Abstract
Purpose
This study was conducted to compare serum xenopsin-related peptide-1 (XP-1) levels in women with polycystic ovary syndrome (PCOS) and in healthy women and to determine the role of XP-1 levels in PCOS.
Methods
Forty patients with PCOS and 38 healthy women were included in the study and matched with age and body mass index. Fasting blood glucose, insulin, high sensitivity C-reactive protein (hs-CRP), XP-1 and total testosterone levels of all participants were measured.
Results
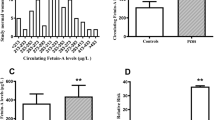
Serum XP-1 levels significantly increased in women with PCOS compared to the control group (6.49 ± 1.57 vs 5.29 ± 1.45 ng/ml, p = 0.001). Serum insulin, hs-CRP, HOMA-IR, total testosterone levels and waist circumference were higher in women with PCOS than in control group. High XP-1 levels were associated with PCOS after adjustment for potential confounders. Receiver operating characteristic (ROC) curve analysis confirmed that the area under ROC curves was 0.703 (95% CI 0.588–0.818, p < 0.002) for XP-1 levels. The optimal cut-off value of XP-1 for detecting PCOS was ≥5.87 ng/ml.
Conclusions
Our results indicate that increased XP-1 levels were associated with PCOS after adjustment for potential confounders, which has been shown to be effective in the function of the insulin signaling pathway.

Similar content being viewed by others
References
Norman RJ, Dewailly D, Legro RS, Hickey TE (2007) Polycystic ovary syndrome. Lancet 370:685–697
Teede H, Deeks A, Moran L (2010) Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med 8:41
Meyer C, McGrath BP, Cameron J, Kotsopoulos D, Teede HJ (2005) Vascular dysfunction and metabolic parameters in polycystic ovary syndrome. J Clin Endocrinol Metab 90:4630–4635
Tarkun I, Arslan BC, Canturk Z, Türemen E, Sahin T, Duman C (2004) Endothelial dysfunction in young women with polycystic ovary syndrome: relationship with insulin resistance and low-grade chronic inflammation. J Clin Endocrinol Metab 89:5592–5596
Carmina E, Lobo RA (1999) Polycystic ovary syndrome (PCOS): arguably the most common endocrinopathy is associated with significant morbidity in women. J Clin Endocrinol Metab 84:1897–1899
Vrbıkova J, Grimmichova T, Dvorakova K, Hill M, Stanická S, Vondra K (2008) Family history of diabetes mellitus determines insulin sensitivity and beta cell function in polycystic ovary syndrome. Physiol Res 57:547–553
Venkatesan AM, Dunaif A, Corbould A (2001) Insulin resistance in polycystic ovary syndrome: progress and paradoxes. Recent Prog Horm Res 56:295–308
Norman RJ, Masters L, Milner CR, Wang JX, Davies MJ (2001) Relative risk of conversion from normoglycaemia to impaired glucose tolerance or non-insulin dependent diabetes mellitus in polycystic ovarian syndrome. Hum Reprod 16(9):1995–1998
Araki K, Tachibana S, Uchiyama M, Nakajima T, Yasuhara T (1975) Isolation and structure of a new active peptide xenopsin on rat stomach strip and some biogenic amines in the skin of Xenopus laevis. Chem Pharm Bull 23:3132–3140
Araki K, Tachibana S, Kato Y, Tajima T (1979) Comparative studies of xenopsin and neurotensin on some biological activities. Yakugaku Zasshi 99:466–470
Arslan N, Sayin O, Tokgöz Y (2014) Evaluation of serum xenin and ghrelin levels and their relationship with nonalcoholic fatty liver disease and insulin resistance in obese adolescents. J endocrinol İnvest 37(11):1091–1097
Carraway RE, Leeman SE (1976) Radioimmunoassay for neurotensin, agastrin immunoreactivity in gastric antral G-cells. Histochemistry hypothalamic peptide. J Biol Chem 251:7035–7044 (1986;85:135–138)
Kawanishi K, Goto A, Ishida T, Kawamura K, Nishina Y, Machida S, Yamamoto S, Ofuji T (1978) The effects of xenopsin of endocrine pancreas and gastric antrum in dogs. Horm Metab Res 10(4):283–286
Zinner MJ, Kasher F, Modlin IM, Jaffe BM (1982) Effect of xenopsin on blood flow, hormone release, and acid secretion. Am J Physiol 243(3):G195–G199
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group (2003) Consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 81:19–25 (revised)
Ferriman D, Gallwey JD (1961) Clinical assessment of body hair growth in women. J Clin Endocrinol Metab 21:1440–1447
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
Salley KE, Wickham EP, Cheang KI, Essah PA, Karjane NW, Nestler JE (2007) Glucose intolerance in polycystic ovary syndrome—a position statement of the Androgen Excess Society. J Clin Endocrinol Metab 92:4546–4556
Willis D, Franks S (1995) Insulin action in human granulosa cells from normal and polycystic ovaries is mediated by the insulin receptor and not the type-I insulin-like growth factor receptor. J Clin Endocrinol Metab 80:3788–3790
Nestler JE (1997) Role of hyperinsulinemia in the pathogenesis of the polycystic ovary syndrome, and its clinical implications. Semin Reprod Endocrinol 15:111–122
Nestler JE, Jakubowicz DJ, de Vargas AF, Brik C, Quintero N, Medina F (1998) Insulin stimulates testosterone biosynthesis by human thecal cells from women with polycystic ovary syndrome by activating its own receptor and using inositolglycan mediators as the signal transduction system. J Clin Endocrinol Metab 83:2001–2015
Chowdhury S, Wang S, Patterson BW, Reeds DN, Wice BM (2013) The combination of GIP plus xenin-25 indirectly increases pancreatic polypeptide release in humans with and without type 2 diabetes mellitus. Regul Pept 187:42–50
Kawanishi K, Goto A, Ishida T, Kawamura K, Nishina Y, Machida S, Yamamoto S, Ofuji T (1978) The effects of xenopsin of endocrine pancreas and gastric antrum in dogs. Horm Metab Res 10(4):283–286
Shaw C, Stöckmann F, Conlon JM (1987) Xenopsin- and neurotensin-like peptides in gastric juice from patients with duodenal ulcers. Eur J Clin Invest 17(4):306–312
Moti M, Amini L, Mirhoseini Ardakani SS, Kamalzadeh S, Masoomikarimi M, Jafarisani M (2015) Oxidative stress and anti-oxidant defense system in Iranian women with polycystic ovary syndrome. Iran J Reprod Med 13:373–378
Cochrane DE, Carraway RE, Boucher W (1991) Generation of xenopsin-related peptides from issue precursors by media conditioned by endotoxin-stimulated rat peritoneal macrophages. Inflammation 15(5):381–390
Carraway RE, Mitra SP, Muraki K (1991) Xenopsin-related peptide(s) are formed from xenopsin precursor by leukocyte protease(s) and cathepsinD. Peptides 12(1):107–112
Acknowledgements
This study did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector. This study was supported by Muzaffer Temur.
Author information
Authors and Affiliations
Contributions
MT, ÖY and SU project development, data collection, and manuscript writing. TÇ and SA did all the analyses, with major contribution and guidance about the statistical methods. EPÖ, MC prepared the figures and data collection. All authors contributed to the study by being part of the Steering Committee or as an international collaborative partner, critical analyses, and discussion of the data and to the revisions of the paper. All authors have read and approved the final version. The authors alone are responsible for the views expressed in the paper.
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Funding
None.
Conflict of interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Temur, M., Özün Özbay, P., Aksun, S. et al. Elevated circulating levels of xenopsin-related peptide-1 are associated with polycystic ovary syndrome. Arch Gynecol Obstet 296, 841–846 (2017). https://doi.org/10.1007/s00404-017-4493-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-017-4493-7