Abstract
Introduction
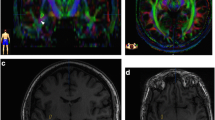
Optic radiations are tracts of particular interest for neurosurgery, especially for temporal lobe resection, because their lesion is responsible for visual field defects. However, histological and MRI studies found a high inter-subject variability of the optic radiation anatomy, especially for their most rostral extent inside the Meyer’s temporal loop. We aimed to better assess inter-subject anatomical variability of the optic radiations, in order to help to reduce the risk of postoperative visual field deficiencies.
Methods
Using an advanced analysis pipeline relying on a whole-brain probabilistic tractography and fiber clustering, we processed the diffusion MRI data of the 1065 subjects of the HCP cohort. After registration in a common space, a cross-subject clustering on the whole cohort was performed to reconstruct the reference optic radiation bundle, from which all optic radiations were segmented on an individual scale.
Results
We found a median distance between the rostral tip of the temporal pole and the rostral tip of the optic radiation of 29.2 mm (standard deviation: 2.1 mm) for the right side and 28.8 mm (standard deviation: 2.3 mm) for the left side. The difference between both hemispheres was statistically significant (p = 1.10–8).
Conclusion
We demonstrated inter-individual variability of the anatomy of the optic radiations on a large-scale study, especially their rostral extension. In order to better guide neurosurgical procedures, we built a MNI-based reference atlas of the optic radiations that can be used for fast optic radiation reconstruction from any individual diffusion MRI tractography.




Similar content being viewed by others
Data availability
This research was performed on the Wu-Minn HCP Database, an open-access MRI database available at https://www.humanconnectome.org/. All analyses were performed using the Ginkgo toolbox developed by the CEA/NeuroSpin team and freely available at https://framagit.org/cpoupon/gkg
References
Abed Rabbo F, Koch G, Lefèvre C, Seizeur R (2018) Stereoscopic visual area connectivity: a diffusion tensor imaging study. Surg Radiol Anat 40:1197–1208. https://doi.org/10.1007/s00276-018-2076-3
Allendorfer JB, Hernando KA, Hossain S, Nenert R, Holland SK, Szaflarski JP (2016) Arcuate fasciculus asymmetry has a hand in language function but not handedness. Hum Brain Mapp 37:3297–3309. https://doi.org/10.1002/hbm.23241
Avants BB, Epstein CL, Grossman M, Gee JC (2008) Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Med Image Anal 12:26–41. https://doi.org/10.1016/j.media.2007.06.004
Basser PJ, Mattiello J, LeBihan D (1994) MR diffusion tensor spectroscopy and imaging. Biophys J 66:259–267. https://doi.org/10.1016/S0006-3495(94)80775-1
Bürgel U, Schormann T, Schleicher A, Zilles K (1999) Mapping of histologically identified long fiber tracts in human cerebral hemispheres to the MRI volume of a reference brain: position and spatial variability of the optic radiation. Neuroimage 10:489–499. https://doi.org/10.1006/nimg.1999.0497
Campello RJGB, Moulavi D, Sander J (2013) Density-Based Clustering Based on Hierarchical Density Estimates. In: Pei J, Tseng VS, Cao L, Motoda H, Xu G (eds) Advances in Knowledge Discovery and Data Mining. Springer, Berlin, Heidelberg, pp 160–172
Catani M, Jones DK, Donato R, ffytche DH, (2003) Occipito-temporal connections in the human brain. Brain 126:2093–2107. https://doi.org/10.1093/brain/awg203
Chen X, Weigel D, Ganslandt O, Buchfelder M, Nimsky C (2009) Prediction of visual field deficits by diffusion tensor imaging in temporal lobe epilepsy surgery. Neuroimage 45:286–297. https://doi.org/10.1016/j.neuroimage.2008.11.038
Clatworthy PL, Williams GB, Acosta-Cabronero J, Jones SP, Harding SG, Johansen-Berg H, Baron J-C (2010) Probabilistic tractography of the optic radiations–an automated method and anatomical validation. Neuroimage 49:2001–2012. https://doi.org/10.1016/j.neuroimage.2009.10.083
Dayan M, Kreutzer S, Clark CA (2015) Tractography of the optic radiation: a repeatability and reproducibility study. NMR Biomed 28:423–431. https://doi.org/10.1002/nbm.3266
Descoteaux M, Angelino E, Fitzgibbons S, Deriche R (2007) Regularized, fast, and robust analytical Q-ball imaging. Magn Reson Med 58:497–510. https://doi.org/10.1002/mrm.21277
Dreessen de Gervai P, Sboto-Frankenstein UN, Bolster RB, Thind S, Gruwel MLH, Smith SD, Tomanek B (2014) Tractography of Meyer’s loop asymmetries. Epilepsy Res 108:872–882. https://doi.org/10.1016/j.eplepsyres.2014.03.006
Ebeling U, Reulen H-J (1988) Neurosurgical topography of the optic radiation in the temporal lobe. Acta Neurochir (Wien) 92:29–36. https://doi.org/10.1007/BF01401969
Guevara P, Poupon C, Rivière D, Cointepas Y, Marrakchi-Kacem L, Descoteaux M, Fillard P, Thirion B, Mangin J-F (2010) Inference of a HARDI fiber bundle atlas using a two-level clustering strategy. Med Image Comput Comput-Assist Interv MICCAI Int Conf Med Image Comput Comput-Assist Interv 13:550–557
James JS, Radhakrishnan A, Thomas B, Madhusoodanan M, Kesavadas C, Abraham M, Menon R, Rathore C, Vilanilam G (2015) Diffusion tensor imaging tractography of Meyer’s loop in planning resective surgery for drug-resistant temporal lobe epilepsy. Epilepsy Res 110:95–104. https://doi.org/10.1016/j.eplepsyres.2014.11.020
Jeelani NUO, Jindahra P, Tamber MS, Poon TL, Kabasele P, James-Galton M, Stevens J, Duncan J, McEvoy AW, Harkness W, Plant GT (2010) “Hemispherical asymmetry in the Meyer’s Loop”: a prospective study of visual-field deficits in 105 cases undergoing anterior temporal lobe resection for epilepsy. J Neurol Neurosurg Psychiatry 81:985–991. https://doi.org/10.1136/jnnp.2009.182378
de Jong L, Kovacs S, Bamps S, Van Calenbergh F, Sunaert S, van Loon J (2009) The arcuate fasciculus: a comparison between diffusion tensor tractography and anatomy using the fiber dissection technique. Surg Neurol 71:153. https://doi.org/10.1016/j.surneu.2008.10.072
Koch MA, Norris DG, Hund-Georgiadis M (2002) An Investigation of functional and anatomical connectivity using magnetic resonance imaging. Neuroimage 16:241–250. https://doi.org/10.1006/nimg.2001.1052
Lavrador JP, Ferreira V, Lourenço M, Alexandre I, Rocha M, Oliveira E, Kailaya-Vasan A, Neto L (2019) White-matter commissures: a clinically focused anatomical review. Surg Radiol Anat 41:613–624. https://doi.org/10.1007/s00276-019-02218-7
Lilja Y, Ljungberg M, Starck G, Malmgren K, Rydenhag B, Nilsson DT (2014) Visualizing Meyer’s loop: a comparison of deterministic and probabilistic tractography. Epilepsy Res 108:481–490. https://doi.org/10.1016/j.eplepsyres.2014.01.017
Ma J, Cheng L, Wang G, Lin S (2014) Surgical management of meningioma of the trigone area of the lateral ventricle. World Neurosurg 82:757–769. https://doi.org/10.1016/j.wneu.2014.05.026
Manji H, Plant G (2000) Epilepsy surgery, visual fields, and driving: a study of the visual field criteria for driving in patients after temporal lobe epilepsy surgery with a comparison of Goldmann and Esterman perimetry. J Neurol Neurosurg Psychiatry 68:80–82. https://doi.org/10.1136/jnnp.68.1.80
Monroy-Sosa A, Navarro-Fernández JO, Chakravarthi SS, Rodríguez-Orozco J, Rovin R, de la Garza J, Kassam A (2021) Minimally invasive trans-sulcal parafascicular surgical resection of cerebral tumors: translating anatomy to early clinical experience. Neurosurg Rev 44:1611–1624. https://doi.org/10.1007/s10143-020-01349-5
Nilsson D, Starck G, Ljungberg M, Ribbelin S, Jönsson L, Malmgren K, Rydenhag B (2007) Intersubject variability in the anterior extent of the optic radiation assessed by tractography. Epilepsy Res 77:11–16. https://doi.org/10.1016/j.eplepsyres.2007.07.012
Palejwala AH, O’Connor KP, Pelargos P, Briggs RG, Milton CK, Conner AK, Milligan TM, O’Donoghue DL, Glenn CA, Sughrue ME (2020) Anatomy and white matter connections of the lateral occipital cortex. Surg Radiol Anat 42:315–328. https://doi.org/10.1007/s00276-019-02371-z
Párraga RG, Ribas GC, Welling LC, Alves RV, de Oliveira E (2012) Microsurgical anatomy of the optic radiation and related fibers in 3-dimensional images. Neurosurgery 71:160–171. https://doi.org/10.1227/NEU.0b013e3182556fde
Pathak-Ray V, Ray A, Walters R, Hatfield R (2002) Detection of visual field defects in patients after anterior temporal lobectomy for mesial temporal sclerosis-establishing eligibility to drive. Eye Lond Engl 16:744–748. https://doi.org/10.1038/sj.eye.6700152
Peltier J, Travers N, Destrieux C, Velut S (2006) Optic radiations: a microsurgical anatomical study. J Neurosurg 105:294–300. https://doi.org/10.3171/jns.2006.105.2.294
Perrin M, Poupon C, Cointepas Y, Rieul B, Golestani N, Pallier C, Rivière D, Constantinesco A, Le Bihan D, Mangin JF (2005) Fiber tracking in q-ball fields using regularized particle trajectories. Inf Process Med Imaging Proc Conf 19:52–63. https://doi.org/10.1007/11505730_5
Peuskens D, van Loon J, Van Calenbergh F, van den Bergh R, Goffin J, Plets C (2004) Anatomy of the anterior temporal lobe and the frontotemporal region demonstrated by fiber dissection. Neurosurgery 55:1174–1184. https://doi.org/10.1227/01.neu.0000140843.62311.24
Rubino PA, Rhoton AL, Tong X, de Oliveira E (2005) Three-dimensional relationships of the optic radiation. Neurosurgery 57:219–227. https://doi.org/10.1227/01.neu.0000176415.83417.16
Shan Y-Z, Wang Z-M, Fan X-T, Zhang H-Q, Ren L-K, Wei P-H, Zhao G-G (2019) Automatic labeling of the fanning and curving shape of Meyer’s loop for epilepsy surgery: an atlas extracted from high-definition fiber tractography. BMC Neurol 19:302. https://doi.org/10.1186/s12883-019-1537-6
Sherbondy AJ, Dougherty RF, Napel S, Wandell BA (2008) Identifying the human optic radiation using diffusion imaging and fiber tractography. J Vis 8:12. https://doi.org/10.1167/8.10.12
Sivakanthan S, Neal E, Murtagh R, Vale FL (2016) The evolving utility of diffusion tensor tractography in the surgical management of temporal lobe epilepsy: a review. Acta Neurochir (Wien) 158:2185–2193. https://doi.org/10.1007/s00701-016-2910-5
Takaya S, Kuperberg GR, Liu H, Greve DN, Makris N, Stufflebeam SM (2015) Asymmetric projections of the arcuate fasciculus to the temporal cortex underlie lateralized language function in the human brain. Front Neuroanat 9:119. https://doi.org/10.3389/fnana.2015.00119
Taoka T, Sakamoto M, Nakagawa H, Nakase H, Iwasaki S, Takayama K, Taoka K, Hoshida T, Sakaki T, Kichikawa K (2008) Diffusion tensor tractography of the Meyer loop in cases of temporal lobe resection for temporal lobe epilepsy: correlation between postsurgical visual field defect and anterior limit of Meyer loop on tractography. AJNR Am J Neuroradiol 29:1329–1334. https://doi.org/10.3174/ajnr.A1101
Thiebaut de Schotten M, Ffytche DH, Bizzi A, Dell’Acqua F, Allin M, Walshe M, Murray R, Williams SC, Murphy DGM, Catani M (2011) Atlasing location, asymmetry and inter-subject variability of white matter tracts in the human brain with MR diffusion tractography. Neuroimage 54:49–59. https://doi.org/10.1016/j.neuroimage.2010.07.055
Thudium MO, Campos AR, Urbach H, Clusmann H (2010) The basal temporal approach for mesial temporal surgery: sparing the Meyer loop with navigated diffusion tensor tractography. Neurosurgery 67:385–390. https://doi.org/10.1227/NEU.0b013e3181f7424b
Van Essen DC, Smith SM, Barch DM, Behrens TEJ, Yacoub E, Ugurbil K, WU-Minn HCP Consortium (2013) The WU-Minn human connectome project: an overview. Neuroimage 80:62–79. https://doi.org/10.1016/j.neuroimage.2013.05.041
Yamamoto T, Yamada K, Nishimura T, Kinoshita S (2005) Tractography to depict three layers of visual field trajectories to the calcarine gyri. Am J Ophthalmol 140:781-785.e1. https://doi.org/10.1016/j.ajo.2005.05.018
Yogarajah M, Focke NK, Bonelli S, Cercignani M, Acheson J, Parker GJM, Alexander DC, McEvoy AW, Symms MR, Koepp MJ, Duncan JS (2009) Defining Meyer’s loop-temporal lobe resections, visual field deficits and diffusion tensor tractography. Brain J Neurol 132:1656–1668. https://doi.org/10.1093/brain/awp114
Acknowledgements
Data were provided by the Human Connectome Project, WU-Minn Consortium (Principal Investigators: David Van Essen and Kamil Ugurbil; 1U54MH091657) funded by the 16 NIH Institutes and Centers that support the NIH Blueprint for Neuroscience Research; and by the McDonnell Center for Systems Neuroscience at Washington University. This research has received funding from the European Union’s Horizon 2020 Framework Program for Research and Innovation under the specific Grant No. 945539 (Human Brain Project SGA3).
Funding
This research has received funding from the European Union’s Horizon 2020 Framework Program for Research and Innovation under the specific Grant No. 945539 (Human Brain Project SGA3). Authors disclose no other financial or non-financial interests that could be directly or indirectly related to the work submitted for publication.
Author information
Authors and Affiliations
Contributions
BH: Protocol/project development, Data collection or management, Data analysis, Manuscript writing. IUi: Protocol/project development, Data management, Data Analysis, Manuscript writing/editing. MC: Protocol/project development, Data analysis. CP: Protocol/project development, Manuscript writing/editing. SD: Protocol/project development, Manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Ethical approval
This research was performed on the Wu-Minn HCP Database, an open-access MRI database available at https://www.humanconnectome.org/. Subjects included had given their written consent to the Wu-Minn Consortium for their data to be published in open-access and reused for other studies. All authors approved the DataUse Terms from the Wu-Minn HCP Consortium, a guideline for accessing, using and performing analyses on this open-access database. We followed the French ethical guidelines and the current French legislation, i.e.: The World Medical Association Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects (https://www.wma.net/wp-content/uploads/2016/11/DoH-Oct2013-JAMA.pdf) and the French Public Health Law n°2004–806 (https://www.legifrance.gouv.fr/jorf/id/JORFTEXT000000787078/).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Herlin, B., Uszynski, I., Chauvel, M. et al. Cross-subject variability of the optic radiation anatomy in a cohort of 1065 healthy subjects. Surg Radiol Anat 45, 849–858 (2023). https://doi.org/10.1007/s00276-023-03161-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-023-03161-4