, Yuan X, Qiu H (2019) AFP-producing hepatoid adenocarcinoma (HAC) of peritoneum and omentum: a case report and literature review. Onco Targets Ther 12:7649–7654. https://doi.org/10.2147/OTT.S216501
Acknowledgements
We thank the colleagues from the Department of Pathology of Semmelweis University for allowing us to use the paraffin blocks from transplanted cases. The English proofreading was done by Cambridge Proofreading LLC.
Funding
Japanese MEXT S01, JSPS KAKENHI Grant Numbers JP22659072, JP24659161, JP26670187, JP16K15256, AMED Grant Numbers JP19ck0106264, JP20ck0106545, and Smoking Research Foundation.
Author information
Authors and Affiliations
Contributions
GS designed the study, participated in the immunoassay and statistical database interpretation, and wrote the manuscript; SH revised the paper and gave scientific approval; SJ performed the statistical assessment and design of the survival curves; SR contributed to data collection and literature review; BC performed the bioinformatic analysis and checked the UALCAN online database and gene network; KL supervised patient follow-up and performed liver transplantation; FD participated in the collection and interpretation of clinical data; JI contributed to interpretation of histopathological and immunohistochemical data, supervised the study design, and approved the final version of the manuscript. The two authors (GS and SJ) contributed equally to the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The present study was conducted after approval obtained from the ethics committees of the George Emil Palade University of Medicine, Pharmacy, Sciences and Technology located in Targu Mures, Romania, and of Semmelweis University in Budapest, Hungary.
Consent to participate
This is a retrospective observational study.
Consent for publication
All authors give their consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

418_2021_2006_MOESM1_ESM.jpg
Supplementary file 1: Figure S1. The expression level of the main EMT genes in HCC, generated using the UALCAN online program.

418_2021_2006_MOESM2_ESM.jpg
Supplementary file 2: Figure S2. Overall survival subgroup analysis of key EMT signaling genes for HCC patients, according to high or low expression level, generated using UALCAN.
Rights and permissions
About this article
Cite this article
Gurzu, S., Sugimura, H., Szederjesi, J. et al. Interaction between cadherins, vimentin, and V-set and immunoglobulin domain containing 1 in gastric-type hepatocellular carcinoma. Histochem Cell Biol 156, 377–390 (2021). https://doi.org/10.1007/s00418-021-02006-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-021-02006-8