Abstract
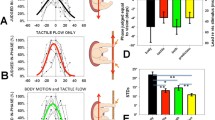
Self-motion perception is based on the integration of visual (optic flow) and vestibular (inertial) sensory information. Previous research has shown that the relative contribution of visual and vestibular cues can change in real time based on the reliability of that information. The present study assessed whether initial velocity and acceleration magnitude influence the relative contribution of these cues to the detection of self-acceleration. Participants performed a simple response time task with visual and vestibular self-acceleration cues as targets. Visual optic flow was presented at three possible initial velocities of 3, 9, or 15 m/s, and accelerated to result in three possible final velocities of 21, 27, or 33 m/s. Corresponding vestibular cues were presented at magnitudes between 0.01 and 0.04 g. The self-acceleration cues were presented at three possible stimulus onset asynchronies (SOAs): visual-first (by 100 ms), in-sync, and vestibular-first (by 100 ms). We found that presenting the cues in-sync resulted in the fastest responses across all velocities and acceleration magnitudes. Interestingly, presenting the visual cue first resulted in a relative advantage over vestibular-first at the slowest initial velocity of 3 m/s, and vice versa for the fastest initial velocity of 15 m/s. The fastest overall responses for visual-first and in-sync were observed at 9 m/s. The present results support the hypothesis that velocity of optic flow can alter the relative contribution of visual and vestibular cues to the detection of self-acceleration.




Similar content being viewed by others
References
Arshad Q, Ortega MC, Goga U, Lobo R, Siddiqui S, Mediratta S, Bednarczuk NF, Kaski D, Bronstein AM (2019) Interhemispheric control of sensory cue integration and self-motion perception. Neuroscience 408:378–387
Ash A, Palmisano S (2012) Vection during conflicting multisensory information about the axis, magnitude, and direction of self-motion. Perception 41:253–267
Barnett-Cowan M (2013) Vestibular perception is slow: a review. Multisensory Res 26(4):387–403
Barnett-Cowan M, Harris LR (2009) Perceived timing of vestibular stimulation relative to touch, light and sound. Exp Brain Res 198(2–3):221–231
Bengtsson H (2018). R.matlab: Read and Write MAT Files and Call MATLAB from Within R. R package version 3.6.2. https://CRAN.R-project.org/package=R.matlab
Berthoz A, Pavard B, Young LR (1975) Perception of linear horizontal self-motion induced by peripheral vision (linear vection) basic characteristics and visual-vestibular interactions. Exp Brain Res 23:471–489
Bertin RJ, Berthoz A (2004) Visuo-vestibular interaction in the reconstruction of travelled trajectories. Exp Brain Res 154:11–21
Brandt T, Dichgans J, Koenig E (1972) Perception of self-rotation (circular vection) induced by optokinetic stimuli. Pflugers Arch 332:R398
Butler JS, Smith ST, Campos JL, Bülthoff HH (2010) Bayesian integration of visual and vestibular signals for heading. J Vis 10(11):1–13
Butler JS, Campos JL, Bülthoff HH (2015) Optimal visual–vestibular integration under conditions of conflicting intersensory motion profiles. Exp Brain Res 233(2):587–597
Calderone JB, Kaiser MK (1989) Visual acceleration detection: effect of sign and motion orientation. Percept Psychophys 45(5):391–394
Chawla D, Buechel C, Edwards R, Howseman A, Josephs O, Ashburner J, Friston KJ (1999) Speed-dependent responses in V5: a replication study. NeuroImage 9:508–515
Cheng K, Hasegawa T, Saleem KS, Tanaka K (1994) Comparison of neuronal selectivity for stimulus speed, length, and contrast in the prestriate visual cortical areas V4, and MT of the macaque monkey. J Neurosci 71(6):2269–2280
Clemens IA, de Vrijer M, Selen LP, van Gisbergen JA, Medendorp WP (2011) Multisensory processing in spatial orientation: an inverse probabilistic approach. J Neurosci 31:5365–5377
Corey DP, Hudspeth AJ (1979) Response latency of vertebrate hair cells. Biophys J 26:499–506
Crane T (2012) Fore-aft translation aftereffects. Exp Brain Res 219(4):477–487
de Bruyn B, Orban GA (1988) Human velocity and direction discrimination measured with random dot patterns. Vision Res 28:1323–1335
de Winkel KN, Soyka F, Barnett-Cowan M, Bülthoff HH, Groen EL, Werkhoven PJ (2013) Integration of visual and inertial cues in the perception of angular self-motion. Exp Brain Res 231:209–218
de Vrijer M, Mendendorp WP, van Gisbergen JA (2009) Accuracy-perception trade-off in visual orientation constancy. J Vis 9:9–15
Drugowitsch J, DeAngelis GC, Klier EM, Angelaki DE, Pouget A (2014) Optimal multisensory decision making in a reaction time task. eLIFE 3:e03005
Fetsch CR, Turner AH, DeAngelis GC, Angelaki DE (2009) Dynamic reweighting of visual and vestibular cues during self-motion perception. J Neurosci 29(49):15601–15612
Fetsch CR, Pouget A, DeAngelis GC, Angelaki DE (2011) Neural correlates of reliability-based cue weighting during multisensory integration. Nat Neurosci 15:146–154
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd Edition. Thousand Oaks, CA https://tinyurl.com/carbook
Gallagher M, Choi M, Ferre ER (2020) Multisensory interactions in virtual reality: optic flow reduces vestibular sensitivity, but only for congruent planes of motion. Multisens Res 1:20. https://doi.org/10.1163/22134808-20201487
Gibson JJ, Olum P, Rosenblatt F (1955) Parallax and perspective during aircraft landings. Am J Psychol 68:372–385
Gottsdanker RM (1956) The ability of human operators to detect acceleration of target motion. Psychol Bull 53(6):477–487
Groen JJ (1956) The semicircular canal system of the organs of equilibrium-I. Phys in Med & Biol 1(2):103
Gu Y, Angelaki DE, DeAngelis GC (2007) A functional link between area MSTd and heading perception based on vestibular signals. Nat Neurosci 10(8):1038–1047
Gu Y, Angelaki DE, DeAngelis GC (2008) Neural correlates of multisensory cue integration in macaque MSTd. Nat Neurosci 11(10):1201–1210
Harris LR, Morgan MJ, Still AW (1981) Moving and the motion after-effect. Nature 293:139–141
Harris LR, Jenkin M, Zikovitz DC (2000) Visual and non-visual cues in the perception of linear self-motion. Exp Brain Res 135:12–21
Heerspink H, Berkouwer W, Stroosma O, van Paassen R, Mulder M, Mulder B (2005). Evaluation of vestibular thresholds for motion detection in the SIMONA research simulator. AIAA modeling and simulation technologies conference and exhibit, pp 6502.
Hlavačka F, Mergner T, Schweigart G (1996) Human self-motion perception during translatory vestibular and proprioceptive stimulation. Neurosci Lett 210(2):83–86
Israel I, Berthoz A (1989) Contribution of the otoliths to the calculation of linear displacement. J Neurophysiol 62:247–263
Jürgens R, Becker W (2006) Perception of angular displacement without landmarks: evidence for Bayesian fusion of vestibular, optokinetic, podokinesethetic, and cognitive information. Exp Brain Res 174:528–543
Karmali F, Lim K, Merfeld DM (2014) Visual and vestibular perceptual thresholds each demonstrate better precision at specific frequencies and also exhibit optimal integration. J Neurophysiol 111:2393–2403
Kassambara A (2019). ggpubr: 'ggplot2' Based Publication Ready Plots. R package version 0.2.3. https://CRAN.R-project.org/package=ggpubr
Keshavarz B, Berti S (2014) Sensory integration precedes the perception of vection as mirrored by the N2 component of the human event-related brain potential. Can J of Exp Psychol 68(4):283–284
Keshavarz B, Speck M, Haycock B, Berti S (2017) Effect of different display types on vection and its interaction with motion direction and field dependence. i-Perception:1–18.
Kovács G, Raabe M, Greenlee MW (2008) Neural correlates of visually induced self-motion illusion in depth. Cereb Cortex 18:1779–1787. https://doi.org/10.1093/cercor/bhm203
Lappe M, Bremmer F, van den Berg AV (1999) Perception of self-motion from visual flow. Trends Cogn Sci 3:329–336
Lepecq J-C, Giannopulu I, Mertz S, Baudonnière P-M (1999) Vestibular sensitivity and vection chronometry along the spinal axis in erect man. Perception 28:63–72
Liu JL, Newsome WT (2005) Correlation between speed perception and neural activity in the middle temporal visual area. J Neurosci 25(3):711–722
Loose R, Probst T (2001) Velocity not acceleration of self-motion mediates visual-vestibular interaction. Perception 30:511–518
Mach E (1875) Grundlinien der Lehre von den Bewegungsempfindungen. Engelmann, Leipzig, Germany
MacNeilage PR, Banks MS, DeAngelis GC, Angelaki DE (2010) Vestibular heading discrimination and sensitivity to linear acceleration in head and world coordinates. J Neurosci 30(27):9084–9094
Maruyama K, Kaneoke Y, Watanabe K, Kakigi R (2002) Human cortical responses to coherent and incoherent motion as measured by magnetoencephalography. Neurosci Res 44:195–205. https://doi.org/10.1016/S0168-0102(02)00129-3
McKee SP (1981) A local mechanism for differential velocity detection. Vision Res 21:491–500
McKee SP, Silverman GH, Nakayama K (1986) Precise velocity discrimination despite random variations in temporal frequency and contrast. Vision Res 26:609–619
Medline Jones GM, Young LR (1978) Subjective detection of vertical acceleration: a velocity-dependent response? Acta Oto-Laryngol 85(1-2):45-53
Mergner T, Schweigart G, Müller M, Hlavacka F, Becker W (2000) Visual contributions to human self-motion perception during horizontal body rotation. Arch Ital Biol 138:139–166
Monen J, Brenner E (1994) Detecting changes in one’s own velocity from the optic flow. Perception 23:681–690
Nakayama R, Motoyoshi I (2017) Sensitivity to Acceleration in the Human Early Visual System. Front Psychol 8:925
Ohmi M (1996) Egocentric perception through interaction among many sensory systems. Cogn Brain Res 5:87–96
Orban GA, DeWolf J, Maes H (1984) Factors influencing velocity encoding in the human visual system. Vision Res 24:33–39
Palmisano S, Allison RS, Pekin F (2008) Accelerating self-motion displays produce more compelling vection in depth. Perception 37(1):22–33
Rodman HR, Albright TD (1987) Coding of visual stimulus velocity in area MT of the macaque. Vision Res 27(12):2035–2048
Schlack A, Krekelberg B, Albright TD (2008) Speed perception during acceleration and deceleration. J Vis 8(8):9–9
Schmerler J (1976) The visual perception of accelerated motion. Perception 5(2):167–185
Soyka F, Bulthoff HH, Barnett-Cowan M (2013) Temporal processing of self-motion: modeling reaction times for rotations and translations. Exp Brain Res 228(1):51–62
Strasburger H, Rentschler I, Juttner M (2011) Peripheral vision and pattern recognition: a review. J Vis 11(5):13
Telford L, Howard IP, Ohmi M (1995) Heading judgments during active and passive self-motion. Exp Brain Res 104:502–510
ter Horst AC, Koppen M, Selen LPJ, Medendorp WP (2015) Reliability-based weighting of visual and vestibular cues in displacement estimation Ben Hamed S, ed. PLoS ONE 10:e0145015
Townsend B, Legere JK, O’Malley S, von Mohrenschildt M, Shedden JM (2019) Attention modulates event-related spectral power in multisensory self-motion perception. NeuroImage 191:68–80
Uesaki M, Ashida H (2015) Optic-flow selective cortical sensory regions associated with self-reported states of vection. Front Psychol 6:775
Vilhelmsen K, van der Weel FR, van der Meer ALH (2015) A high-density EEG study of differences between three high speeds of simulated forward motion from optic flow in adult participants. Front Syst Neurosci 9:146
Watamaniuk SN, Heinen SJ (2003) Perceptual and oculomotor evidence of limitations on processing accelerating motion. J Vis 3:698–709. https://doi.org/10.1167/3.11.5
Werkhoven P, Snippe HP, Toet A (1992) Visual Processing of Optic Acceleration. Vision Res 31(12):2313–2329
Wickham H (2017). Tidyverse: Easily Install and Load the 'Tidyverse'. R package version 1.2.1. https://CRAN.R-project.org/package=tidyverse
Zacharias GL, Young LR (1981) Influence of combined visual and vestibular cues on human perception and control of horizontal rotation. Exp Brain Res 41:159–171
Acknowledgements
The authors thank Dr. Hongjin Sun for discussions about the design of the experiment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests. Funding for this study was provided to J.M.S. and M.vM. by the Natural Sciences and Engineering Research Council of Canada (RGPGP-2014-00051) and the Canadian Foundation for Innovation (2009M00034). These funding sources were not involved in the study design, the collection, analysis and interpretation of data, in the writing of the report, and in the decision to submit this article for publication.
Additional information
Communicated by Melvyn A. Goodale.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Significance statement The present study contributes novel evidence to the emerging literature on how visual–vestibular integration can result in reliable self-motion perception.
Rights and permissions
About this article
Cite this article
Kenney, D.M., O’Malley, S., Song, H.M. et al. Velocity influences the relative contributions of visual and vestibular cues to self-acceleration. Exp Brain Res 238, 1423–1432 (2020). https://doi.org/10.1007/s00221-020-05824-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-020-05824-9