Abstract
Background
Severe asthma, characterized by inflammation and airway remodeling, involves fibroblast differentiation into myofibroblasts expressing α-SMA. This process leads to the production of fibronectin and connective tissue growth factor (CTGF), driven by factors such as transforming growth factor (TGF)-β. Furthermore, the persistent presence of myofibroblasts is associated with resistance to apoptosis and mitochondrial dysfunction. The chemokine (C-X3-C motif) ligand 1 (CX3CL1) plays a role in tissue fibrosis. However, it is currently unknown whether neutralization of CX3CL1 decreases TGF-β-induced fibroblast differentiation and mitochondrial dysfunction in normal human lung fibroblasts (NHLFs).
Methods
CX3CL1/C-X3-C motif chemokine receptor 1 (CX3CR1), CX3CL1 was analyzed by immunofluorescence (IF) or immunohistochemical (IHC) staining of ovalbumin-challenged mice. CX3CL1 release was detected by ELISA. TGF-β-induced CTGF, fibronectin, and α-SMA expression were evaluated in NHLFs following neutralization of CX3CL1 (TP213) treatment for the indicated times by Western blotting or IF staining. Mitochondrion function was detected by a JC-1 assay and seahorse assay. Cell apoptosis was observed by a terminal uridine nick-end labeling (TUNEL) assay.
Results
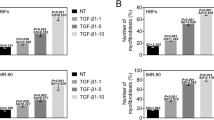
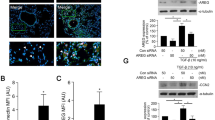
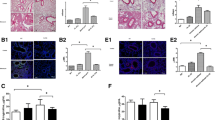
An increase in CX3CL1 expression was observed in lung tissues from mice with ovalbumin-induced asthma by IF staining. CX3CR1 was increased in the subepithelial layer of the airway by IHC staining. Moreover, CX3CR1 small interfering (si)RNA downregulated TGF-β-induced CTGF and fibronectin expression in NHLFs. CX3CL1 induced CTGF and fibronectin expression in NHLFs. TGF-β-induced CX3CL1 secretion from NHLFs. Furthermore, TP213 decreased TGF-β-induced CTGF, fibronectin, and α-SMA expression in NHLFs. Mitochondrion-related differentially expressed genes (DEGs) were examined after CX3CL1 neutralization in TGF-β-treated NHLFs. TP213 alleviated TGF-β-induced mitochondrial dysfunction and apoptosis resistance in NHLFs. CX3CL1 induced p65, IκBα, and IKKα phosphorylation in a time-dependent manner. Furthermore, CX3CL1-induced fibronectin expression and JC-1 monomer were decreased by p65 siRNA. TP213 reduced TGF-β-induced p65 and α-SMA expression in NHLFs.
Conclusions
These findings suggest that neutralizing CX3CL1 attenuates lung fibroblast activation and mitochondrial dysfunction. Understanding the impacts of CX3CL1 neutralization on fibroblast mitochondrial function could contribute to the development of therapeutic strategies for managing airway remodeling in severe asthma.








Similar content being viewed by others
Availability of Data and Materials
Not applicable.
Abbreviations
- ADAM17:
-
A disintegrin and metalloproteinase 17
- α-SMA:
-
α-Smooth muscle actin
- BSA:
-
Bovine serum albumin
- CTGF:
-
Connective tissue growth factor
- CX3CL1:
-
(C-X3-C motif) ligand 1
- CX3CR1:
-
C-X3-C motif chemokine receptor 1
- DEG:
-
Differentially expressed gene
- DMEM:
-
Dulbecco’s modified minimal essential/Eagle medium
- ECM:
-
Extracellular matrix
- EGFR:
-
Epidermal growth factor receptor
- ELISA:
-
Enzyme-linked immunosorbent assay
- EMT:
-
Epithelial-mesenchymal transition
- ETC:
-
Electron transport chain
- FBS:
-
Fetal bovine serum
- HRP:
-
Horseradish peroxidase
- IF:
-
Immunofluorescence
- IHC:
-
Immunohistochemical
- IgG:
-
Immunoglobulin G
- IL:
-
Interleukin
- mAb:
-
Monoclonal antibody
- NES:
-
Normalized enrichment score
- OVA:
-
Ovalbumin
- NEAAs:
-
Nonessential amino acids
- NF-κB:
-
Nuclear factor-κB
- NHLF:
-
Normal human lung fibroblast
- phospho:
-
Phosphorylated
- OXPHOS:
-
Oxidative phosphorylation
- PBS:
-
Phosphate-buffered saline
- PCR:
-
Polymerase chain reaction
- PDGF:
-
Platelet-derived growth factor
- PVDF:
-
Polyvinylidene difluoride
- SDS-PAGE:
-
Sodium dodecylsulfate polyacrylamide gel electrophoresis
- siRNA:
-
Small interfering RNA
- TGF:
-
Transforming growth factor
- TH2:
-
T helper 2
- TUNEL:
-
Terminal uridine nick-end labeling
References
Vos T, Lim SS, Abbafati C et al (2020) Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1204–1222
Hough KP, Curtiss ML, Blain TJ et al (2020) Airway remodeling in asthma. Front Med 7:191
Hinz B, Lagares D (2020) Evasion of apoptosis by myofibroblasts: a hallmark of fibrotic diseases. Nat Rev Rheumatol 16:11–31
Bueno M, Calyeca J, Rojas M, Mora AL (2020) Mitochondria dysfunction and metabolic reprogramming as drivers of idiopathic pulmonary fibrosis. Redox Biol 33:101509
Lee M, Lee Y, Song J et al (2018) Tissue-specific role of CX3CR1 expressing immune cells and their relationships with human disease. Immune Netw 18:e5
Cormican S, Griffin MD (2021) Fractalkine (CX3CL1) and its receptor CX3CR1: a promising therapeutic target in chronic kidney disease? Front Immunol 12:664202
Zheng C, Xuan W, Chen Z et al (2022) CX3CL1 worsens cardiorenal dysfunction and serves as a therapeutic target of canagliflozin for cardiorenal syndrome. Front Pharmacol 13:848310
Dong J, Ma Q (2019) In vivo activation and pro-fibrotic function of NF-κB in fibroblastic cells during pulmonary inflammation and fibrosis induced by carbon nanotubes. Front Pharmacol 10:1140
Wright JG, Christman JW (2003) The role of nuclear factor kappa B in the pathogenesis of pulmonary diseases: implications for therapy. Am J Respir Med 2:211–219
Edwards MR, Bartlett NW, Clarke D et al (2009) Targeting the NF-κB pathway in asthma and chronic obstructive pulmonary disease. Pharmacol Ther 121:1–13
Korbecki J, Simińska D, Kojder K et al (2020) Fractalkine/CX3CL1 in neoplastic processes. Int J Mol Sci 21:3723
Reddy PH (2011) Mitochondrial dysfunction and oxidative stress in asthma: implications for mitochondria-targeted antioxidant therapeutics. Pharmaceuticals 4:429–456
Qian L, Mehrabi Nasab E, Athari SM, Athari SS (2022) Mitochondria signaling pathways in allergic asthma. J Investig Med 70:863–882
Huang M, Nasab EM, Athari SS (2021) Immunoregulatory effect of mesenchymal stem cell via mitochondria signaling pathways in allergic asthma. Saudi J Biol Sci 28:6957–6962
Liu R-M, Desai LP (2015) Reciprocal regulation of TGF-β and reactive oxygen species: a perverse cycle for fibrosis. Redox Biol 6:565–577
Li X, Zhang W, Cao Q et al (2020) Mitochondrial dysfunction in fibrotic diseases. Cell Death Discov 6:80
Chen J-Y, Cheng W-H, Lee K-Y et al (2021) Abnormal ADAM17 expression causes airway fibrosis in chronic obstructive asthma. Biomed Pharmacother 140:111701
Cheng W-H, Lee K-Y, Yu M-C et al (2021) Pref-1 induced lung fibroblast differentiation by hypoxia through integrin α5β1/ERK/AP-1 cascade. Eur J Pharmacol 909:174385
Feng K, Meng P, Zou X et al (2022) IL-37 protects against airway remodeling by reversing bronchial epithelial–mesenchymal transition via IL-24 signaling pathway in chronic asthma. Respir Res 23:1–17
Mionnet C, Buatois V, Kanda A et al (2010) CX3CR1 is required for airway inflammation by promoting T helper cell survival and maintenance in inflamed lung. Nat Med 16:1305–1312
Julia V (2012) CX3CL1 in allergic diseases: not just a chemotactic molecule. Allergy 67:1106–1110
Rimaniol A-C, Till SJ, Garcia G et al (2003) The CX3C chemokine fractalkine in allergic asthma and rhinitis. J Allergy Clin Immunol 112:1139–1146
Green G, Johnson SM, Costello H et al (2021) CX3CR1 is a receptor for human respiratory syncytial virus in cotton rats. J Virol 95:10–1128
Cunoosamy D, Zhou X-H, Rehnberg M et al (2014) Expression of CX3CL1 and CX3CR1 in human asthmatic lung in relation to cell survival. Eur Respir J 44:3863
Jones BA, Beamer M, Ahmed S (2010) Fractalkine/CX3CL1: a potential new target for inflammatory diseases. Mol Interv 10:263
Moss ML, Minond D (2017) Recent advances in ADAM17 research: a promising target for cancer and inflammation. Mediators Inflamm 2017:9673537
Ishida Y, Kimura A, Nosaka M et al (2017) Essential involvement of the CX3CL1-CX3CR1 axis in bleomycin-induced pulmonary fibrosis via regulation of fibrocyte and M2 macrophage migration. Sci Rep 7:1–12
Guo R, Gu J, Zong S et al (2018) Structure and mechanism of mitochondrial electron transport chain. Biomed J 41:9–20
Sebag SC, Koval OM, Paschke JD et al (2017) Mitochondrial CaMKII inhibition in airway epithelium protects against allergic asthma. JCI Insight 2:e88297
Zhang Y, Do DC, Hu X et al (2021) CaMKII oxidation regulates cockroach allergen-induced mitophagy in asthma. J Allergy Clin Immunol 147:1464–1477
Susin SA, Lorenzo HK, Zamzami N et al (1999) Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397:441–446
Beere HM (2005) Death versus survival: functional interaction between the apoptotic and stress-inducible heat shock protein pathways. J Clin Investig 115:2633–2639
Drakopanagiotakis F, Xifteri A, Polychronopoulos V, Bouros D (2008) Apoptosis in lung injury and fibrosis. Eur Respir J 32:1631–1638
Albensi BC (2019) What is nuclear factor kappa B (NF-κB) doing in and to the mitochondrion? Front Cell Dev Biol 7:154
Galán-Ganga M, García-Yagüe ÁJ, Lastres-Becker I (2019) Role of MSK1 in the induction of NF-κB by the chemokine CX3CL1 in microglial cells. Cell Mol Neurobiol 39:331–340
Mizutani S, Nishio J, Kondo K et al (2021) Treatment with an anti-Cx3cl1 antibody suppresses M1 macrophage infiltration in interstitial lung disease in Skg mice. Pharmaceuticals 14:474
Acknowledgements
This study was supported by grant (NSTC112-2320-B-038-001 and NSTC112-2320-B-038-022) from the National Science and Technology Council of Taiwan, R.O.C. DP2-TMU-112-T-05 from the Ministry of Education (MOE) in Taiwan, R.O.C. TMU112-AE1-B10 from the Taipei Medical University (TMU), Taiwan, R.O.C.
Funding
NSTC112-2320-B-038-001 and NSTC112-2320-B-038-022 from the National Science and Technology Council of Taiwan, R.O.C. DP2-TMU-112-T-05 from Ministry of Education (MOE) in Taiwan, R.O.C. TMU112-AE1-B10 from the Taipei Medical University (TMU), Taiwan, R.O.C.
Author information
Authors and Affiliations
Contributions
Conceptualization: B-CC. Data curation: W-HC and P-LC. Formal analysis: W-HC and FSY. Funding acquisition: B-CC. Investigation: W-HC, S-AW, F-LH, M-MN, and P-LC. Methodology: B-CC, Y-CW, S-AW and C-LC. Project administration: B-CC. Resources: B-CC, Y-CW. Software: W-HC and B-CC. Supervision: B-CC and C-HL. Validation: W-HC, P-LC, L-YL, F-SY, and B-CC. Visualization: W-HC, F-SY, L-YL, C-HL, and B-CC. Writing—original draft: W-HC. Writing—review and editing: B-CC, W-HC, C-LC and C-HL.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics Approval and Consent to Participate
All animal protocols were approved by the Animal Ethics Committee of Taipei Medical University (approval No. LAC-2019-0042).
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheng, WH., Chang, PL., Wu, YC. et al. Neutralization of CX3CL1 Attenuates TGF-β-Induced Fibroblast Differentiation Through NF-κB Activation and Mitochondrial Dysfunction in Airway Fibrosis. Lung 202, 343–356 (2024). https://doi.org/10.1007/s00408-024-00701-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-024-00701-6