Abstract
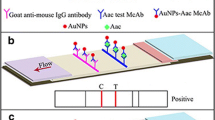
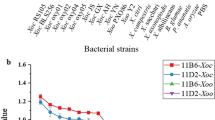
A rapid lateral flow immunochromatographic strip (ICS) using fluorescein isothiocyanate (FITC) labeled antigen and antibody was developed for the detection of Acidovorax citrulli (Ac) in melons and vegetable samples. In the ICS, signal amplification was realized based on antigen Ac and anti-Ac monoclonal antibody (McAb) 4F conjugated with FITC, respectively, which were forming two probes. The control line and the test line were obtained by immobilizing the goat anti-mouse IgG antibody and anti-Ac McAb 6D on both sides of the nitrocellulose membrane. The visual detection limit of the strip was 105 CFU/mL, which was 10-fold sensitive compared to the strip of FITC only labeling antigen or antibody. Signal amplification ICS was successfully applied to the detection of Ac in melon and vegetable samples with less detection time and operation procedures compared to the traditional enzyme-linked immunosorbent assay (ELISA) and PCR methods. This is the first report of using FITC labeled antigen and McAb as dual fluorescent probes to develop a direct-type immunofluorescence strip for the rapid and sensitive detection of Ac, which demonstrates a powerful tool for rapidly screening Ac in plant materials and other samples.

The schematic presentation of the test strip (a) and the positive result (b) or negative result (c) of the test strip







Similar content being viewed by others
References
Feng JJ, Li JQ, Walcott RR, Zhang GM, Luo LX, Kang L, et al. Advances in detection of Acidovorax citrulli, the causal agent of bacterial fruit blotch of cucurbits. Seed Sci Technol. 2013;41(1):1–15.
Rane KK, Latin RX. Bacterial fruit blotch of watermelon: association of the pathogen with seed. Plant Dis. 1992;76(5):761–5.
Webb RE, Goth RW. A seedborne bacterium isolated from watermelon. Plant Dis Rep. 1965;49:818–21.
Bahar O, Kritzman G, Burdman S. Bacterial fruit blotch of melon: screens for disease tolerance and role of seed transmission in pathogenicity. Eur J Plant Pathol. 2009;123(1):71–83.
Singh J, Sharma S, Nara S. Evaluation of gold nanoparticle based lateral flow assays for diagnosis of enterobacteriaceae members in food and water. Food Chem. 2015;170:470–83.
Ren W, Huang Z, Xu Y, Li Y, Ji Y, Su B. Urchin-like gold nanoparticle-based immunochromatographic strip test for rapid detection of fumonisin B1 in grains. Anal Bioanal Chem. 2015;407(24):7341–8.
Liu Y, Wu A, Hu J, Lin M, Wen M, Zhang X, et al. Detection of 3-phenoxybenzoic acid in river water with a colloidal gold based lateral flow immunoassay. Anal Biochem. 2015;483(1):7–11.
Zhou Y, Li YS, Meng XY, Zhang YY, Yang L, Li ZH, et al. Production of a monoclonal antibody and development of an immunoassay for detection of Cr(III) in water samples. Chemosphere. 2013;93(10):2467–72.
Zhang MZ, Wang MZ, Chen ZL, Fang JH, Fang MM, Liu J, et al. Development of a colloidal gold-based lateral-flow immunoassay for the rapid simultaneous detection of clenbuterol and ractopamine in swine urine. Anal Bioanal Chem. 2009;395(8):2591–9.
Byzova NA, Smirnova NI, Zherdev AV, Eremin SA, Shanin IA, Lei HT, et al. Rapid immunochromatographic assay for ofloxacin in animal original foodstuffs using native antisera labeled by colloidal gold. Talanta. 2014;119:125–1232.
Di Nardo F, Anfossi L, Giovannoli C, Passini C, Goftman VV, Goryacheva IY, et al. A fluorescent immunochromatographic strip test using quantum dots for fumonisins detection. Talanta. 2016;150:463–8.
Sirkka N, Lyytikäinen A, Savukoski T, Soukka T. Upconverting nanophosphors as reporters in a highly sensitive heterogeneous immunoassay for cardiac troponin I. Anal Chim Acta. 2016;925:82–7.
Anfossi L, Di Nardo F, Giovannoli C, Passini C, Baggiani C. Increased sensitivity of lateral flow immunoassay for ochratoxin A through silver enhancement. Anal Bioanal Chem. 2013;405(30):9859–67.
Liu D, Huang Y, Wang S, Liu K, Chen M, Xiong Y, et al. A modified lateral flow immunoassay for the detection of trace aflatoxin M1 based on immunomagnetic nanobeads with different antibody concentrations. Food Control. 2015;51:218–24.
Nagatani N, Yamanaka K, Ushijima H, Koketsu R, Sasaki T, Ikuta K, et al. Detection of influenza virus using a lateral flow immunoassay for amplified DNA by a microfluidic RT-PCR chip. Analyst. 2012;137(137):3422–6.
Liu Q, Liu XP, Wei YP, Mao CJ, Niu HL, Song JM, et al. Electrochemiluminescence immunoassay for the carcinoembryonic antigen using CdSe:Eu nanocrystals. Microchim Acta. 2017;184(5):1353–60.
Wang C, Wu J, Zong C, Ju H, Yan F. Highly sensitive rapid chemiluminescent immunoassay using the DNAzyme label for signal amplification. Analyst. 2011;136(20):4295–300.
Saniee P, Siavoshi F, Nikbakht BG, Khormali M, Sarrafnejad A, Malekzadeh R. Localization of H.pylori within the vacuole of Candida yeast by direct immunofluorescence technique. Arch Iran Med. 2013;16(12):705–10.
Yeo KB, Kim HB, Choi YS, Pack SP. Highly effective detection of inflamed cells using a modified bradykinin ligand labeled with FITC fluorescence. Enzyme Microb Technol. 2016;82:191–6.
Kim BS, Oh JM, Kim KS, Seo KS, Cho JS, Khang G, et al. BSA-FITC-loaded microcapsules for in vivo delivery. Biomaterials. 2009;30(5):902–9.
Murányi J, Gyulavári P, Varga A, Bökönyi G, Tanai H, Vántus T, et al. Synthesis, characterization, and systematic comparison of FITC-labelled GnRH-I, -II and -III analogues on various tumour cells. J Peptide Sci. 2016;22(8):552–60.
Blajman JE, Astesana DM, Zimmermann JA, Rossler E, Scharpen AR, Berisvil AP, et al. Quantification of FITC-labelled probiotic Lactobacillus salivarius DSPV 001P during gastrointestinal transit in broilers. Beneficial Microbes. 2017;8(1):55–64.
Zeng H, Guo W, Liang B, Li J, Zhai X, Song C, et al. Self-paired monoclonal antibody lateral flow immunoassay strip for rapid detection of Acidovorax avenae subsp. citrulli. Anal Bioanal Chem. 2016;408(22):6071–8.
Hu C, Chiang C, Hong P, Yeh M. Influence of charge on FITC-BSA-loaded chondroitin sulfate-chitosan nanoparticles upon cell uptake in human Caco-2 cell monolayers. Int J Nanomed. 2012;7(8):4861–72.
Himananto O, Luxananil P, Kumpoosiri P. Novel and highly specific monoclonal antibody to Acidovorax citrulli and development of ELISA-based detection in cucurbit leaves and seed. Plant Dis. 2011;95(95):1172–8.
Charlermroj R, Himananto O, Seepiban C, Kumpoosiri M, Warin N, Oplatowska M, et al. Multiplex detection of plant pathogens using a microsphere immunoassay technology. Plos One. 2013;8(4):e62344.
Bahar O, Efrat M, Hadar E, Dutta B, Walcott RR, Burdman S. New subspecies-specific polymerase chain reaction-based assay for the detection of Acidovorax avenae subsp. citrulli. Plant Pathol. 2008;57(4):754–63.
Ha Y, Fessehaie A, Ling KS, Wechter WP, Keinath AP, Walcott RR. Simultaneous detection of Acidovorax avenae subsp. citrulli and Didymella bryoniae in cucurbit seedlots using magnetic capture hybridization and real-time polymerase chain reaction. Phytopathology. 2009;99(6):666–78.
Zhao W, Lu J, Ma W, Xu C, Kuang H, Zhu S. Rapid on-site detection of Acidovorax avenae subsp. citrulli by gold-labeled DNA strip sensor. Biosens Bioelectron. 2011;26(10):4241–4.
Qu H, Zhang Y, Qu B, Kong H, Qin G, Liu S, et al. Rapid lateral-flow immunoassay for the quantum dot-based detection of puerarin. Biosens Bioelectron. 2016;81:358–62.
Acknowledgements
This research is supported by the Natural Science Foundation of China (No. 31371776), Science and Technology Innovation Plan of Shanghai: Yangtze River Delta joint research (No. 15395810900), Minimally Motivated Innovation Fund (YS30809102). and Graduate Education Innovation Program of Shanghai.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors confirm that no conflict of interest exists for this manuscript and all of the authors agree to the submission of this paper.
Rights and permissions
About this article
Cite this article
Zeng, H., Zhang, D., Zhai, X. et al. Enhancing the immunofluorescent sensitivity for detection of Acidovorax citrulli using fluorescein isothiocyanate labeled antigen and antibody. Anal Bioanal Chem 410, 71–77 (2018). https://doi.org/10.1007/s00216-017-0690-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0690-2